Chemistry OCR A
1/16
Earn XP
Description and Tags
random bits i get wrong
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
how to purify aromatic compounds SOLIDS
Dissolve in minimum volume of hot solvent
Allow to cool
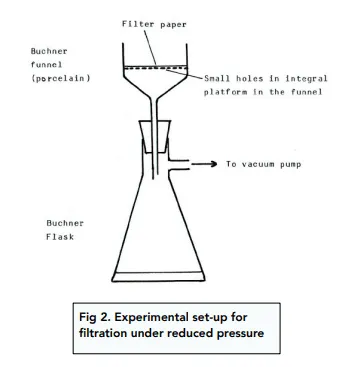
Filter under reduced pressure (buchner funnel)
wash with cold solvent and dry
scratch with glass rod - speeds up crystallisation
how to purify organic LIQUIDS
use separating funnel to separate organic layer from aqueous layer
wash with NaHCO3 to remove acidic impurities
add drying agent ( CaCl2 )
redistill at specific boiling point
what does heterolytic fission mean
the breaking of a covalent bond where both electrons from shared pair moves to ONE atom
what does homolytic fission mean
when a covalent bond breaks, each bonding atom receives one electron from the bonding pair, forming 2 radicals.
what are the naturally occurring elements you need to know
S8, P4
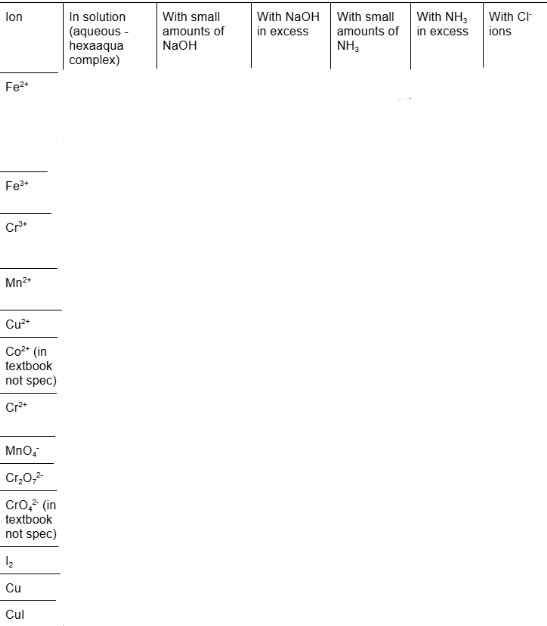
fill in blanks for colour of compounds
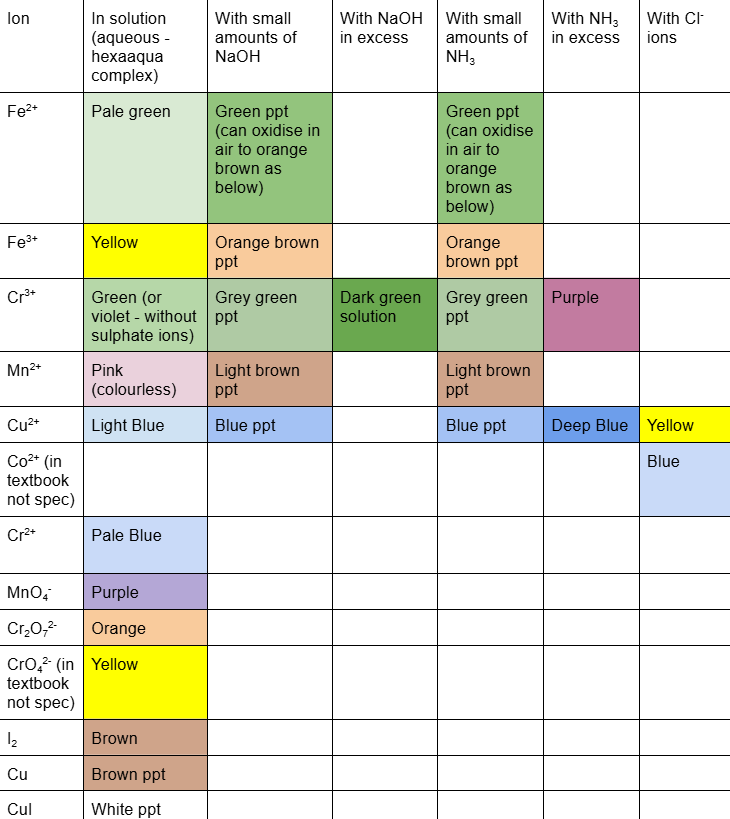
what do u add to hexaaqua structures to have ligand substitution
excess NaOH, Excess NH3, Cl- ions
happens with Cu, Cr
what compounds are used for Proton NMR
D2O- proton exchange ( for -OH -NH -SH)
CDCL3 - solvent
what compounds are used in CNMR
Si(CH3)4 - used as standard reference for calibration
CDCl3 - used as solvent (no protons to interfere w sample)
definition on a functional group
specific atom that is responsible for reactivity
why does stereoisomerism happen
different group on each carbon in double bond
double bond doesnt rotate
evidence for delocalised modol of benzene
all C-C bonds same length
enthalpy of hydrogenation of benzene is less exothermic than expected
describe diagram of filtration under reduced pressure
what are the reasons for a lower enthalpy change than expected in an experiment
incomplete combustion
heat loss to surroundings
evaporation of water
what are the colours of the halides in solution and organic
Cl2 + 2Br- → Br2 + 2Cl- (orange in cyclohexane)
Br2 + 2I- → I2 + 2Br- (purple in cyclohexane)

what transition metal forms square planar ions
Platinum
how to explain buffer formation
excess weak acid with Salt/strong base. partially neutralises. contains [acid] and [conjugate base] / [salt or H+]