Polarity
1/6
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Electronegativity
The ability of an atom to attract the bonding electron in a covalent bond
Non-Polar bonds
These occur between covalent bonds in diatomic gases as the atoms have equal electronegativity and so the bonded electrons are equally attracted to both nuclei.
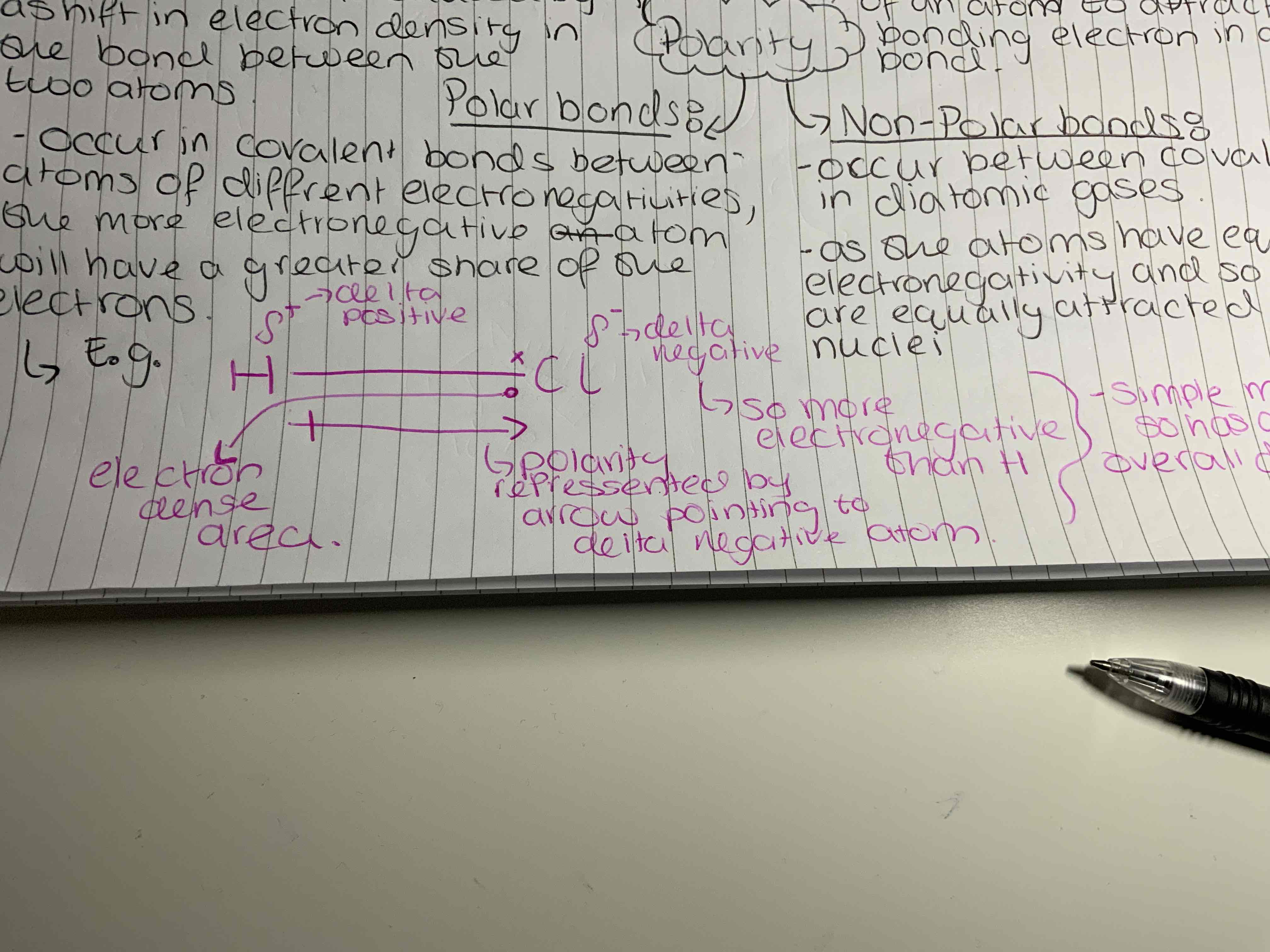
Polar bonds
Occur in covalent bonds between atoms of different electronegativities as the more electronegative atom will have a greater share of the bonded electrons.
Dipole
A difference in charge between two atoms caused by a shift in electron density in the bond between the two atoms.
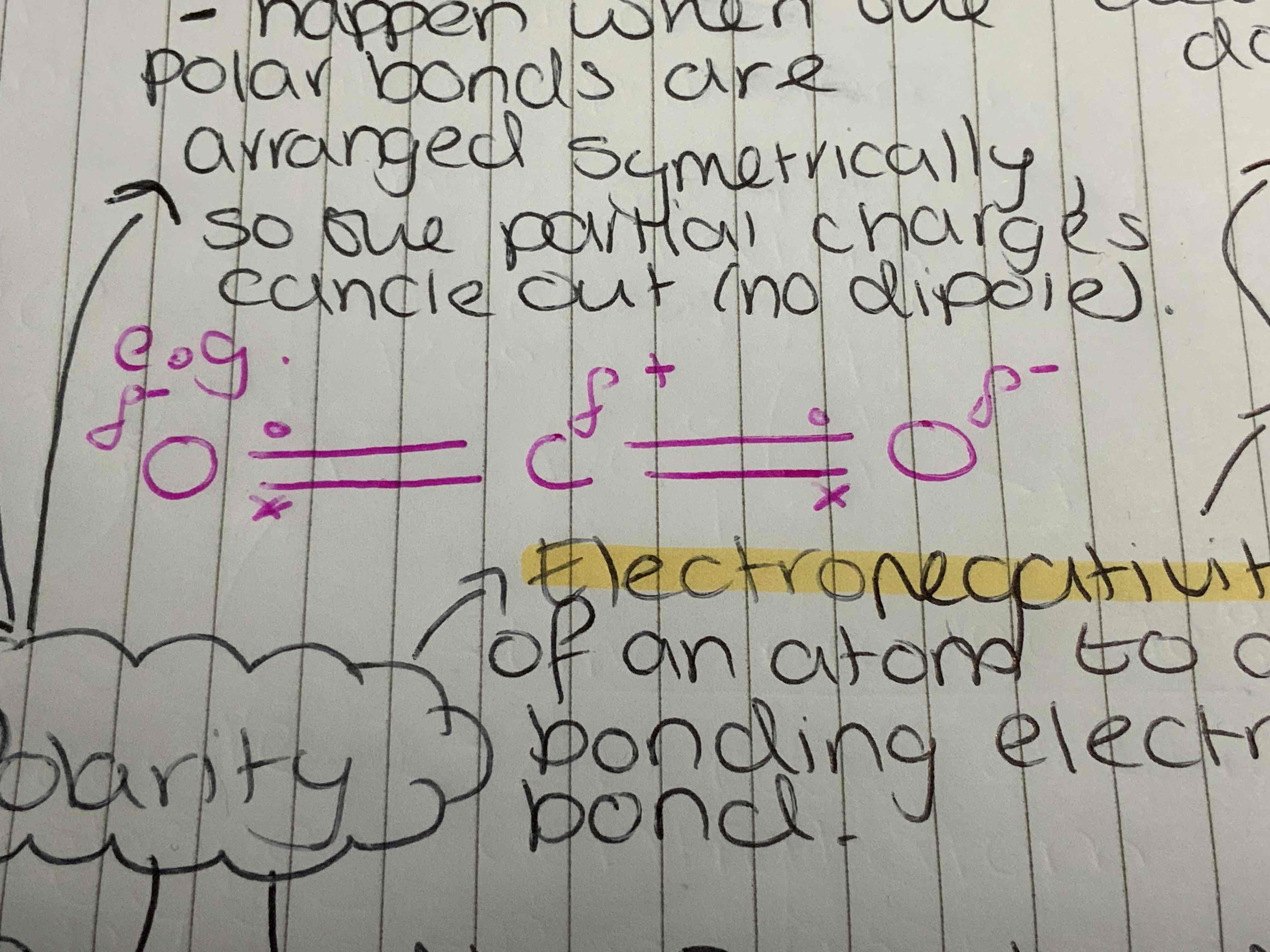
Non-Polar molecules
Happened when the polar bonds between atoms are arranged symmetrically so cancel out.

Polar molecules
Occur if the polar bonds between atoms are arranged asymmetrically so don’t cancel out.
Pauling scale
Measures the electronegativity of an atom. Tends to increase across periods and decrease down groups(ignoring the noble gases)