ORGO 2 lab Final
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Please select true below. this question is worth zero points:
True
For esters the C=O stretch occurs at:
1750 - 1730 cm^-1
A good separation on a TLC plate is achieved when each compound:
has a different Rf value
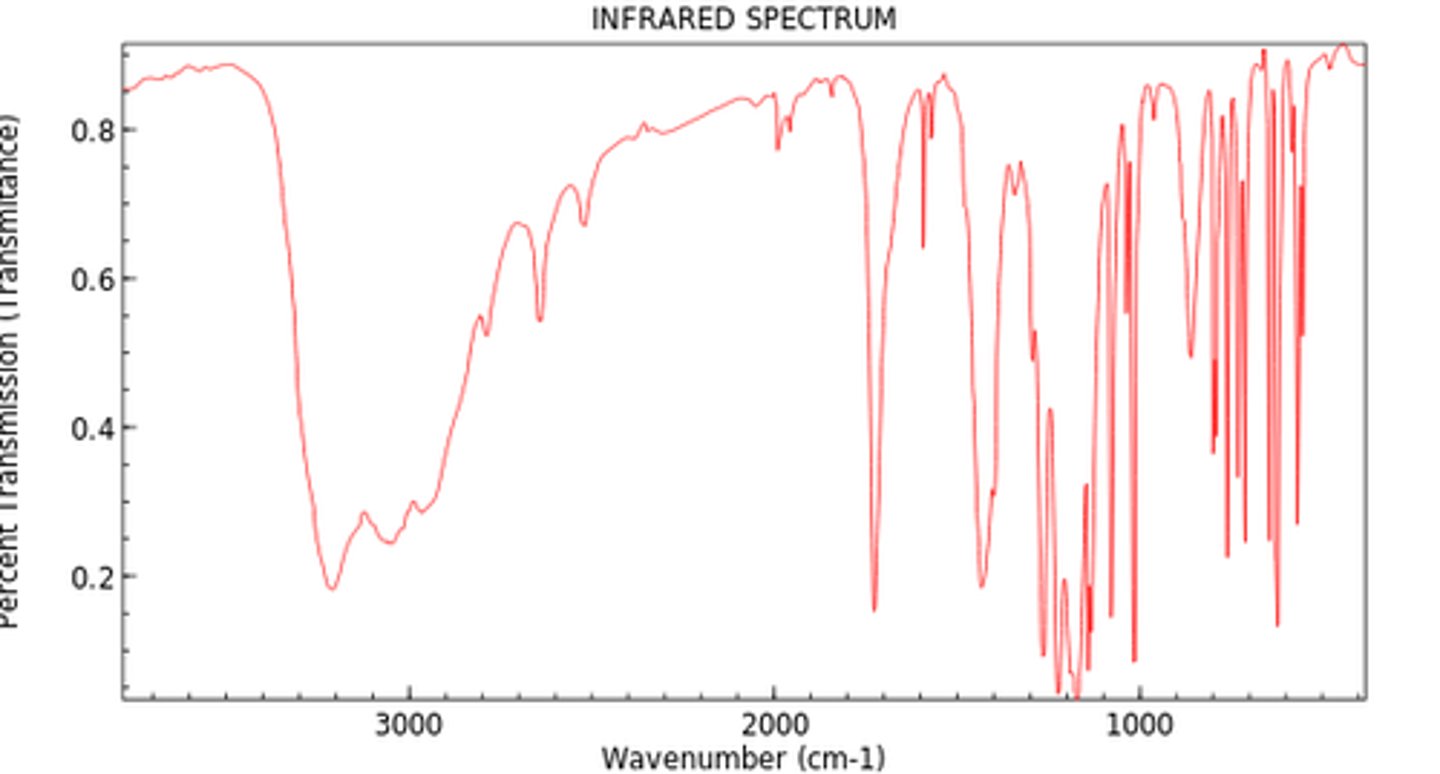
What functional group can be seen in the following IR spectra:
carboxylic acid
Rank the following compounds in order of decreasing speed of reactivity in an electrophilic aromatic substitution reaction:
3 > 1 >2
Select the correct mechanism to form a Grignard reagent
Which solvent(s) can be used for a Grignard reaction? select all that apply.
THF
diethyl ether
What way can radical chain growth polymerization terminate?
disproportionation, chain combination or reaction with an impurity
Step-growth polymers have three possible mechanisms; radical polymerization, cationic polymerization, or anionic polymerization
False
A pure solid sample will have a melting point range of approximately of 1 °C or less
True
What is missing from the box shown below
Acid
It is important that the melting point of the solid being recrystallized is above the boiling point of the recrystallization solvent to prevent "oiling out".
True
The best recrystallization solvent dissolves a large amount of your solid at high temperatures, and a small amount of your solid at low temperature
True
Grignard reagents can react with O2 and CO2 to form peroxides and carboxylic acids, respectively
True
In a Fischer esterification reaction, IR analysis can be used to determine whether or not the carboxylic acid has been converted to an ester, because we will see an O-H stretch in the product, but not in the starting material
False
The purpose of adding Na2SO4 to the ester products (from the Fischer esterification reaction) was to remove water
True
The first step of the Fischer esterification mechanism is the protonation of the carbonyl oxygen by an acid catalyst
True
During an electrophilic aromatic substitution reaction, the presence of an electron-donating group on the aromatic ring will _________ the reaction rate
Increase
(Nitration of methyl benzoate reaction)
It is important to add the methyl benzoate to the nitrating solution quickly to prevent the formation of side products.
False
The polymerization of this molecule, goes through three district phases. What are these phases - must be in the correct order
1 Initiation, 2 propagation, 3 termination
The name of this polymer is _________
Nylon 6,8
Using the given reagent bank, select the corresponding number for the missing reagent(s).
21 1,6-hexanediamine
What is the correct temperature for this reaction
0° C
Using the given reagent bank, select the corresponding number for the missing reagent(s).
19 Propanol and H2SO4
What is the correct solvent for this reaction ?
Diethyl ether
Using the given reagent bank, select the corresponding number for the missing reagent(s).
1 HNO3 and H2SO4
Using the given reagent bank, select the corresponding number for the missing reagent(s).
18 Methanol and H2SO4
What is the correct solvent for this reaction ?
0.5 M NaOH
What is the correct temperature for this reaction?
0° C
What is the correct first step in the mechanism for the nitration of methyl benzoate? select all that are correct
What is the correct second step in the reaction?? select all that are correct
Select the main product(s) formed in the nitration of methyl benzoate? select all that are correct.
During the polymer lab, we made Nylon 6,10 from a di-acid chloride and a diamine. This is an example of ___________ polymerization
18. Step-Growth
During an electrophilic aromatic substitution reaction, the rate-determining step involves the aromatic ring attacking a(n)
2. Electrophile
During the polymer lab we made slime from poly(vinyl alcohol) and borax. The strongest force responsible for cross0linking this polymer chains is:
21. Hydrogen-bonding
__________ contain the function group -COOR
25. Esters
is the following spectra methyl propionate, methanol or propionic acid?
methyl propionate
is the following spectra methyl propionate, methanol or propionic acid?
Methanol
is the following spectra methyl propionate, methanol or propionic acid?
propionic acid
Please select true below. this question is worth zero points:
True