LAB 5: Isolating Clove Oil from Cloves Using Steam Distillation
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What precautions must be taken when mixing reagents in a separatory funnel or centrifuge tube?
One should frequently release the pressure, CO2 gas within the separatory funnel by spinning the stopcock
Codistillation
Distillation performed on two immiscible liquids
Raoult's Law
The total vapor pressure of the system is determined by adding together the products of the vapor pressure and the mole fraction of each compound. PT=X1P1°+X2P2°
Dalton's Law
The total vapor pressure of a mixture of gases (when 2 immiscible liquids are distilled) above the liquid is equal to the sum of the vapor pressure of each compound. PT=P1°+P2°
Essential oil
A natural, volatile oil extracted from plants that is commonly obtained using distillation. Carries the fragrance or taste of the plant. The "essence" of the plant
Why is steam distillation preferable to simple distillation for isolating high-boiling natural products?
It avoids high temperature that may lead to the decomposition of the product. Steam distillation allows volatile compounds that are unstable or have high boiling points to codistill with water at low temps
What are the positive tests for KMnO4 and Br2?
KMnO4: The disappearance of MnO4 as a muddy brown precipitate
Br2: The disappearance of the red
What is the purpose of using anhydrous sodium sulfate (NaSO4)?
It is used as a drying agent to remove traces of water from organic solutions.
Which distillation is used for a miscible mixture? What law describes it?
Fractional and Simple distillation is used from miscible mixtures. Raoult's Law describes it.
What are two main components of clove oil?
Eugenol and Eugenol acetate
Purpose of the rotovap?
Used for compounds with lower BP to purify and dry solvents from the sample through evaporation. Avoids "bumping"
System boils
when the total pressure equals one atmosphere the system boils
Steam Distillation
involves temperatures below 100 degrees Celsius and provides a powerful way to isolate compounds that are
1) not miscible with water
2) may have boiling points too high to allow for use of normal distillation
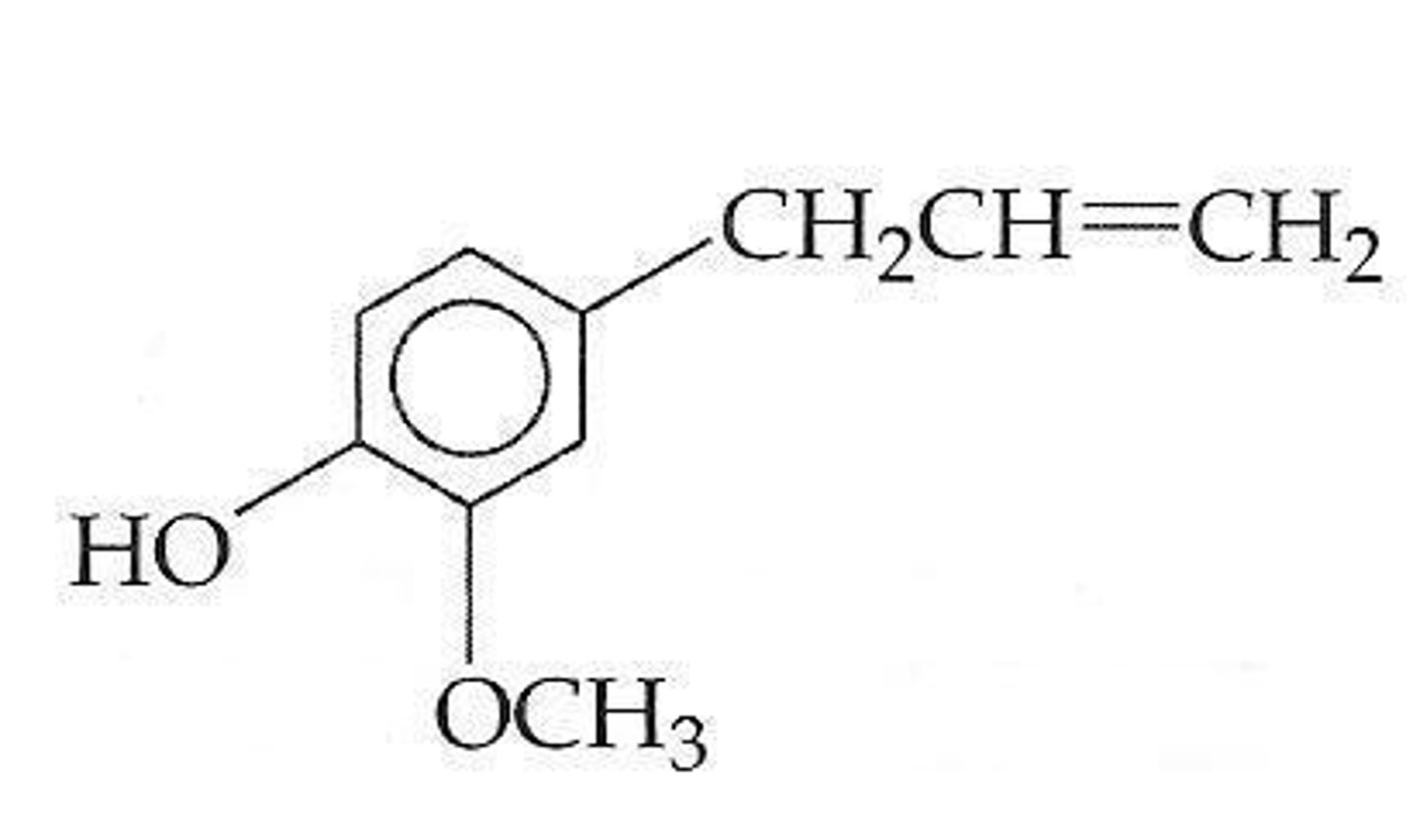
Eugenol
a terpene and also a phenol or an aromatic hydroxyl compound
boiling point is 250 degrees Celsius

Procedure
build apparatus (claisen adapter, stopper, e flask receiver), put cloves water and boiling chips into rbf, put water in addition funnel, distill until collected 30 mL, transfer to separatory funnel, rinse e flask w/ ether and add, mix/vent/drain, remove organic out top, return aq and repeat extraction two more times, combine organic layers, add sodium sulfate, dry/decant, rinse sodium sulfate w/ ether 3 times, rotovap, dissolve in methanol, add 1mL methanol and product to tubes, add bromine to other third, add potassium permanganate, TLC
Why do we use steam distillation to isolate eugenol rather than purify it by simple distillation?
Eugenol is isolated via steam distillation rather than simple distillation because it has a boiling point of around 250 degrees Celsius. This temperature would have to be obtained in simple distillation, risking the decomposition of the sample, which would produce poor and impure yield. Instead, steam distillation lowers the boiling point of the compound to around 100 degrees Celsius since the initial mixture is heterogeneous (two immiscible liquids). The vapor pressures are independent of mole fractions, leading to a quicker boiling point.
Dichloromethane/methylene chloride
is NOT flammable
Miscible Mixtures
Simple and fractional distillations are carried out in miscible mixtures
Co-Distillation
Distillation that can also be done in mixture that are immiscible
Steam distillation is carries out in 1 or 2 methods:
1st method is used in class, an excess of water is added to the compound of interest in a distilling flask. the mixture is then heated to boiling point. Resulting vapor is condensed and collected in the receiver flask. The compound of interest is the n separated from the water, often by extraction.
Purpose:
will isolate clove oil from cloves by semi-microscale steam distillation and extraction. The product will be characterizing by using reaction with bromine, potassium, permanganate, and Iron III chloride. TLC will demonstrate and analyze the purity of the product.
In the isolation of clove experiment, several tests were used to classify eugenol. Give the reagents for One of the two tests, the product of tat reaction, and briefly describe the observation one would expect for that test?
Bromine, when mixed in clove oil it turned red.
Raoult's Law addresses the thermodynamics that allow steam distillation to work. Give the equation for Raoult's law, for a two component system, and state what each variable stands for?
Pt=P1X1+P2X2
Pt=Total Pressure
P1=Pressure of cmpd 1
X1= Mole fraction fo cmpd 1
P2=Pressure of cmpd 2
X2=Mole fraction of cmpd 2
Give you test result for the reaction of your eugenol product with each of the test reagents
Eugenol product with bromine test resulted in 12 drops until pale yellow color persisted. Eugenol product with permanganate resulted in a brown substance after 3 drops. The control group needed only one drop for color to persist when tested with bromine and remained purple when tested with permanganate.
1) codistillation
2) Steam distillation
1) is distillation of two compounds are not miscible.
2) distillation when one of the compounds is water.
- avoids decomposition that might occur at the normal point.
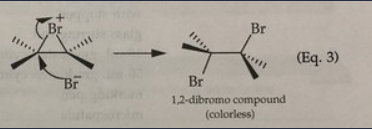
Bromine (Br2)
-It reacts with the double bond to form a colorless compound. A positive test is the disappearance of the red color
- Test the presence of unsturation
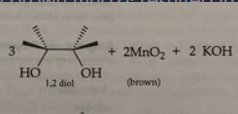
Potassium permanganate
-A positive test is the disappearance of the purple KMnO4 and the appearance of MnO2 as a brown percipitate.
- test for the presence of double or triple bonds in a molecule, s
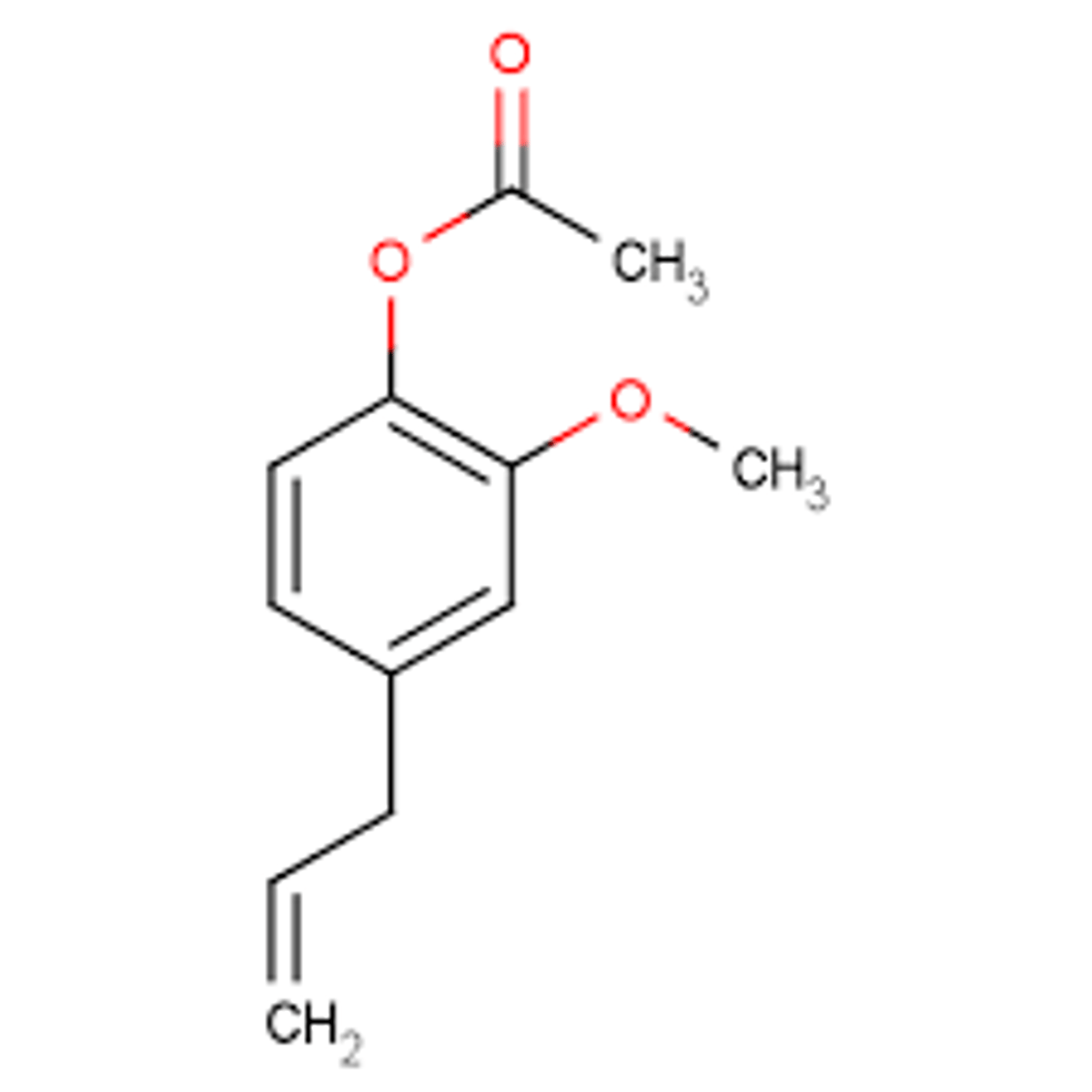
Eugenol acetate
has a higher Rf value than eugenol.
When all of the clove oil reacted with Br2,
A pale yellow color will remain.
T or F: Bromine (Br2) (red brown will react with the double bond to form a purple compound.
False, it removes color
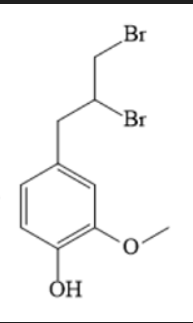
The compound below is ___________
eugenol
The compound below is ___________
eugenol acetate
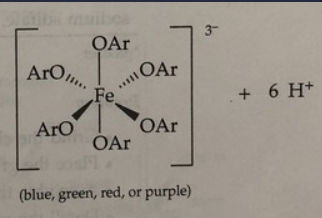
T or F: The ferric ion (Fe3+) reacts with eugenol acetate. A positive test is indicated by the appearance of either blue, green, red or purple color.
F
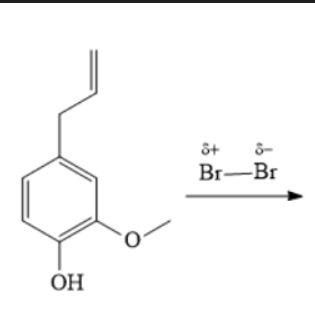
rxn: Eugenol + Br2
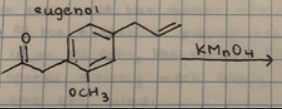
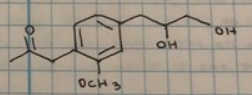
rxn: eugenol acetate + KMnO4
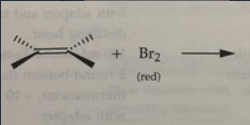
rxn: bromine (red) in dichloromethane decolorizes as br2 reacts with double bond to form
colorless compound (positive = disappearance of red br2 color); resulted in yellow test tubes
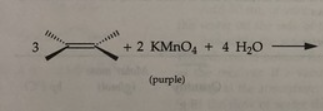
rxn: kmno4 (purple) can oxidize double bond to form
brown mno2 (positive = disappearance of purple kmno4 and appearance of muddy brown mno2); resulted in brown/maroon test tubes
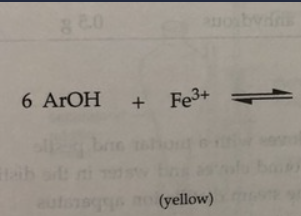
rxn: phenols Ar-OH react with Fe 3+ (yellow) in fecl3 to form
complexes that are blue, green, red, or purple