College Biology Final
1/238
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
239 Terms
cell respiration definition
the process in which energy stored in food molecules is transferred to ATP
lewis acids
release electrons
lewis bases
absorb electrons
important lewis acids and bases
NAD+ --> NADH
FAD --> FADH2
two types of cell respiration
anaerobic and aerobic
anaerobic respiration
does not need oxygen, most ancient form, came before aerobic, 2 forms (glycolysis and fermentation)
glycolysis
1 glucose (6Cs) + 2 ATP --> 2 pyruvic acids (3Cs) + 4 ATP
-takes place in the cytoplasm
-2 ATP profit
-breaks glucose in half
fermentation
1 pyruvate (3Cs) --> 2C molecule waste products* + 1 ATP
-an add on to glycolysis
-used by yeast and some bacteria
*acetic acid (bacteria) or ethanol (yeast)
aerobic respiration
requires oxygen, occurs in mitochondria
four steps in aerobic respiration
oxidative decarboxylation, Krebs cycle, electron transport chain, chemiosmosis
CoA
coenzyme a
oxidative decarboxylation
2 pyruvates + 2NAD+ + 2CoA --> 2CO2 + 2 acetyl-CoA + 2NADH
Krebs cycle (citric acid cycle)
2 acetyl-CoA + 4NAD+ + 2FADH --> 4CO2 + 2ATP + 4NADH + 2FADH2
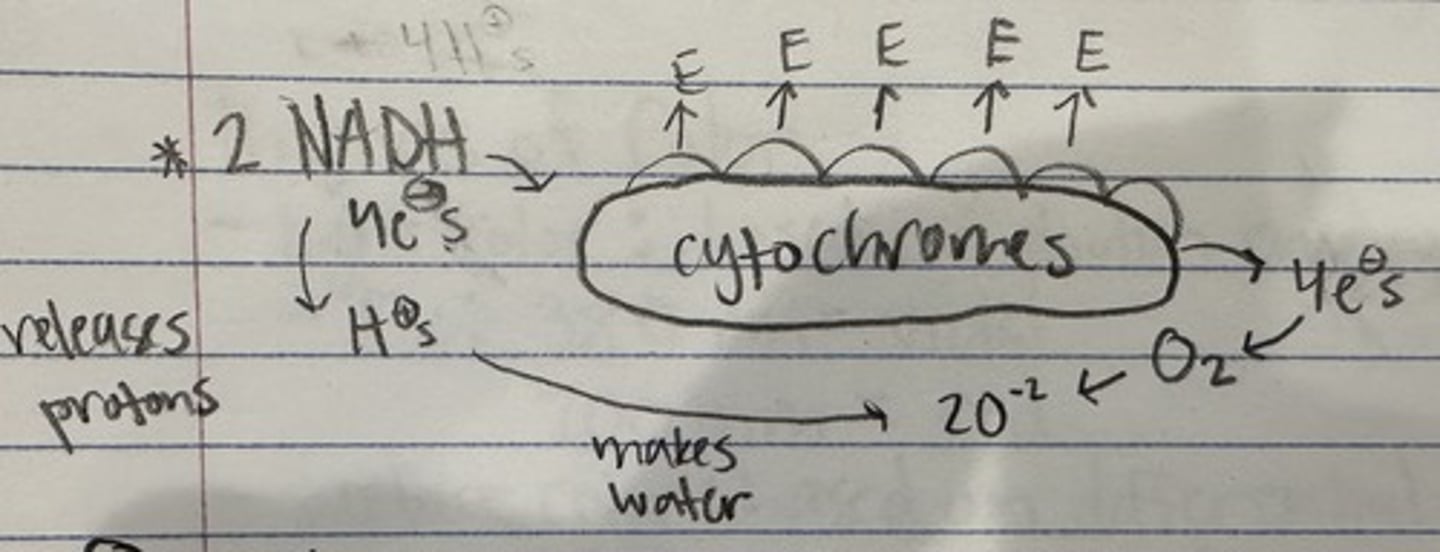
electron transport chain
-the basis is an enzyme complex called a cytochrome
(NADH or FADH2) + O2 --> electrons --> cytochromes --> low energy electrons + H2O
electron transport chain diagram
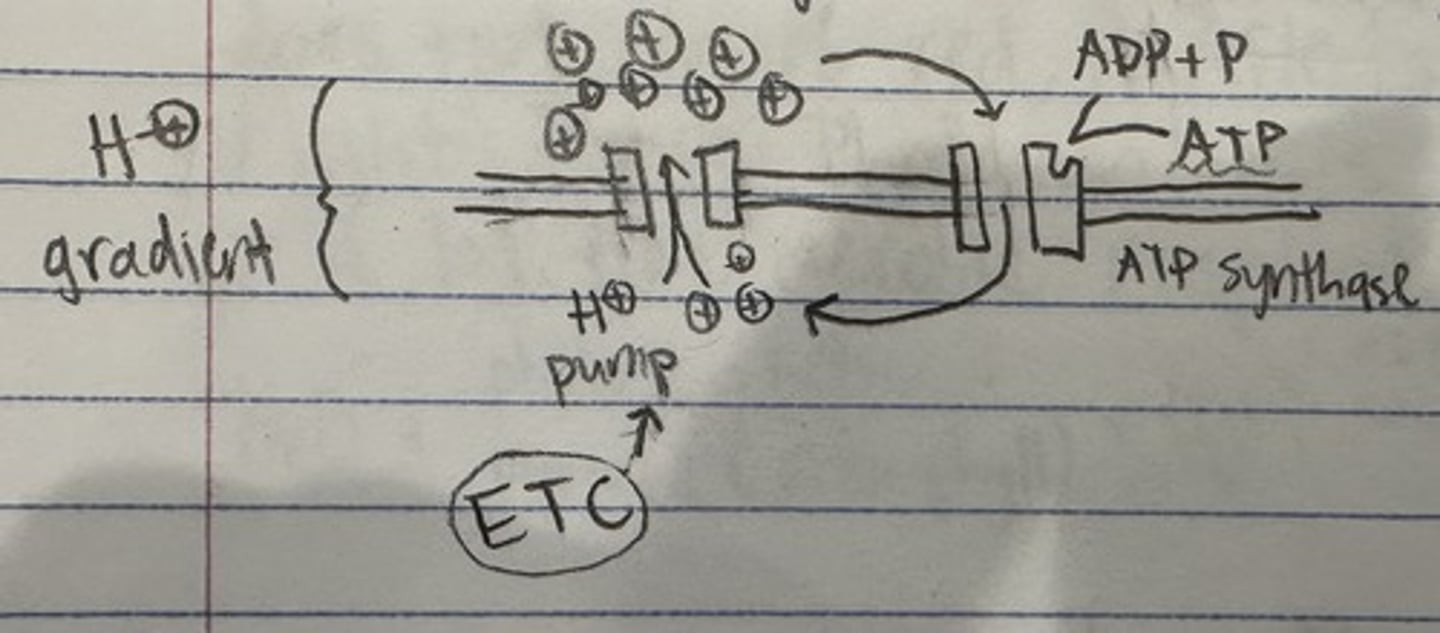
chemiosmosis
-process where bulk of ATP is produced
-occurs along the membrane folds of mitochondria
-2 active factors (H+ pumps and ATP synthase)
ATP synthase
proton gate, always open, binds ADP + P to create ATP
chemiosmosis diagram
photosynthesis stages
light phase and dark phase
light color
based on photon energy level, Ex: 700 nm = red
surface color
based on differential absorption of light by a surface, black absorbs all colors, white reflects all colors, other colors absorb everything but themselves
light phase photosynthesis
H2O + chlorophyll + light + ADP + P + NADP+ --> ATP + NADPH + O2
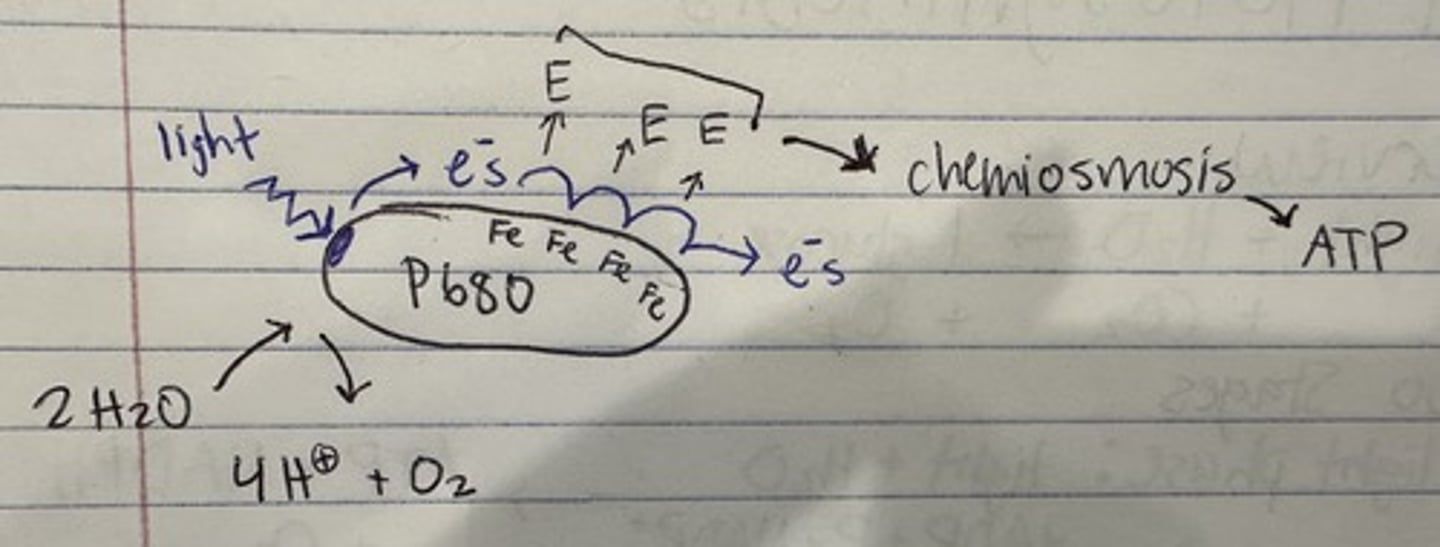
two stages of light phase
photosystem II and photosystem I
photosystem II
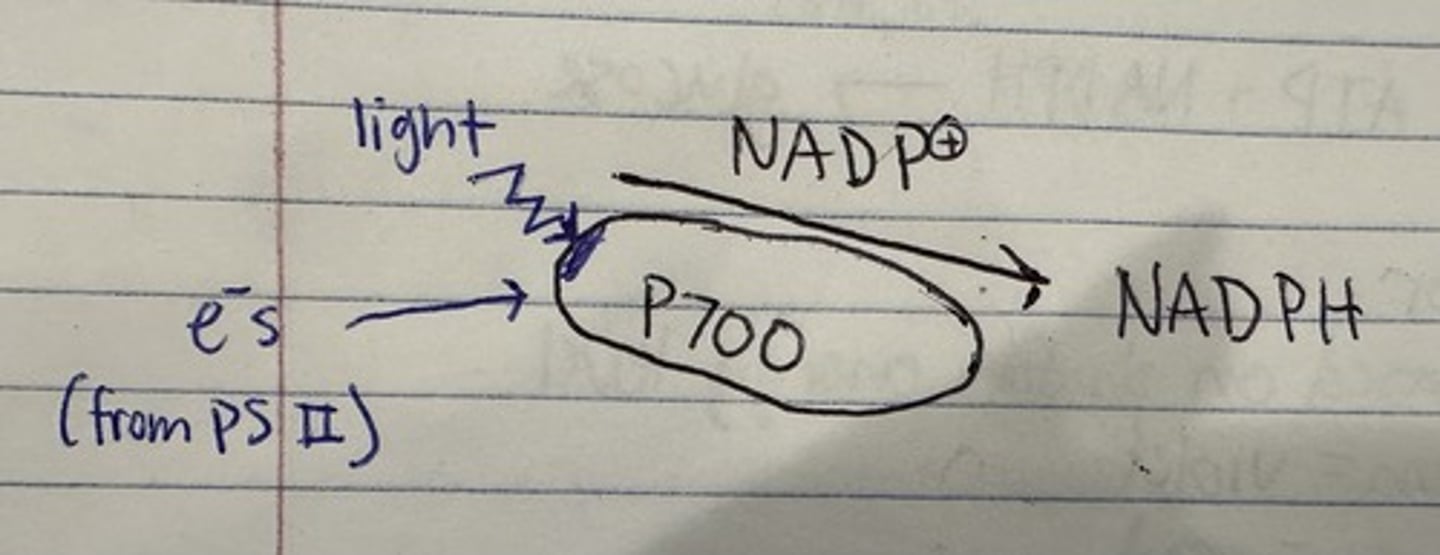
photosytem I
dark phase photosynthesis
6 CO2 + 6H2O + ATP + NADPH ---> glucose
pathways in dark phase photosynthesis
C3 (calvin cycle), C4, CAM (found in cacti)
C3 vs. C4 plants
C3 plants- cool weather grass, woody plants, some grains (wheat, barley, oats, rye)
-high water and CO2 needs
C4 plants- tropical or warm weather grass, palm trees, tropical grains (rice, corn, sorgum, millet)
-water efficient and low CO2 needs
electronegativity
the tendency for atoms to draw electrons toward themselves
electronegativity and covalent bonds
some atoms will "grab" the electron more in a covalent bond, some share it equally
polar molecule
have an uneven charge distribution while being electrically neutral, formed when atoms show strong electronegativity differences
non polar molecules
have even charge distributions, formed when atoms have similar electronegativity
ion
a charged atom or molecule
nucleus in an atom
contains protons and neutrons
atomic number
number of protons in an atom
atomic weight
protons plus neutrons
chemical bonding
process by which atoms becomes physically attached through electron exchange or sharing (involves valence electrons)
ionic bonding
transfer of electrons
covalent bonding
sharing of electrons
hydrogen bonding
a weak charge attraction between positive and negative charges
solute
a substance that is dissolved in a solute
solvent
the substance in which the solute is dissolved
reference solvent for polar molecules
water (H20)
reference solvent for non polar molecules
liquid fats (oils)
hydrophilic
loves water (polar)
hydrophobic
hates water (non polar)
acid
any molecule that releases a proton (H+) in water solution
base
any molecule that absorbs a proton in water solution
pH
a measure of the acidity or alkalinity of a water solution
pH 7
neutral
pH < 7
acidic
pH > 7
basic
buffer
any molecule that can act as an acid or base, minimizes pH shifts in solutions
definition of organic chemistry
the chemistry of carbon, hydrogen, and any atom that can replace a hydrogen around a carbon
number of bonds in a carbon containing molecule
four
hydrocarbons
nonpolar, consist entirely of C and H, building blocks of all other organic molecules
hydrocarbons functional group
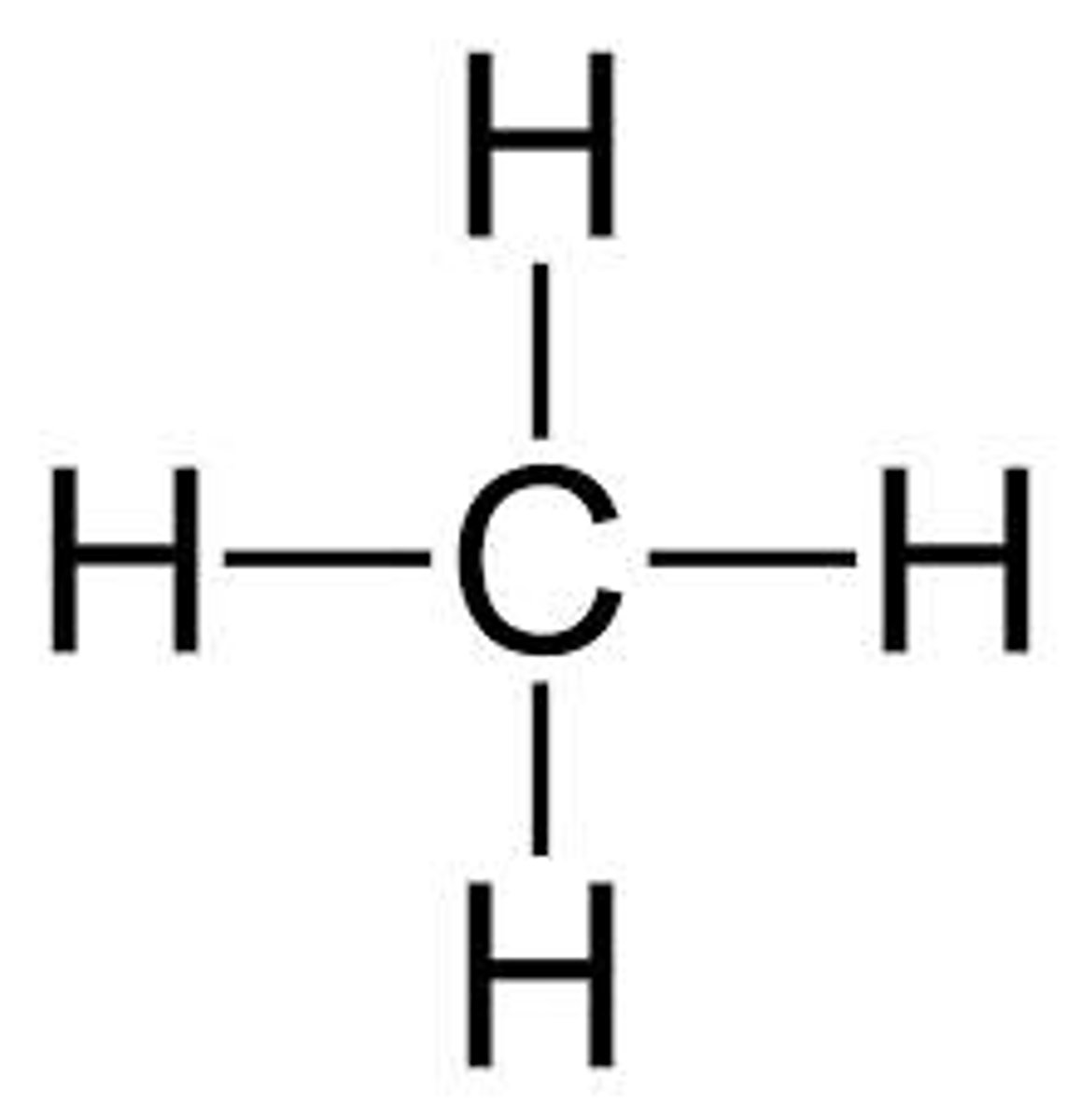
methane
alcohols
polar, modified water molecules
alcohols functional group
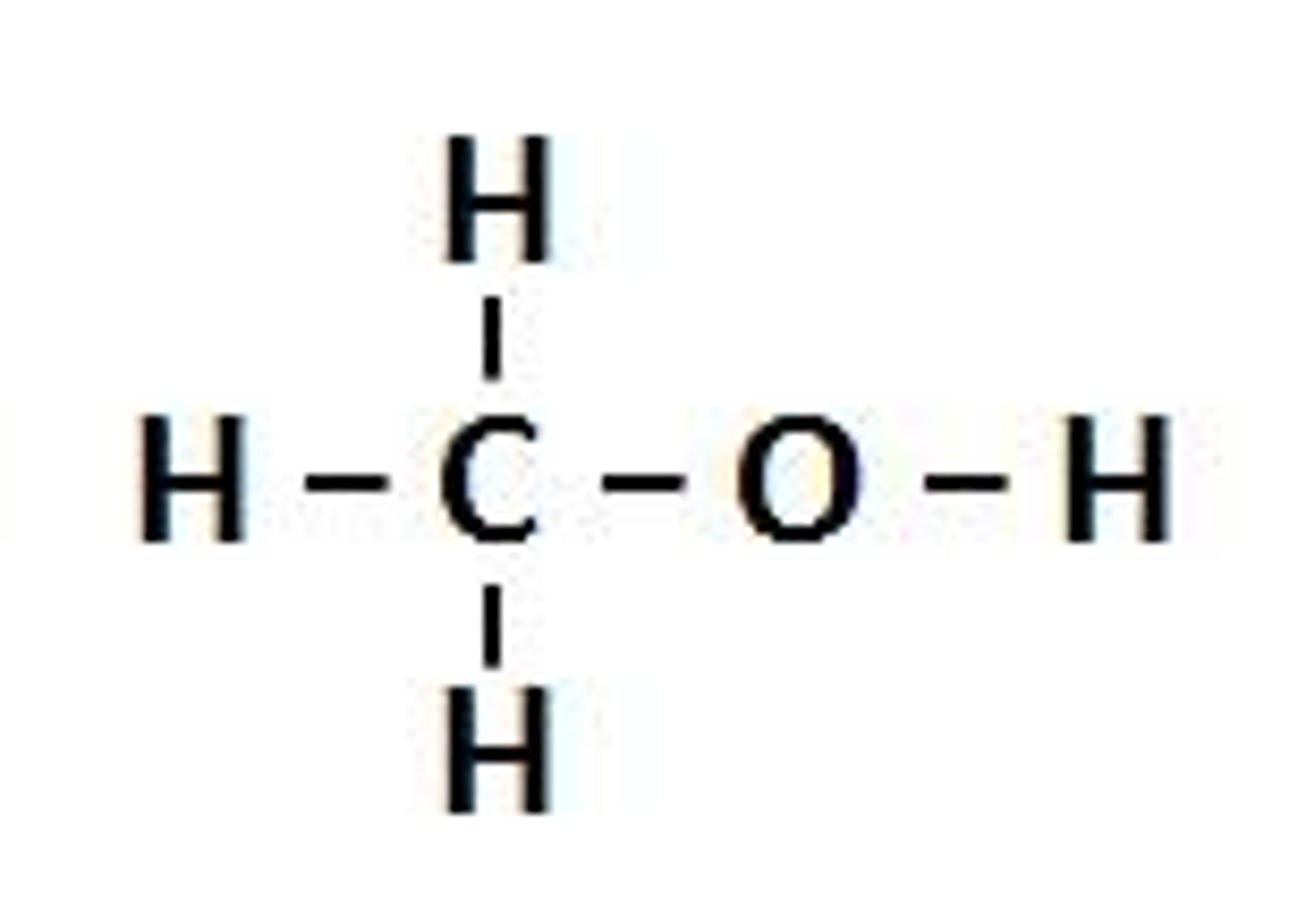
special case of alcohols
waxes are large multicarbon alcohols that do not dissolve in water because they have large hydrocarbon tails
amines
polar, modified ammonia molecules, can be powerful bases
amines functional group
C-NH2
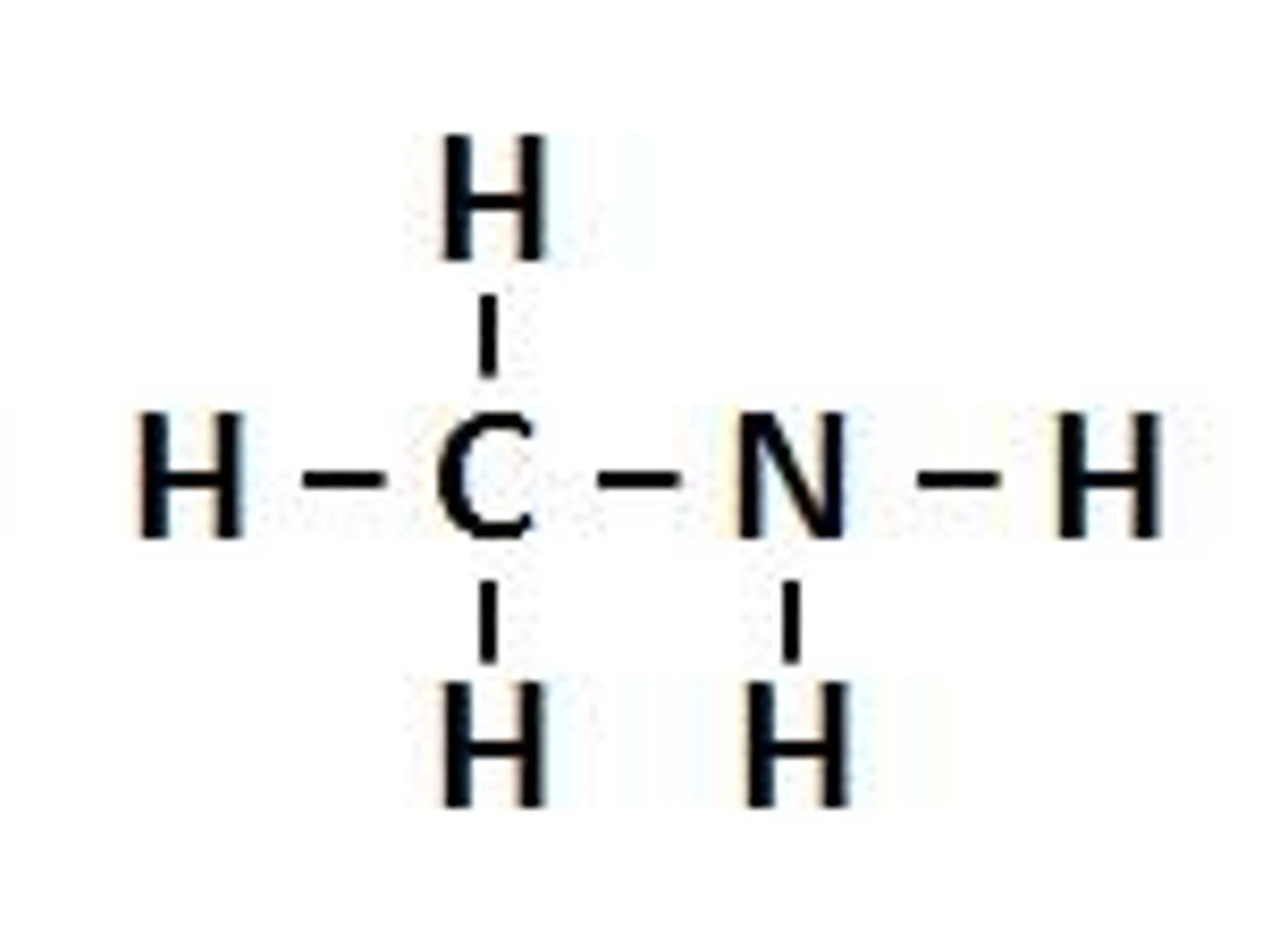
aldehydes and ketones
polar
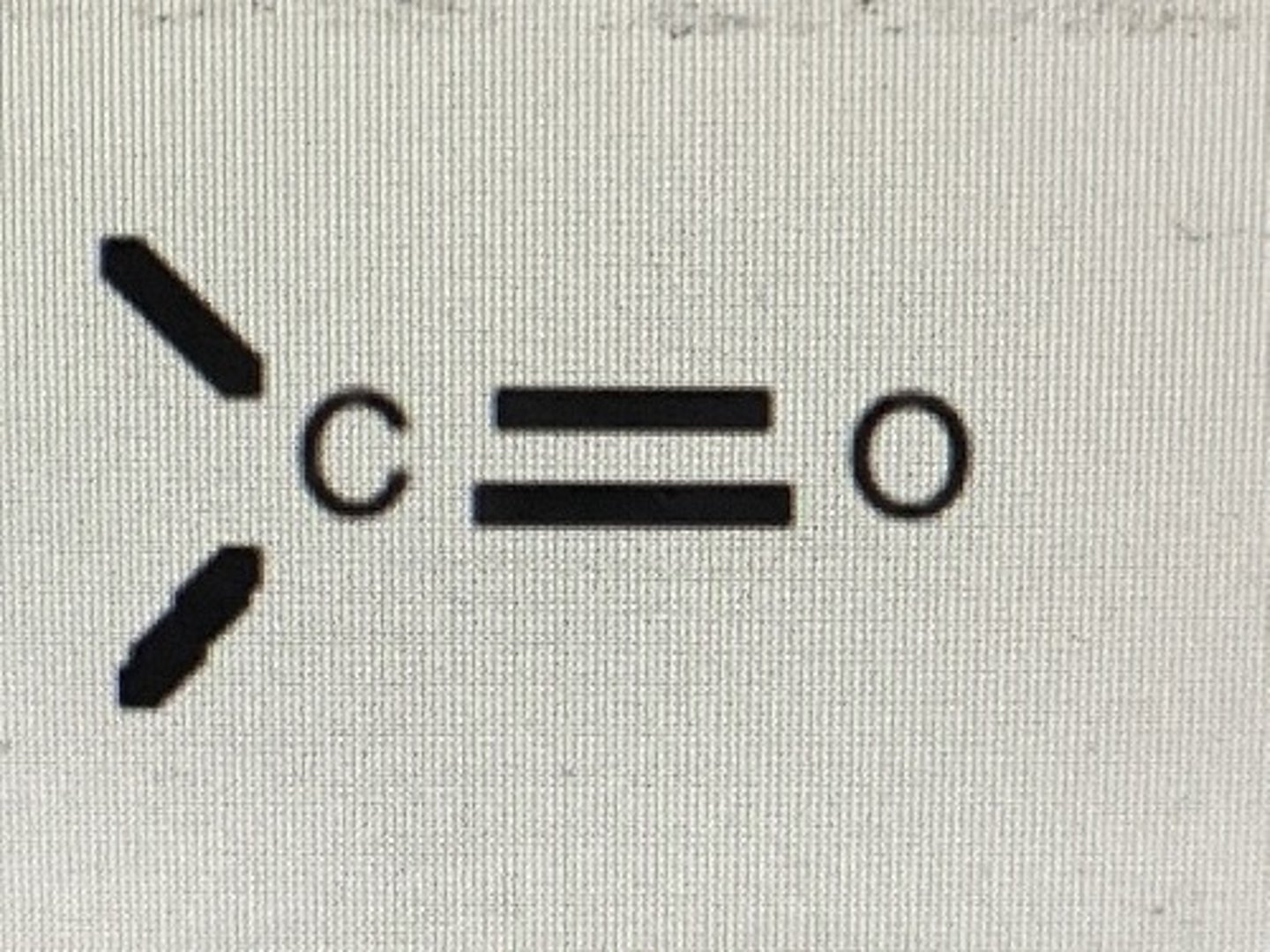
aldehydes and ketones functional group
carbonyl
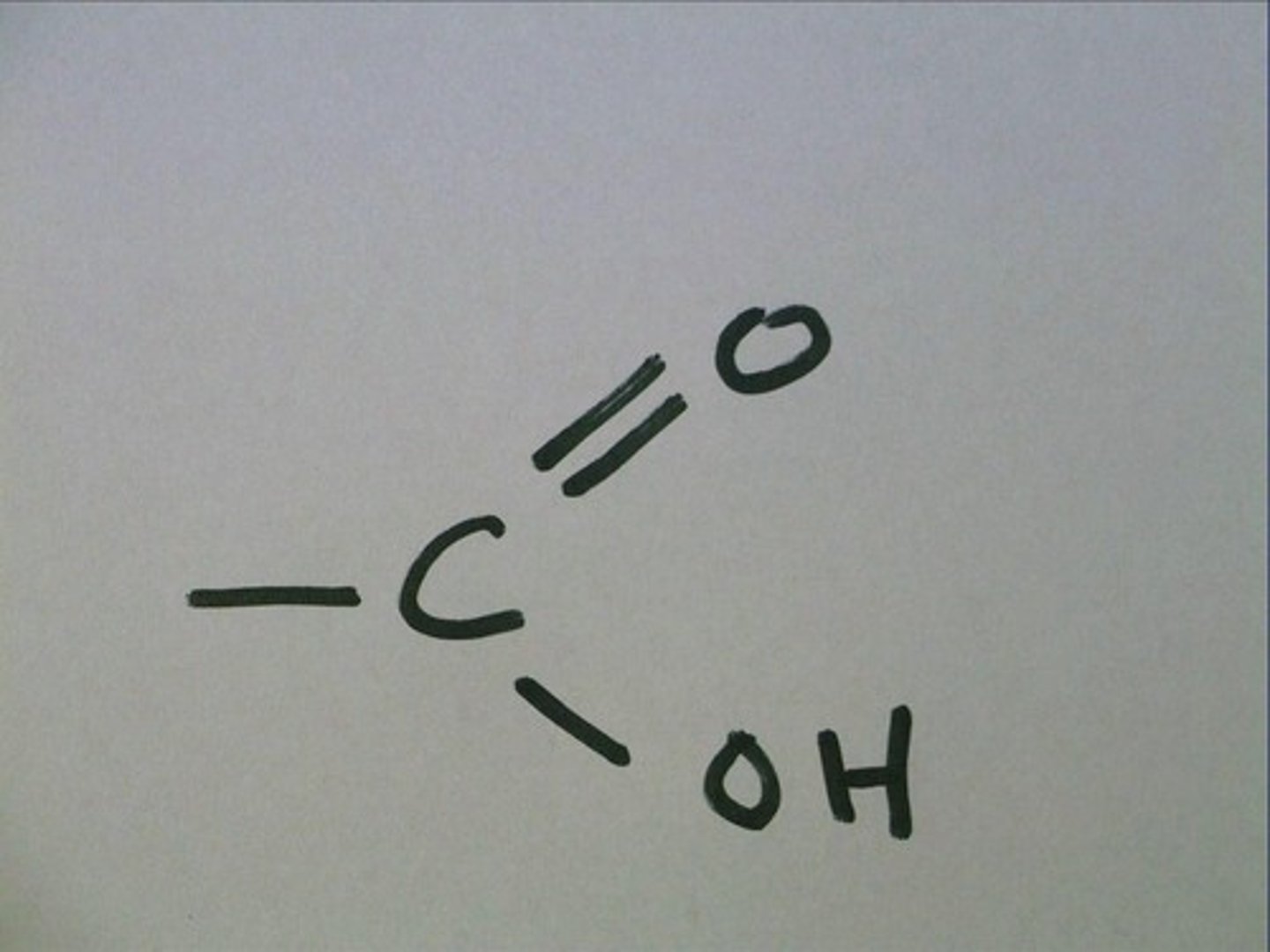
carboxylic acids
polar, weak acids
carboxylic acids functional group
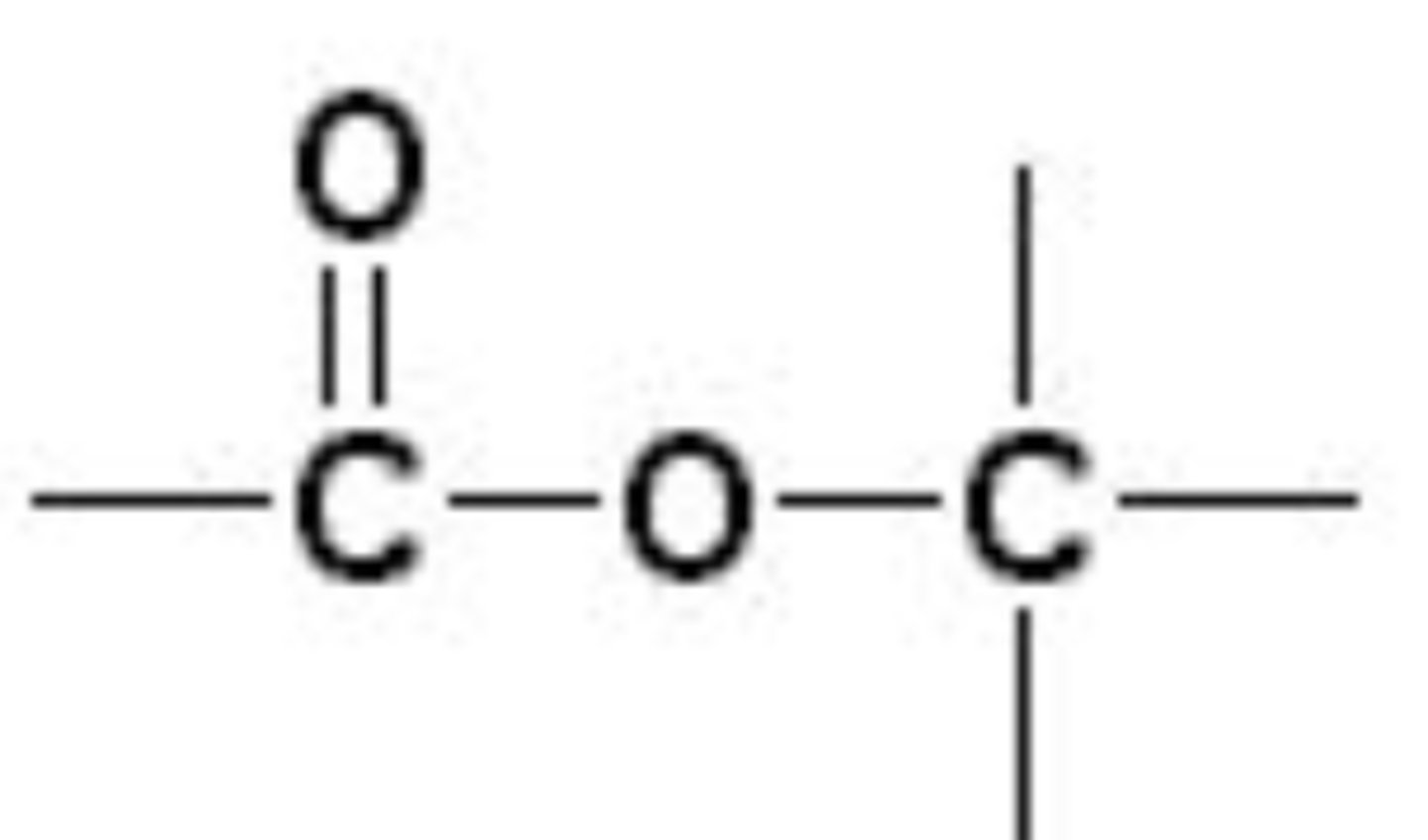
esters
non polar, combination of an alcohol and a carboxylic acid, fats are special cases, formed by dehydration, responsible for scents and flavors
esters functional group
carbohydrates definition
polyhyrdoxy aldehyde or ketone, aka sugars
biologically important carbohydrates
have 3-7 Cs (triose, tetrose, pentose, hexose)
monosaccharides definition
single hexose units
disaccharides definition
two conjoined monosaccharides
glucose
monosaccharide
-universal cell food
-found in honey and maple syrup
fructose
monosaccharide
-found in all sweet fruit and corn
galactose
monosaccharide
-found in milk only
sucrose
disaccharide
-cane or table sugar
-composed of a glucose and a fructose
maltose
disaccharide
-found in dark grains
-composed of two glucoses
lactose
disaccharide
-found in mammal milk
-composed of a glucose and a galactose
hexose polymers
fructose: fructans
galactose: galactans
the 2 glucose polymers
two glucose polymers
starches and celluloses
starches
-animals (glycogen)
-plants (starch)
-purpose: glucose storage
-amylase: enzyme that breaks up glucose for use
celluloses
-purpose: structural material
-used by plants to build bark, wood, leaves, etc
cellulase
enzyme that breaks down cellulose into glucose, made by bacteria
lipids definition
esters that are made of an alcohol called glycerol and 1-3 large carboxylic acids (aka fatty acids)
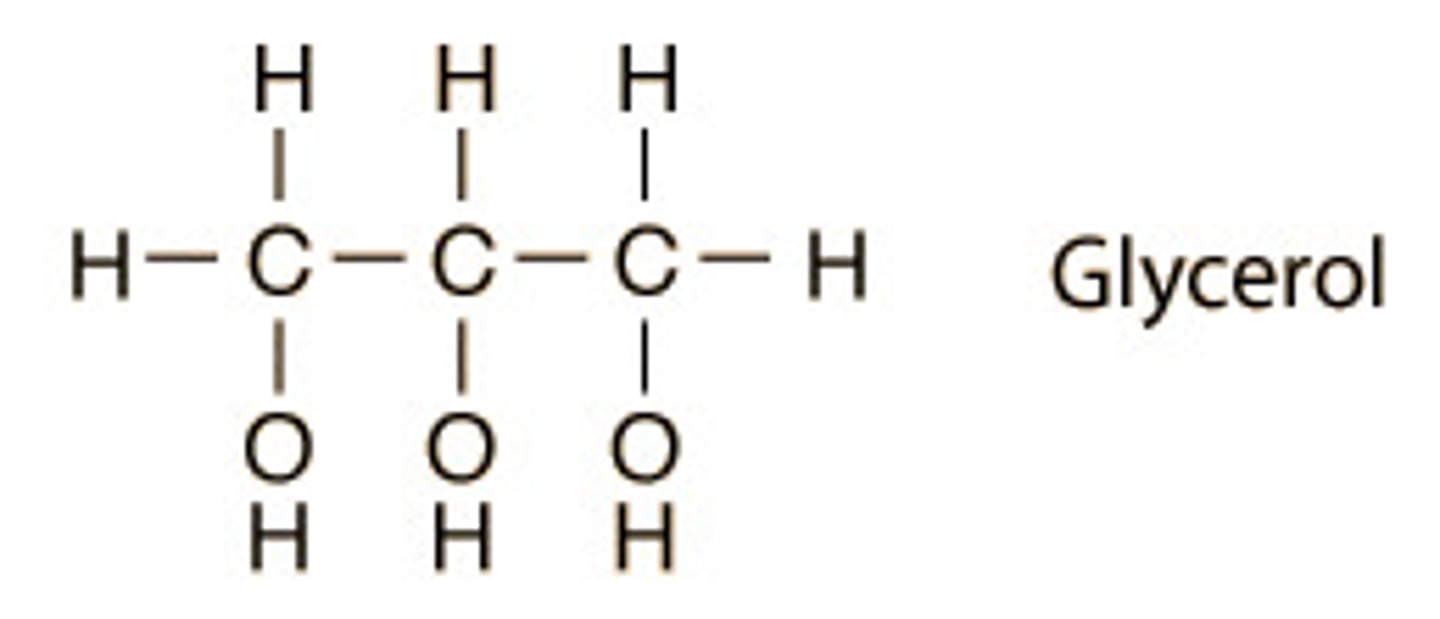
glycerol
3 carbon alcohol
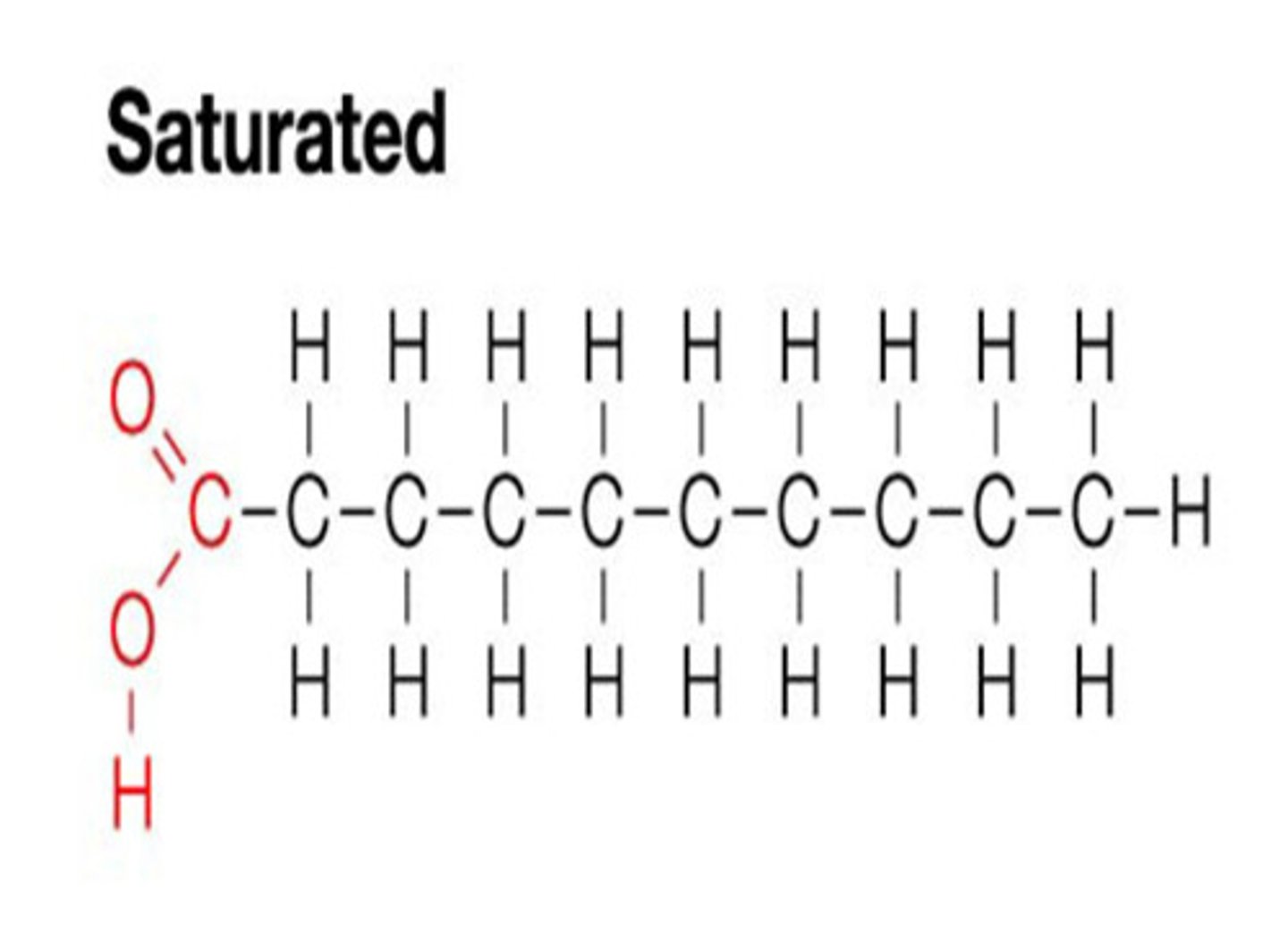
fatty acids
large 10-28 C carboxylic acids
basic lipid
mono acyl glyceride
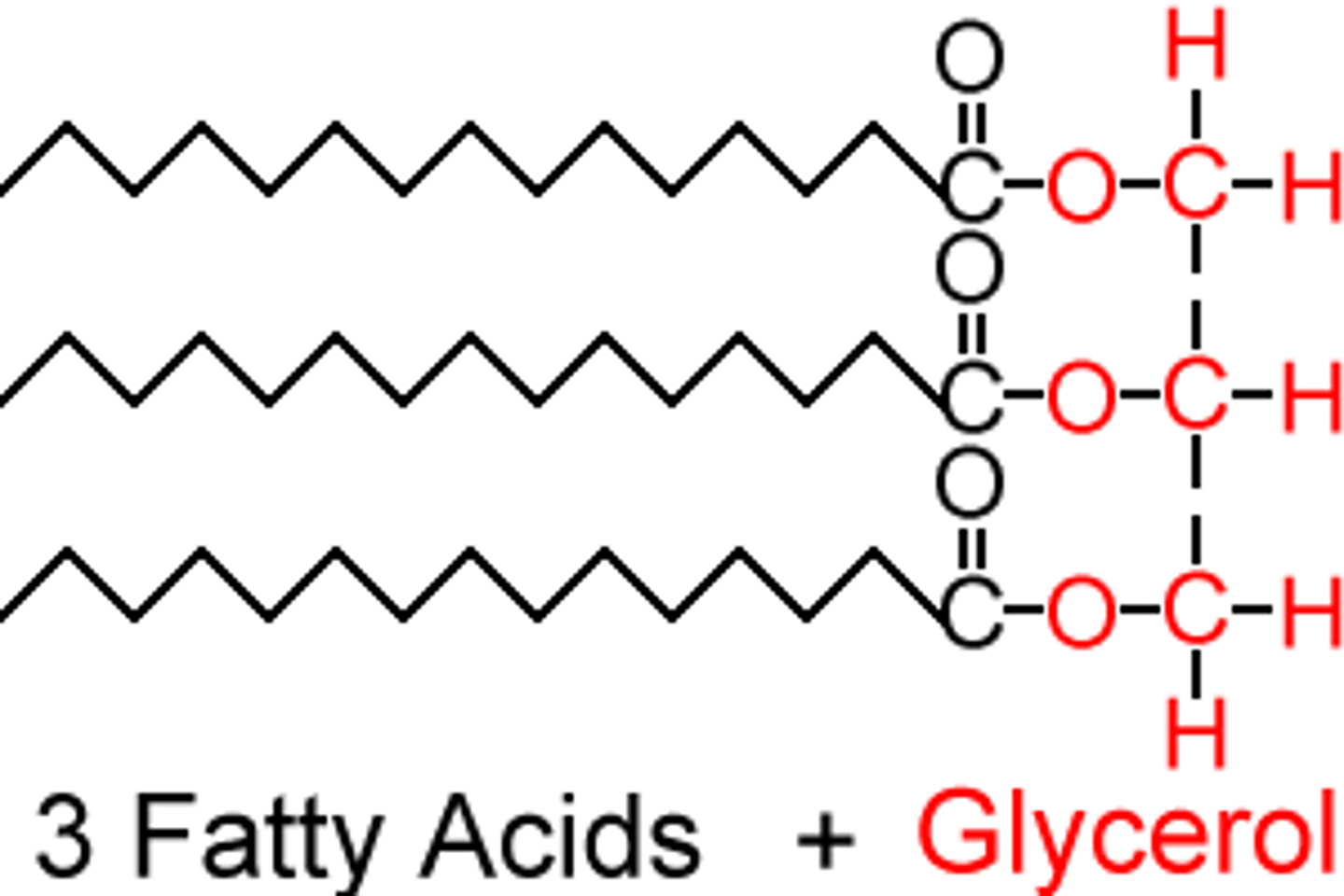
saturated vs unsaturated lipids
saturated: no double bonds, solid
unsaturated: has double bonds, liquid
double bond in saturation
double bond has the potential to absorb hydrogen, double bonds create kinks that make it harder for molecules to stick together (liquids)
lipids biological significance
building blocks of cell membranes, used for long term energy storage, high energy density
carbs vs fats energy storage
carbs are used for short term energy short falls, fat is used longterm to prevent starvation
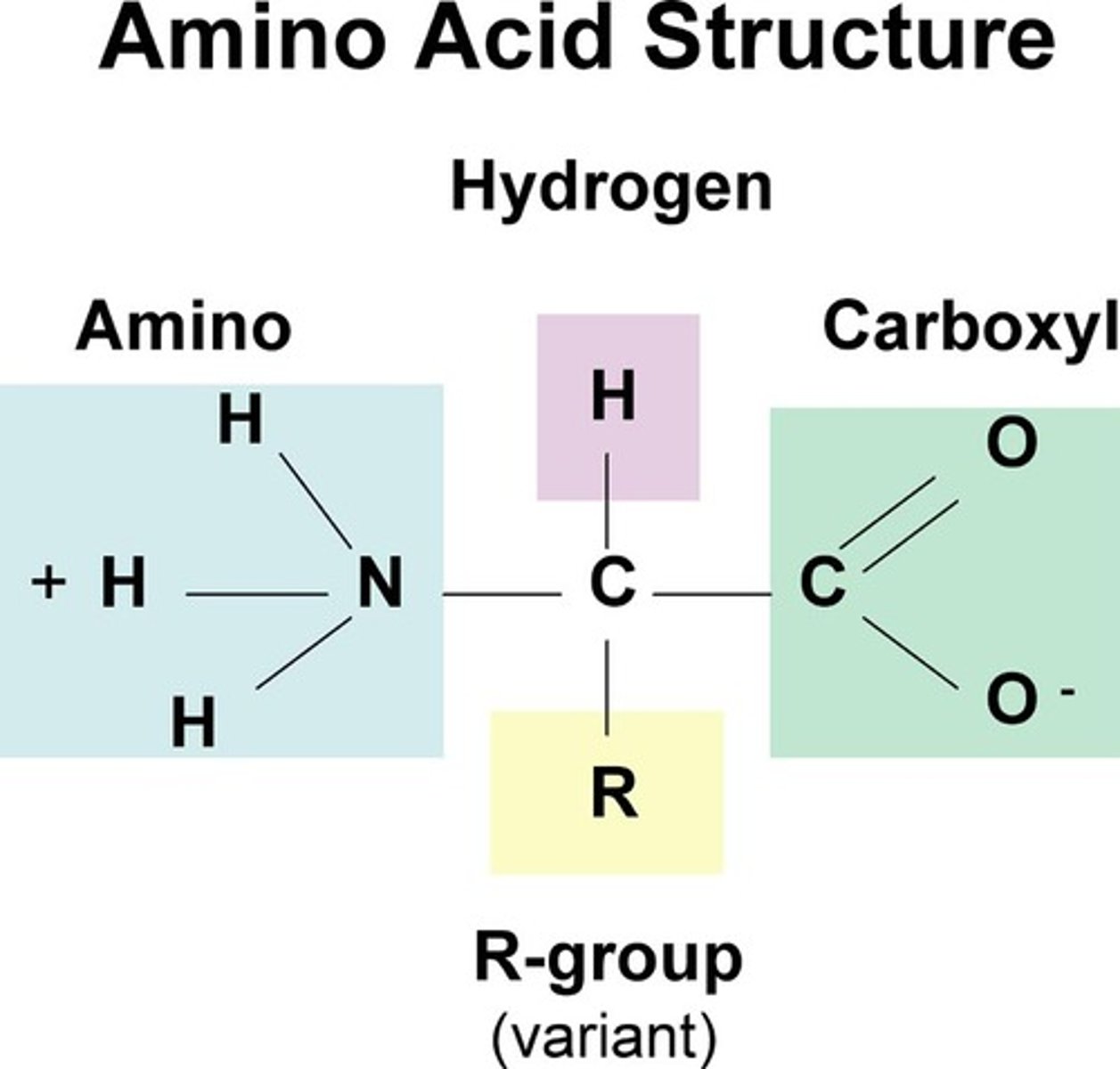
amino acid definition
monomers of proteins, all have the same generic structure
amino acid structure
R group can be anything
number of amino acids used by living things
20
three R group categories
nonpolar, polar uncharged, charged
charged R groups
can be negative or positive, can switch between neg and pos from pH shifts
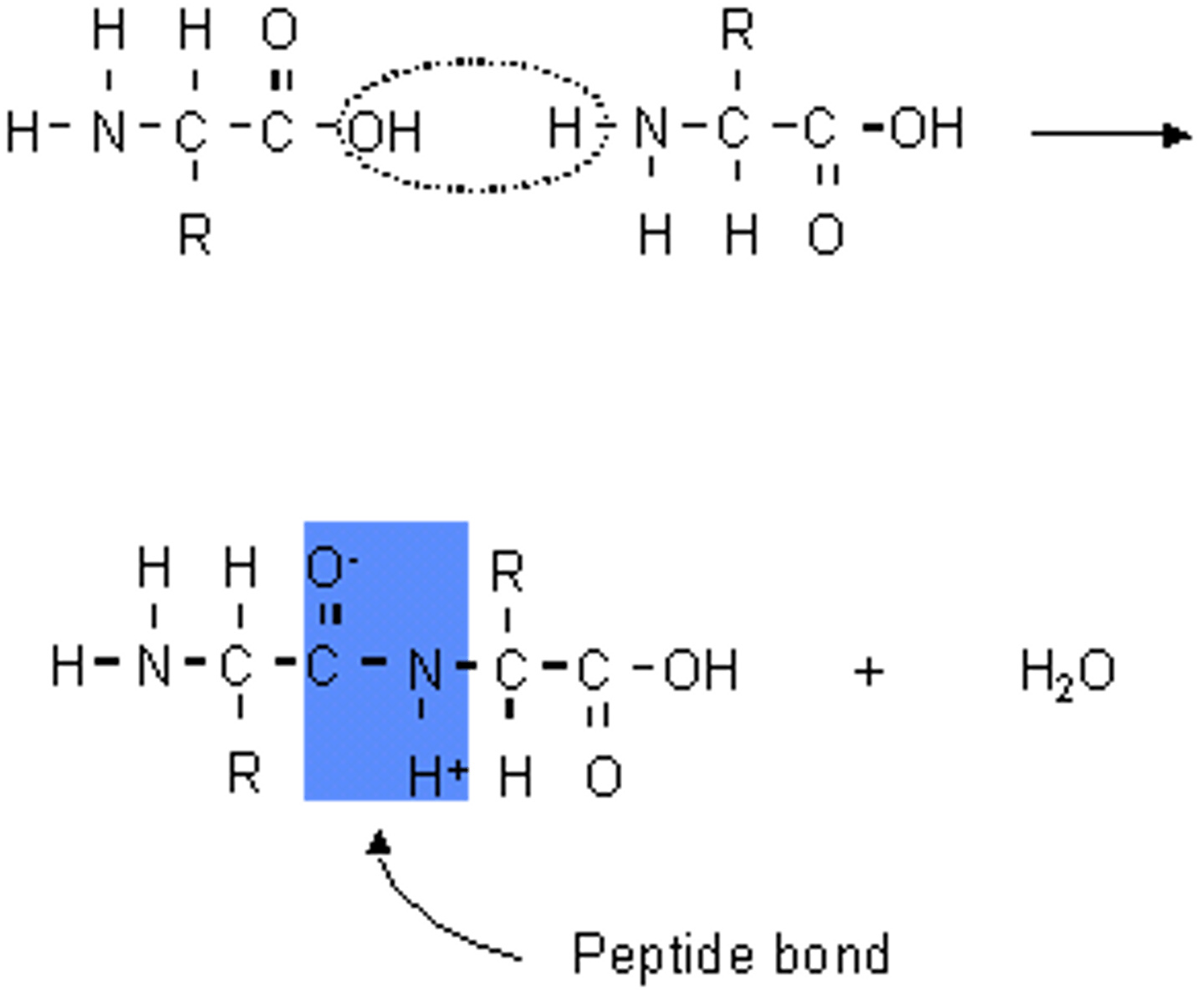
dipeptides
two amino acids joined together by a peptide bond
peptide bond
proteins definition
amino acid polymers
three structural levels of proteins
primary, secondary, tertiary