OCR A A level foundations in chemistry flashcards
1/84
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
ionic bond definition
the strong electrostatic attraction between two or more oppositely charged ions
covalent bond definition
the elctrostatic attraction between between the bonding pair of electrons and their nuclei
what’s the maximum number of bonding pairs?
equivalent to the number of electrons in the outer shell.
valence
the measure of many electrons an atom can fit on its outer shell
dative bond
a covalent bond in which both electrons come from the same atom
MOLECULE- 2BP 0LP
linear, 180°
MOLECULE-3BP 0LP
trigonal planar, 120°
MOLECULE-4BP 0LP
tetrahedral, 109.5°
MOLECULE-6BP 0LP
octahedral, 90°
MOLECULE- 5BP 0LP
trigonal bipyramical, 90°, 120°
MOLECULE-3BP 1LP
trigonal pyramidal, 107°
MOLECULE- 2BP 2LP
non-linear/bent, 104.5°
order of repulsion of electrons most to least
lp/lp, lp/bp, bp/bp
electronegativity definition
the ability of an atom to attract the bonding electrons in a covalent bond
what increases electronegativity and why?
in a lower period because there’s less space therefore more attraction between the outer shell and the nucleus, higher group because it has more protons/positive charge to attract the electron pair
dipole definition
the separation of opposite partial charges in a molecule, represented by ẟ+/ẟ-
what determines whether a bond is ionic or covalent?
the difference in electronegativity. covalent is lower, then froms a dipole, then at a high enough difference it forms an ionic bond
average bond enthalpy definition
the amount of energy required to break one mole of bonds of a particular type between two atoms in a gaseous state, caulculated using a variety of different compounds
induced dipole-dipole interactions (london dispersion forces)
the weakest intermolecular force that exists between all molecules due to electron movement
simple molecular lattice
the structure simple covalent molecules form in the solid state. held in place by london forces
solubility of non-polar simple covalent compounds
soluble in non-polar solvents but not in polar solvents
solubility of polar simple covalent compounds
may dissolve in polar substances. doesn’t dissolve in non-polar substances.
conductivity of simple molecular structures
aren’t electrical conductors because there are no mobile charged particles. however some polar structures can ionise
hydrogen bond
a type of permanent dipole-dipole interaction found between H and O,N or F. strongest IM force
anomalous properties of water
solid less dense than liquid. high melting/boiling point. high surface tension
permanent dipole-dipole interactions
the intermolecular force between polar molecules because of their dipoles.
ionic lattice structure
ions arranged in a repeating pattern
solubility of giant covalent structures
generally don’t dissolve because the energy needed to overcome the covalent bonds is too great
conductivity of giant covalent strucutres
sometimes can conduct due to delocalised electrons
what is n=1 n=2 n=3
shell1 shell2 shell3
sub shells
the sum of the one or more orbitals making up the shell of an atom
how many sub shells does each shell have
n=1 has s, n=2 has s + p, n=3 has s + p + d etc
orbital definition
a region of space around the nucleus where an electron is likely to be found. can hold 2 electrons
s orbital shape
spherical
p orbital shape
dumbbell shape
what is in electron configuration
principal quantum no., sub shell, electron occupancy in sub shell
metallic bonding definition
the electrostatic attraction between positive metal ions and negative delocalised electrons
isotope definition
atoms of the same element with a different number of neutrons
relative isotopic mass definition
mass of an isotope relative to 1/12 of a carbon-12 atom
relative atomic mass
the average mass of an atom of an element, considering the isotopic abundances, relative to 1/12 a carbon 12 atom.
empirical formula
the simplest whole number ratio of atoms of each element present in a compound
anhydrous definition
a substance containing no water
hydrated definition
compounds containing water molecules
water of crystallisation meaning
water that is found in the crystalline framework of a metal or salt, but not directly bonded.
how many electrons can fill the first four shells?
2, 8, 18, 32 2n²
hydrochloric acid formula
HCl
sulphuric acid formula
H2SO4
nitric acid formula
HNO3
ethanoic acid formula
CH3COOH
sodium hydroxide formula
NaOH
potassium hydroxide formula
KOH
what is an acid
a proton donor aqueous solution
what is a base
a substance that reacts with an acid to form salt and water/ proton accepting species
what is an alkalai
any base that is soluble in water
what makes an acid strong/weak
how much the acid dissociates in water, releasing H+ ions. if all of its Hs become H+ ions it is strong and fully dissociated, if only some dissociate it is weak and partially dissociated.
what is neutralisation
the exothermic reactions between H+ and OH- to from water. also the reaction between acids and bases to form salts.
general oxidation no. of O
-2
general oxidation number of H
+1
oxidation no. of O in peroxide
-1
oxidation no. of H in metal hydrides
-1
oxidation no. for group 7(halides)
-1 usually but can be -1 to +7
oxidation group 6
-2 usually but can be -2 to +6
oxidation group 5
-3 usually but can be -3 to +5
oxidation no. group 2
+2
oxidation no. group 1
+1
oxidation no. fluorine
-1 always
chlorine oxidation no.
-1 except in compounds with O or F
O oxidation no. in F2O
+2
oxidation no. transition metals
whatever ionic charge it has, e.g Iron(II) is +2, or whatever is left
oxidation no. in group 4
whatevers left/+4
oxidation no. of elements in their natural forms
0
oxidation no. of ions
whatever the charge is (e.g Cl- is -1, Ca2+ is +2)
acid dissociation constant (Ka) meaning
a measure of the strength of an acid in solution. It represents the concentration of hydrogen ions (H+) in solution produced by an acid.
acid dissocation constant formula
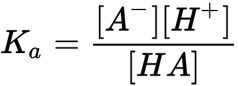
amount of substance definition
the measure of how many sets of 6.02×10²³, measured in mol
percentage yield
Percentage of product obtained compared to the maximum possible mass
Avogadro's constant
6.022×10²³
molar mass meaning
the mass in g of one mole of a particular substance
volume of 1 mol of any given gas at room temp and pressure
24dm³
molecular formula meaning
the number of atoms of each element in a compound
ideal gas equation
pV=nRT
(presure*volume=number of moles*gas constant*temperature)
Pa, m³, mol, 8.314, K
Atom economy
the percentage of desired product formed in a reaction over total products formed
why is it good to keep atom economy high?
environmentally sustainable, less waste product formed, less natural resources used
Oxidation definition
The loss of electrons or an increase in oxidation number
Reduction definition
The gain of electrons or the decrease in oxidation number