PHAR 203 - Gene Therapy
1/110
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
111 Terms
What is gene therapy?
a person’s genes are modified to treat or cure a disease by introducing foreign genetic material, such as RNA or DNA
Why develop gene therapies?
Rare genetic disorders have limited therapeutic options and lack early diagnosis, gene therapy treats these diseases at the root, and patients produce their own therapeutic gene product
What is the difference between enzyme replacement and gene augmentation
abnormal enzymes are non-functional and they’re treated by injecting purified enzyme into patient, which was produced by recombinant system
gene augmentation replace the mutant version with a new functional version that produces correct protein → healthy cell
There are two ways to deliver genetic material to patients, what are they?
In vivo and Ex vivo
What is in vivo way to deliver genetic material to patients
direct administration of a vector that has a genetic material
What is ex vivo way to deliver genetic material to patients
extract patient cells
genetic modification by a vector carrying a therapeutic transgene
selection and expansion in culture
infusion to reintroduce the engineered cells back into patient
Do Genetic Materials need a delivery vector and why?
yes, because nucleic acids are large and negatively charged, they poorly accumulate and penetrate into deeper tissues, biological enzymes rapidly degrade naked RNA / DNA molecules, and rapid clearance by immune cells.
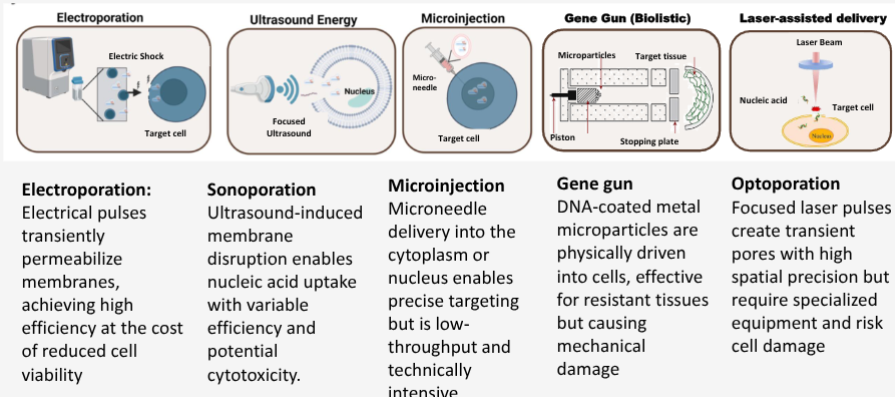
What are the physical methods of gene delivery types (5)
electroporation
sonoporation
microinjection
gene gun
optoporation
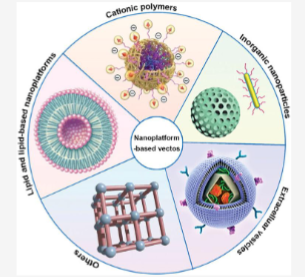
What are the non-viral methods of gene delivery (5)
Lipid based Nanoparticles (LNPs)
Lipid—Polymer Hybrid Nanoparticles
Extracellular vesicles
inorganic nanoparticles
cationic nanoparticles
Gene gun (biolistics) delivery involved coating DNA onto metal particles and firing them onto tissue. Which metal is most commonly used, and why is particle size a critical parameter for this technique
Gold or tungsten particles are used; particle size must be 0.6-2 microm to penetrate the cell membrane
What are the viral methods of gene delivery types (4)
retrovirus (8 kb)
lentivirus (9-10 kb)
AAV (adeno-associated virus) (4.7 kb)
adenovirus (36 kb)
What is the expression of retrovirus
stable
What is the expression of lentivirus
stable
what is the expression of AAV
transient or stable
what is the expression adenovirus
transient
does the retrovirus have target cell genome integration
yes
does the lentivirus have target cell genome integration
yes
does the AAV have target cell genome integration
No
Recombinant AAV has low frequency of host genome integration
does the adenovirus have target cell genome integration
No
What are the characteristics of retrovirus
high risk of insertional mutagenesis
can only target dividing cells
what are the characteristics of lentivirus
can trigger immune reaction
can target dividing and non-dividing cells
what are the characteristics of AAV
small cargo size, sustained expression
need a helper lipid to infect
can target dividing and non-dividing cells
what are characteristics of adenovirus
highly immunogenic
short expression duration / transient
does not integrate with host genome
What is an AAV?
A small, non-pathogenic virus used as a vector in gene therapy
What is the size of AAV?
18-26 nm in diameter
What kind of genome does AAV contain?
non enveloped
icosahedral capsid
Does AAV cause disease?
No, AAV is non-pathogenic (doesn’t cause disease)
Can AAV replicate independently?
No, It needs a helper virus for replication
Which viruses help AAV replicate?
adenovirus
herpesvirus
vaccinia virus
Why is AAV widely used in gene therapy?
Because it is:
safe (non-pathogenic)
low immune response
can infect many cell types
What does it mean that AAV has a broad host range?
It can infect many different cell types, making it versatile
What are AAV serotypes
variants with different surface proteins, leading to different tissue targeting
Why are different AAV serotypes useful?
they allow targeting of specific tissues (tissue tropism)
What is tissue tropism?
the ability of virus to target specific cell types or tissues
What does minimal immunogenicity mean?
it causes little immune response, making it safer for therapy
Summarize AAV in one line
AAV is a safe, non-pathogenic ssDNA virus with broad host range and low immunogenicity, making it idea for gene therapy
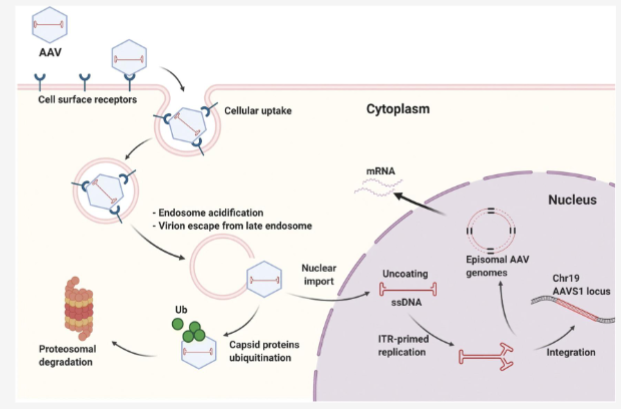
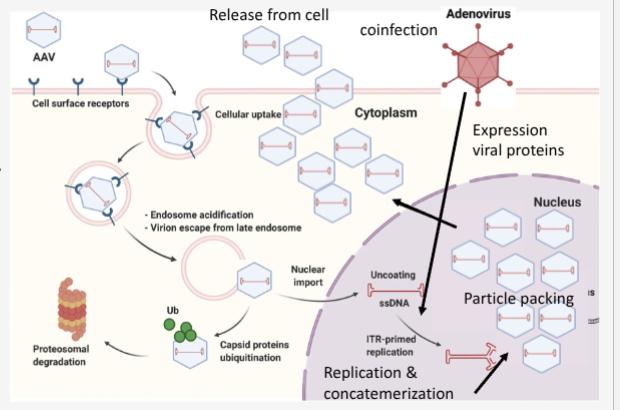
Walk through AAV lysogenic lifecycle
AAV binds receptor
endocytosed into cell
endosomal escape by degradation and transit to nucleus
coat removal
second strand DNA synthesis
Integration into host genome (AAVS1 site on Chr 19)
Viral DNA as Episomal DNA
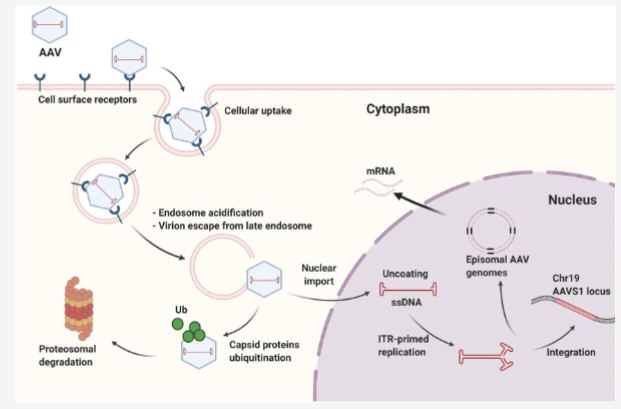
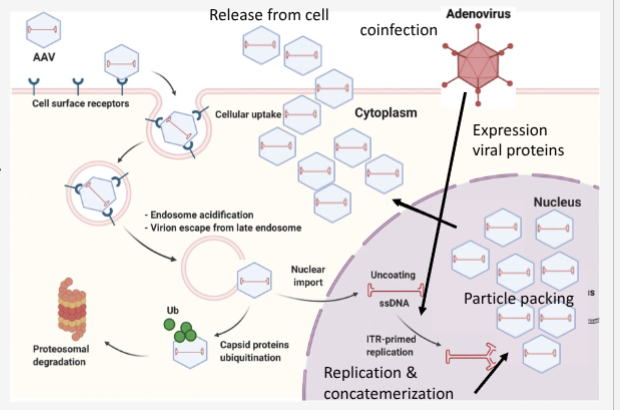
Walk through AAV lytic lifecycle
AAV binds to receptors
endocytosed into cell
endosomal escape
coat removal
second strand DNA synthesis
Co-infection with helper virus (specific gene products needed to express AAV replication and capsid proteins)
DNA replicates
Capsid made
virions assemble
release
What is the first step in creating an AAV gene therapy vector
the viral genome is cloned
What is done to the original AAV viral genes
they are removed to prevent viral replication and pathogenicity
What is inserted into the AAV genome
the target (therapeutic) genes
why are viral genes removed from AAV
to make the virus safe and unable to replicate on its own
What is done after the recombinant DNA is created?
it is used to transform bacterial cells for amplification
Why are bacteria used in this process?
to replicate and produce large amounts of plasmid DNA
why are helper virus genes needed
they provide functions required for AAV replication and packaging
What is the plasmid used for in AAV gene therapy?
A DNA vector carrying:
therapeutic gene
necessary regulatory elements
What happens when AAV infects target cells
the DNA can integrate into the genome (or persist episomally)
What are recombinant AAV (rAAV) particles?
Engineered AAV viruses that carry the therapeutic gene
Can recombinant AAV replicate on its own?
No it can’t replicate independently
Why are rAAV vectors considered safe?
because they
lac viral genes
can’
Why is AAV called a biological nanoparticles?
Because it is a tiny, engineered particle used to deliver genes into cells
What is the overall process of AAV gene therapy
clone AAV genome
remove viral genes
insert therapeutic gene
amplify DNA in bacteria
provide helper virus functions
produce rAAV particles
deliver gene to target cells
Which of the following best explains why AAV-based gene therapy is generally administered as a single lifetime dose, and what is the main obstacle to re-administation if a booster dose were needed?
AAV integrates into every cell of the target tissue simultaneously, providing permanent genetic correction.
3 Major Limitations of AAV-mediated Gene Therapy
high cost of treatment
high cost of viral vector production & purification
ineffective re-administration due to vector - neutralizing antibodies
Which limitation is most characteristic of physical transfection methods when compared to chemical nanoparticle-based delivery?
A. Low transfection efficiency
B. High dependence on endocytic pathways
C. Reduced scalability and increased cell damage
D. Poor Nucleic acid protection from degradation
Nucleic acids are
genetic materials used for gene therapy
Nucleic acids have (3)
high specificity
functional diversity
limited toxicity
What are polyplexes in gene delivery?
Complexes formed between cationic polymers and nucleic acids
Why are cationic polymers used in gene delivery?
They are positively charged, so they bind to negatively charged DNA/RNA
What is an example of cationic polymers used for gene delivery?
Diethylaminoethyl (DEAE)-dextran
Which cationic polymer is considered one of the most efficient for gene delivery?
Polyethyleneimine (PEI)
What is a major bottleneck of cationic polymers Gene delivery systems
toxicity and low efficiency
Why do toxicity and low efficiency limit clinical translation?
they reduce safety and effectiveness, making them less suitable for use in patients
What are recent advancements in cationic polymers design?
Development of
biodegradable polymers
bio-reducible polymers
stimuli-responsive polymer
targeted polymers
What is the goal of designing new types of cationic polymers
to reduce toxicity and improve delivery efficiency and specificity
What makes up the building block of living cells?
Lipids
What is a phase transition temperature (Tm) of phospholipids
the temperature that a lipid membrane goes from solid gel to fluid liquid
Tm (phase transition temperature of phospholipids) can be measured by
differential scanning calorimetry (DSC)
What happens to Tm (phase transition temperature of phospholipids) when there is an increased chain length → saturated chain
Tm increases
Lipid self assembly is governed by
molecular geometry and charge
Lipid shape depends on
headgroup size and hydration
fatty chain volume
electrostatic interactions
Formula of Critical Packing Parameter (CPP)

Which factor would most increase the transition temperature (Tm) of a phospholipid
A. increasing the number of double bonds
B. shortening fatty acid chain length
C. Increasing saturation of fatty acid chains
D. Introducing cis double bonds
C. Increasing saturation of fatty acid chains
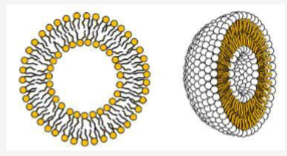
Liposomes are often consisting of
phospholipids and cholesterol
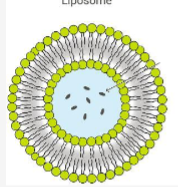
Describe liposomes
lipid bilayer made of ampipathic molecule surrounding an aqueous core, they’re neutral and has poor encapsulation
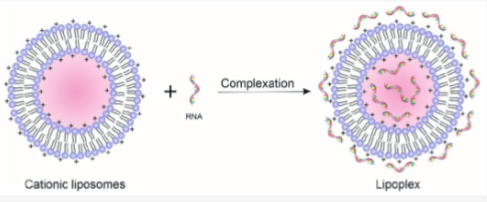
Describe the lipoplex
Lipoplex is formed by electrostatic interactions between positive and negatively charged nucleic acids, condenses into small particles when mixing.
What are the limitations of Lipoplexes
aggregation with serum proteins
cleared rapidly by immune cells
unencapsulated nucleic acid = activated immune system
less tolerability in vivo
3 examples of cationic lipids used in lipoplexes
DOTMA
DOTAP
DSTAP
Lipid nanoparticles are composed of 4 different components, each having a critical role in determining size, entrapment efficiency, stability and in vivo fate, what are these 4 components
ionizable cationic lipids
PEG lipid
Cholesterol
Helper lipids
Function of ionizable cationic lipids in LNPs
ionizable cationic lipids protonate at acidic pH, bind to nucleic acids and are neutral at physiological pH
Function of PEG Lipids in LNPs
PEG have stealth properties, minimize opsonization by serum proteins
Function of cholesterol in LNPs
cholesterol enhances stability and aid in transfection
Function of Helper lipids in LNPs
Helper Lipids promote cell binding, encapsulation efficiency and endosomal escape
Benefits of PEGylation (Shielding effect)
increase particle stability
increase circulation time
decrease particle side
What does the Shielding effect of PEGylation do
the shielding effect can decrease cell uptake
Anti-PEG antibodies may lead to
reduced efficiency
Two LNP formulations have the same size, but only one results in high protein expression. What ionizable lipid property most likely explains this difference?
A. the charged ionizable headgroup
B. Length of the hydrophobic lipid tail
C. Optimized pKa and unsaturated tail
D. Increased carbon saturation in the tail
C. Optimized pKa and unsaturated tail
Because they escape endosome → high protein expression
First, the LNPs are tested on animal (Preclinical studies) - Human trials, what is the process when we wanna study about LDL
LNP administered IV to mouse
Accumulation of LNP in hepatocytes
Mechanistic study = show interaction between LNPs and LDL receptors
How nanoparticles are taken up inside cells
Endocytosis
cell taking up LNPs (engulf)
What is the first step in LNP uptake after IV administration
rapid release of PEG-lipids from LNP particle
Why is PEG release important for LNP function
allows binding of ApoE to the LNP
What is ApoE
an exchangeable apolipoprotein found on lipid particles like VLDL (very-low-density lipoproteins)
What types of particles can ApoE bind to
endogenous particles (lipoprotein)
exogenous particles (LNPs)
What is Step 2 of LNP uptake?
Extravasation into the liver from circulation
What properties allow LNPs to enter the liver barrier?
small size (<100nm)
neutral charge
What happens in Step 3 of LNP uptake
ApoE-coated LNPs bind to LDL receptor on hepatocytes
What is the result of ApoE binding to LDL receptors
Receptor mediated endocytosis of LNPs
What cellular structure forms after LNP uptake?
endosome (intracellular vesicle)
What happens to pH inside the endosome?
it decreases (pH < 6.2) → becomes acidic
What happens to ionizable lipids in acidic endosomes?
they become protonated (positively charged)