CHEM 270 MIDTERM 3
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
![<p>7.1</p><p>Using the (E )–(Z ) designation [and in parts (e) and (f) the (R )–(S ) designation as well], give IUPAC names for each of the following:</p>](https://assets.knowt.com/user-attachments/51ed16ee-abb2-4cff-85a6-2e9660460646.png)
7.1
Using the (E )–(Z ) designation [and in parts (e) and (f) the (R )–(S ) designation as well], give IUPAC names for each of the following:
7.2
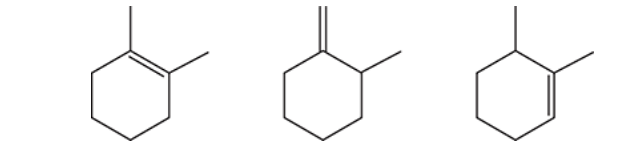
Rank the following cycloalkenes in order of increasing stability.
7.3
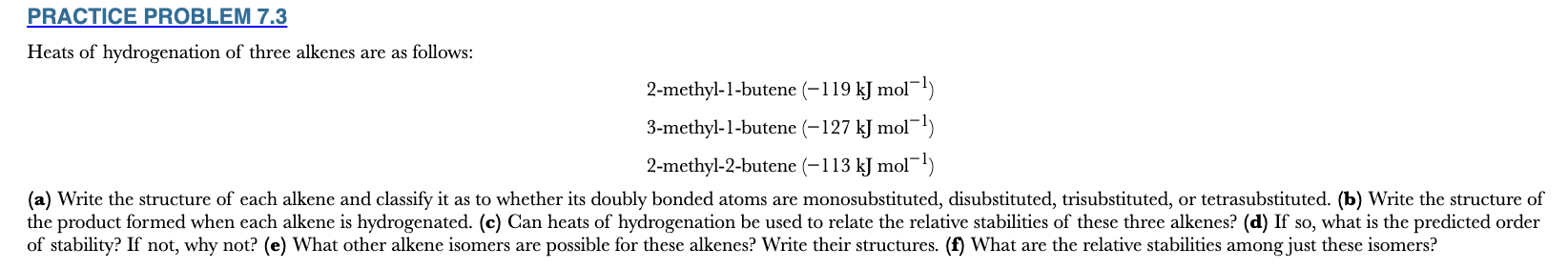
Heats of hydrogenation of three alkenes are as follows:
2-methyl-1-butene (−119 kJ mol−1)
3-methyl-1-butene (−127 kJ mol−1)
2-methyl-2-butene (−113 kJ mol−1)
(a) Write the structure of each alkene and classify it as to whether its doubly bonded atoms are monosubstituted, disubstituted, trisubstituted, or tetrasubstituted. (b) Write the structure of the product formed when each alkene is hydrogenated. (c) Can heats of hydrogenation be used to relate the relative stabilities of these three alkenes? (d) If so, what is the predicted order of stability? If not, why not? (e) What other alkene isomers are possible for these alkenes? Write their structures. (f) What are the relative stabilities among just these isomers?
7.4
Predict the more stable alkene of each pair: (a) 2-methyl-2-pentene or 2,3-dimethyl-2-butene; (b) cis-3-hexene or trans-3-hexene; (c) 1-hexene or cis-3-hexene; (d) trans-2-hexene or 2-methyl-2-pentene.
7.5
How many stereoisomers are possible for 4-methyl-2-hexene, and how many fractions would you obtain if you distilled the mixture?
7.6
Predict the major product formed when 2-bromobutane is subjected to dehydrobromination using sodium ethoxide in ethanol at 55 °C.
7.7
List the alkenes that would be formed when each of the following alkyl halides is subjected to dehydrohalogenation with potassium ethoxide in ethanol and use Zaitsev’s rule to predict the major product of each reaction: (a) 2-bromo-3-methylbutane and (b) 2-bromo-2,3-dimethylbutane.
7.9
Consider a simple molecule such as ethyl bromide and show with Newman projection formulas how the anti coplanar transition state would be favored over the syn coplanar one.
7.10
When cis-1-bromo-4-tert-butylcyclohexane is treated with sodium ethoxide in ethanol, it reacts rapidly; the product is 4-tert-butylcyclohexene. Under the same conditions, trans-1-bromo-4-tert-butylcyclohexane reacts very slowly. Write conformational structures and explain the difference in reactivity of these cis–trans isomers.
7.11
(a) When cis-1-bromo-2-methylcyclohexane undergoes an E2 reaction, two products (cycloalkenes) are formed. What are these two cycloalkenes, and which would you expect to be the major product? Write conformational structures showing how each is formed. (b) When trans-1-bromo-2-methylcyclohexane reacts in an E2 reaction, only one cycloalkene is formed. What is this product? Write conformational structures showing why it is the only product.
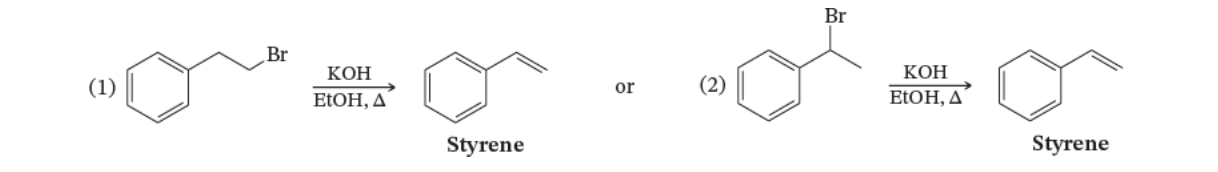
7.12
Your task is to prepare styrene by one of the following reactions. Which reaction would you choose to obtain the better yield of styrene? Explain your answer.
7.13
Although ethyl bromide and isobutyl bromide are both primary halides, ethyl bromide undergoes SN2 reactions more than 10 times faster than isobutyl bromide does. When each compound is treated with a strong base/nucleophile (EtO−), isobutyl bromide gives a greater yield of elimination products than substitution products, whereas with ethyl bromide this behavior is reversed. What factor accounts for these results?
7.14
Dehydration of 2-propanol occurs in 14 M H2SO4 at 100 °C. (a) Using curved arrows, write all steps in a mechanism for the dehydration. (b) Explain the essential role performed in alcohol dehydrations by the acid catalyst. (Hint: Consider what would have to happen if no acid were present.)
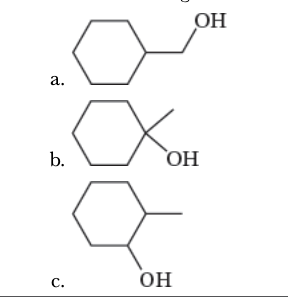
7.15
Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration.
7.16
Acid-catalyzed dehydration of 3,3-dimethyl-1-butanol yields 2-methyl-2-butene as the major product. Outline a mechanism showing all steps in its formation.
7.17
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.

7.19
Predict the products of the following acid–base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case, label the stronger acid, the stronger base, the weaker acid, and the weaker base:
CH3CH═CH2 + NaNH2 ⟶
CH3C≡CH + NaNH2 ⟶
CH3CH2CH3 + NaNH2 ⟶
CH3C≡C: ⁻ + CH3CH2OH ⟶
CH3C≡C: ⁻ + NH4Cl ⟶
7.20
Show how you might synthesize ethynylbenzene from methyl phenyl ketone.

7.21
Outline all steps in a synthesis of propyne from each of the following:
a. CH3COCH3
b. CH3CH2CHBr2
c. CH3CHBrCH2Br
d. CH3CH═ch2
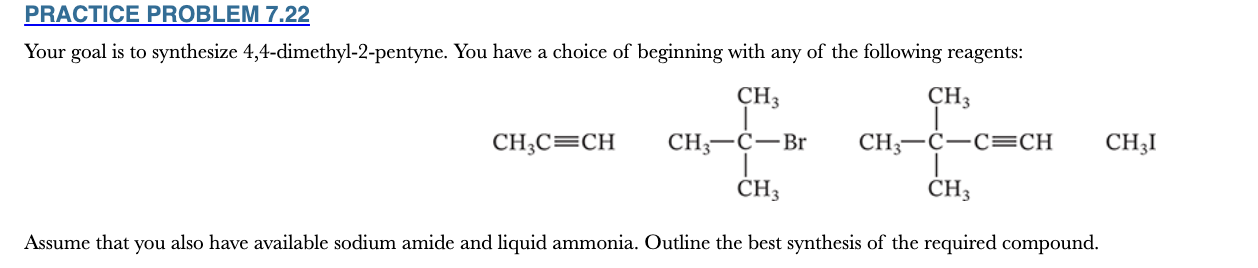
7.22
Your goal is to synthesize 4,4-dimethyl-2-pentyne. You have a choice of beginning with any of the following reagents:
Assume that you also have available sodium amide and liquid ammonia. Outline the best synthesis of the required compound.
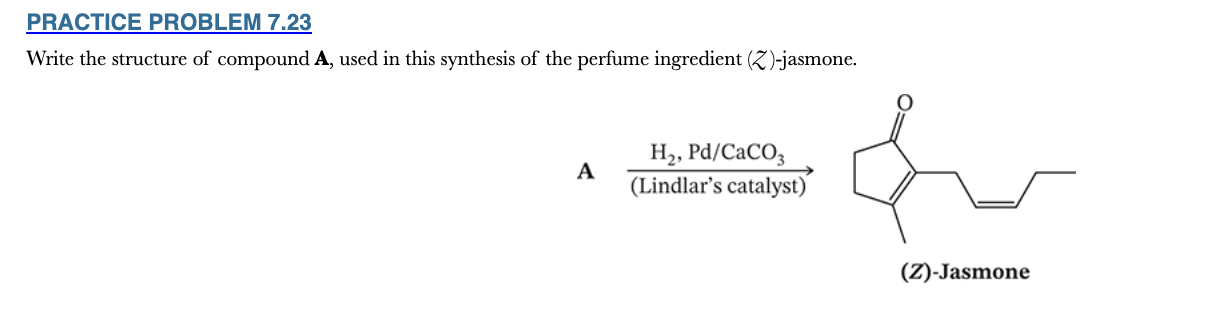
7.23
Write the structure of compound A, used in this synthesis of the perfume ingredient (Z )-jasmone.
7.24
Write the structure of compound A, used in this synthesis of the perfume ingredient (Z )-jasmone.
7.25
Referring to the retrosynthetic analysis for 2-methylhexane in this section, write reactions for those synthesis routes that are feasible.
7.26
(a) Devise retrosynthetic schemes for all conceivable alkynide anion alkylation syntheses of the insect pheromones undecane and 2-methylheptadecane (see “The Environmental Chemistry of . . . Pheromones.” box in Chapter 4). (b) Write reactions for two feasible syntheses of each pheromone.
7.28
Write a structural formula for each of the following:
a. 3-Methylcyclobutene
b. 1-Methylcyclopentene
c. 2,3-Dimethyl-2-pentene
d. (Z)-3-Hexene
e. (E)-2-Pentene
f. 3,3,3-Tribromopropene
g. (Z,4R)-4-Methyl-2-hexene
h. (E,4S)-4-Chloro-2-pentene
i. (Z)-1-Cyclopropyl-1-pentene
j. 5-Cyclobutyl-1-pentene
k. (R)-4-Chloro-2-pentyne
l. (E)-4-Methylhex-4-en-1-yne
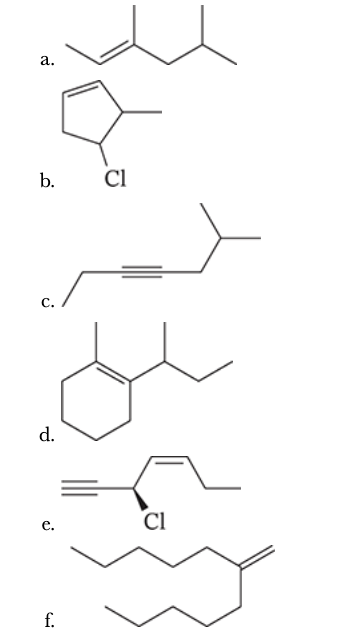
7.30
Give the IUPAC names for each of the following:
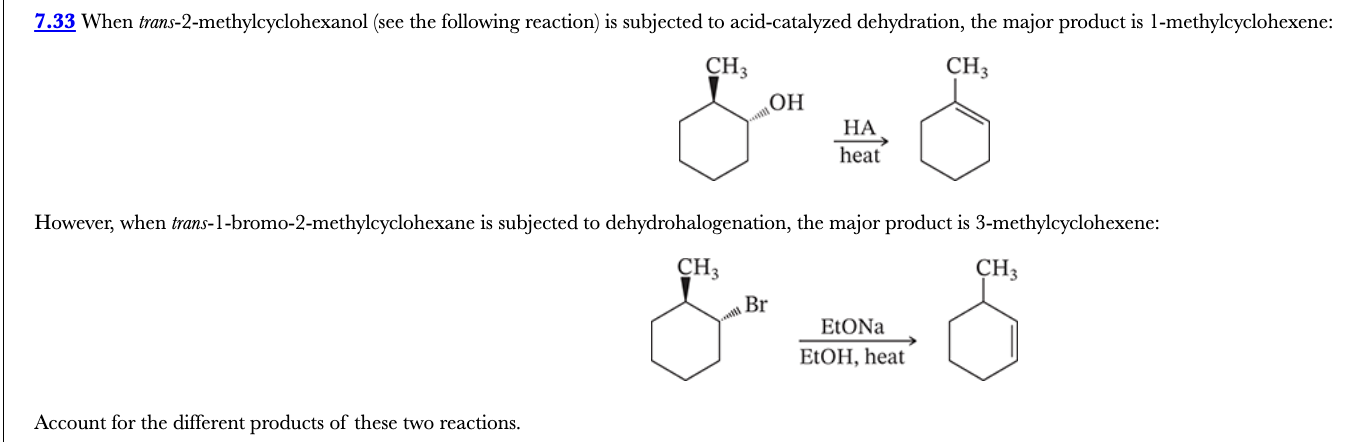
7.33
When trans-2-methylcyclohexanol (see the following reaction) is subjected to acid-catalyzed dehydration, the major product is 1-methylcyclohexene:
However, when trans-1-bromo-2-methylcyclohexane is subjected to dehydrohalogenation, the major product is 3-methylcyclohexene:
Account for the different products of these two reactions.
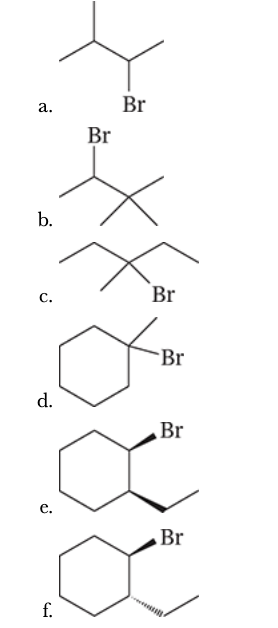
7.34
Write structural formulas for all the products that would be obtained when each of the following alkyl halides is heated with sodium ethoxide in ethanol. When more than one product results, you should indicate which would be the major product and which would be the minor product(s). You may neglect cis–trans isomerism of the products when answering this question.
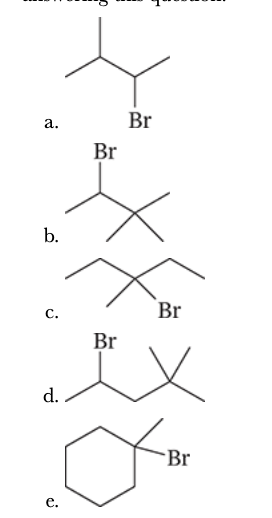
7.35
Write structural formulas for all the products that would be obtained when each of the following alkyl halides is heated with potassium tert-butoxide in tert-butyl alcohol. When more than one product results, you should indicate which would be the major product and which would be the minor product(s). You may neglect cis–trans isomerism of the products when answering this question.
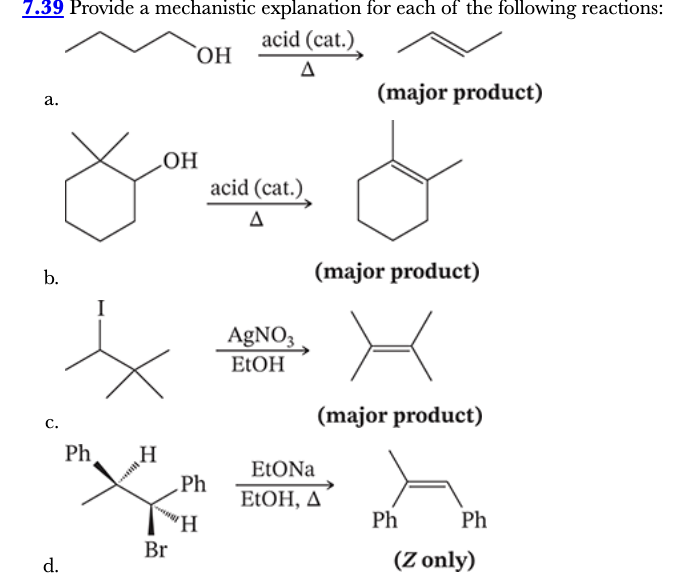
7.39
Provide a mechanistic explanation for each of the following reactions:
7.48
Starting with ethyne, outline syntheses of each of the following. You may use any other needed reagents, and you need not show the synthesis of compounds prepared in earlier parts of this problem.
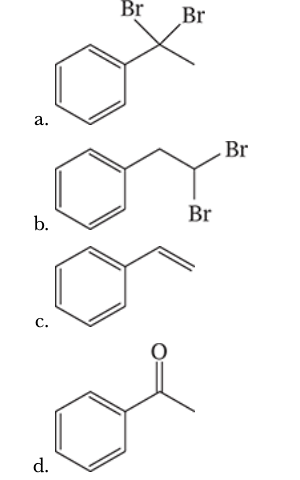
7.54
Outline a synthesis of phenylethyne from each of the following:

7.55
For each of the following questions, please provide a route that could reasonably be expected to convert the starting material into the final product. In each case, more than one reaction is required, and reactions you have learned in previous chapters may be needed to solve the problem
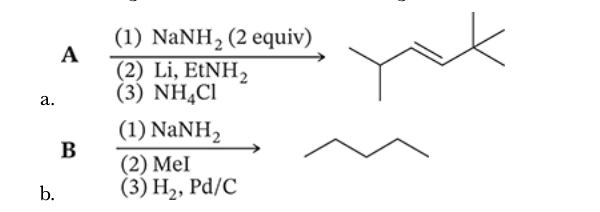
7.56
Working backwards, deduce the starting material that led to the indicated product through the defined reactions
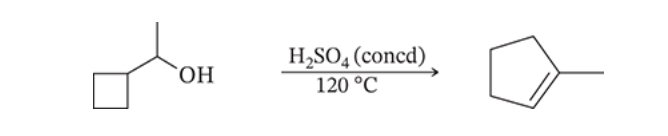
7.57
When 1-cyclobutylethanol (shown below) is treated with concentrated H2SO4 at 120 °C, one of the products that is formed is methylcyclopentene. Please write a mechanism that can account for the formation of this new product.
8.1
Give the structure and name of the product that would be obtained from the ionic addition of IBr to propene.
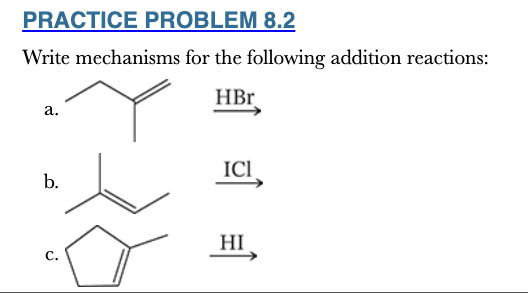
8.2
Write mechanisms for the following addition reactions:
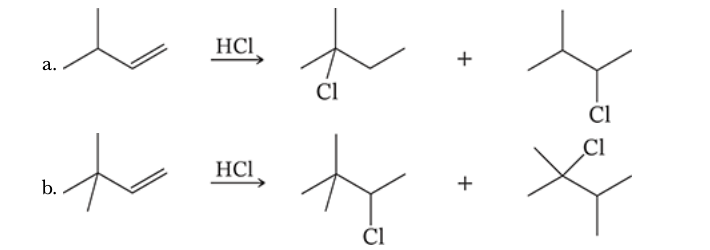
8.3
Provide mechanistic explanations for the following observations:
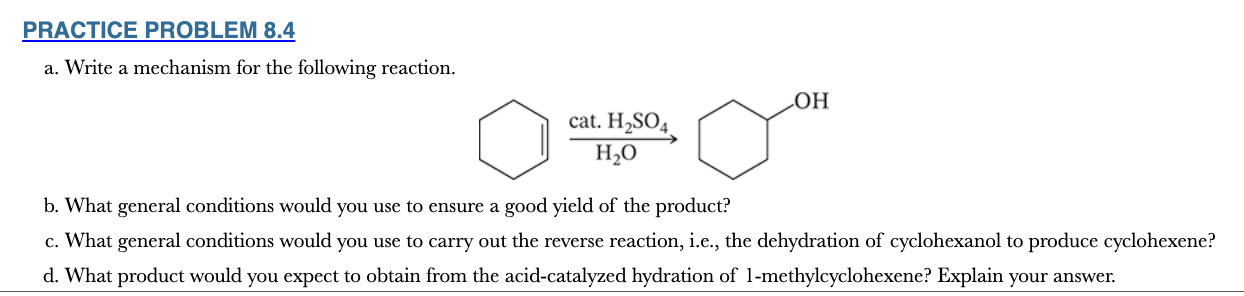
8.4
a. Write a mechanism for the following reaction.
b. What general conditions would you use to ensure a good yield of the product?
c. What general conditions would you use to carry out the reverse reaction, i.e., the dehydration of cyclohexanol to produce cyclohexene?
d. What product would you expect to obtain from the acid-catalyzed hydration of 1-methylcyclohexene? Explain your answer.
8.6
Write a mechanism that shows how 2,3-dimethyl-2-butanol is formed in the acid-catalyzed hydration of 3,3-dimethyl-1-butene.
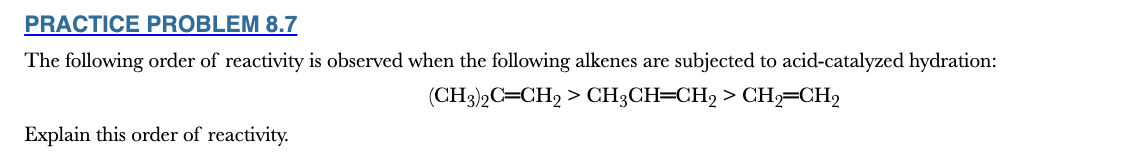
8.7
The following order of reactivity is observed when the following alkenes are subjected to acid-catalyzed hydration:
(CH3)2C═CH2 > CH3CH═CH2 > CH2═CH2
Explain this order of reactivity.
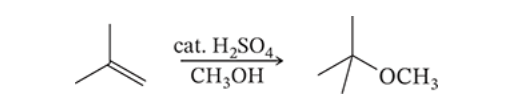
8.8
Write a mechanism for the following reaction.

8.9
Write the structure of the appropriate alkene starting materials and specify the reagents needed to synthesize each of the following alcohols by oxymercuration–demercuration
8.10
(a) Write a mechanism for the solvomercuration step of the ether synthesis just shown. (b) Show how you would use solvomercuration–demercuration to prepare tert-butyl methyl ether. (c) Propose a reason why Hg(O2CCF3)2 is more reactive than Hg(OAc)2.
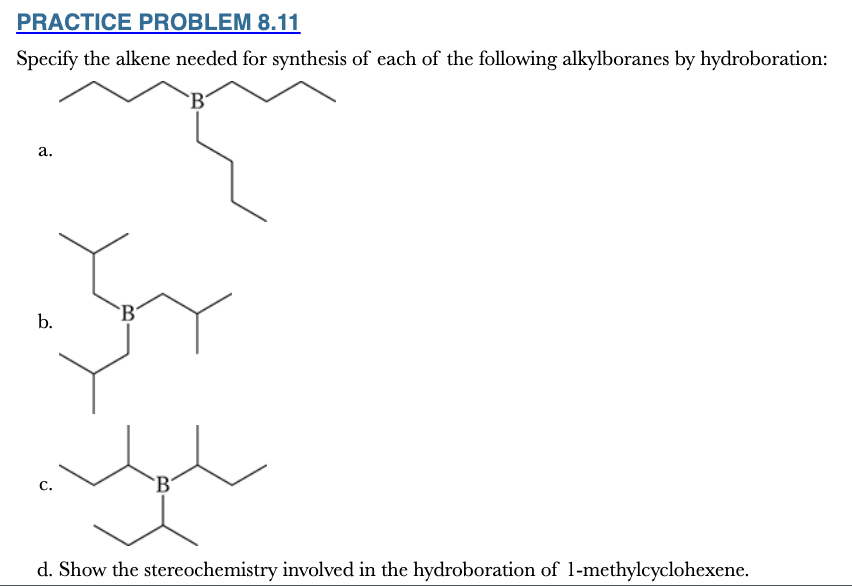
8.11
Specify the alkene needed for synthesis of each of the following alkylboranes by hydroboration:
8.12
Treating a hindered alkene such as 2-methyl-2-butene with BH3:THF leads to the formation of a dialkylborane instead of a trialkylborane. When 2 mol of 2-methyl-2-butene is added to 1 mol of BH3, the product formed is bis(3-methyl-2-butyl)borane, nicknamed “disiamylborane.” Write its structure. Bis(3-methyl-2-butyl)borane is a useful reagent in certain syntheses that require a sterically hindered borane. (The name “disiamyl” comes from “disecondary-iso-amyl,” a completely unsystematic and unacceptable name. The name “amyl” is an old common name for a five-carbon alkyl group.)
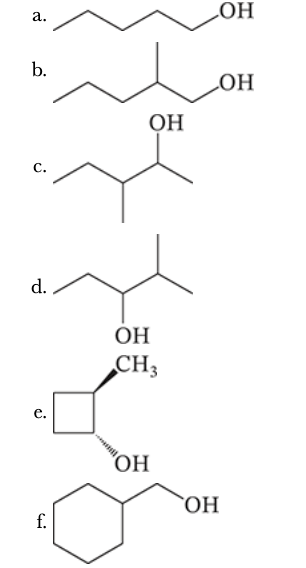
8.13
Specify the appropriate alkene and reagents for synthesis of each of the following alcohols by hydroboration–oxidation.
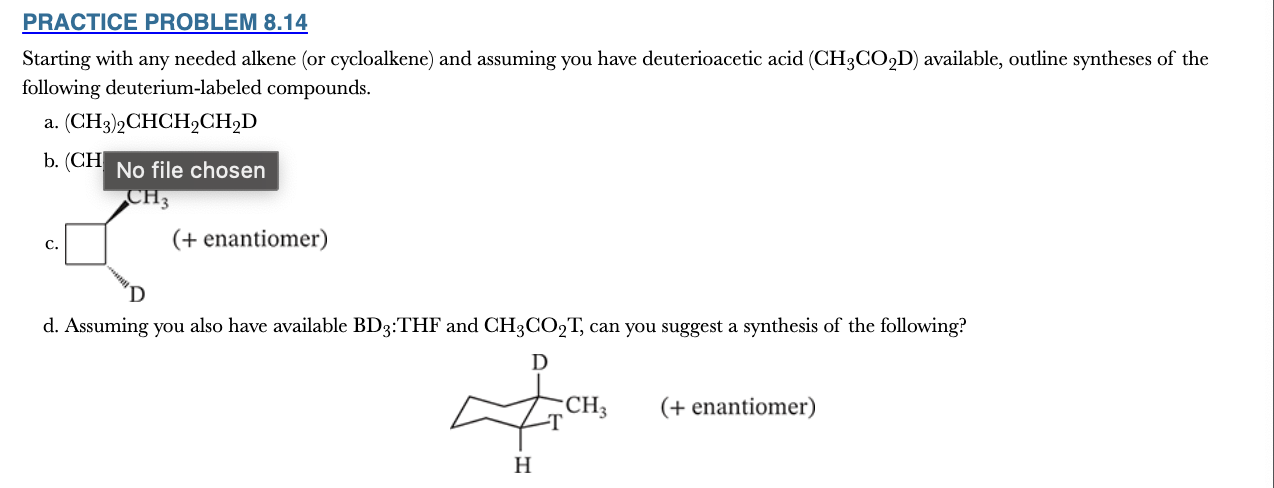
8.14
Starting with any needed alkene (or cycloalkene) and assuming you have deuterioacetic acid (CH3CO2D) available, outline syntheses of the following deuterium-labeled compounds.
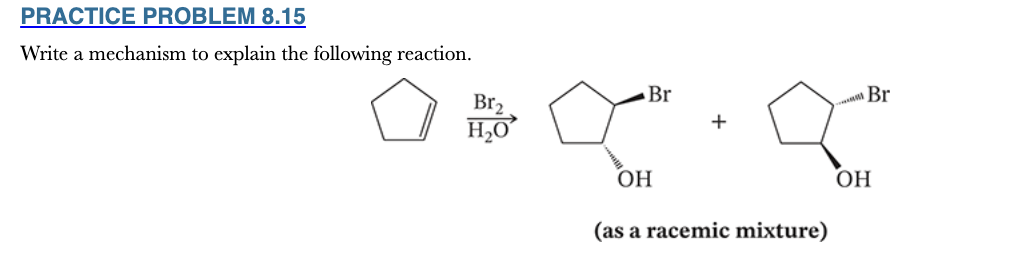
8.15
Write a mechanism to explain the following reaction.

8.17
What products would you expect from each of the following reactions?
8.18
Starting with cyclohexene and using any other needed reagents, outline a synthesis of 7,7-dibromobicyclo[4.1.0]heptane.
8.19
Treating cyclohexene with 1,1-diiodoethane and a zinc–copper couple leads to two isomeric products. What are their structures?
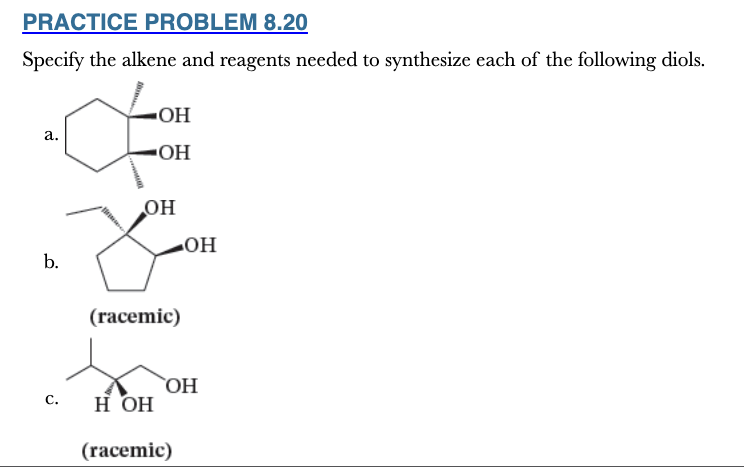
8.20
Specify the alkene and reagents needed to synthesize each of the following diols.
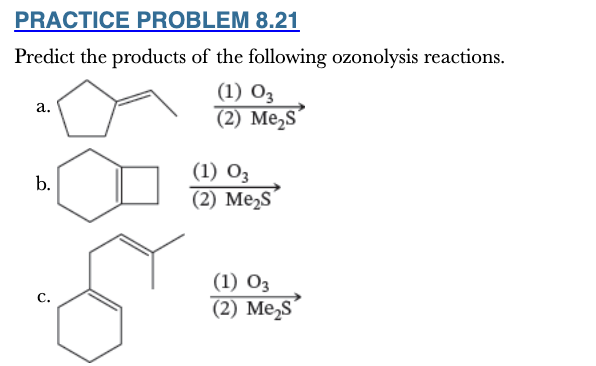
8.21
Predict the products of the following ozonolysis reactions.

8.22
Write the structures of the alkenes that would yield the following carbonyl compounds when treated with ozone and then with dimethyl sulfide.
8.23
Alkenes are more reactive than alkynes toward addition of electrophilic reagents (i.e., Br2, Cl2, or HCl). Yet when alkynes are treated with one molar equivalent of these same electrophilic reagents, it is easy to stop the addition at the “alkene stage.” This appears to be a paradox and yet it is not. Explain.
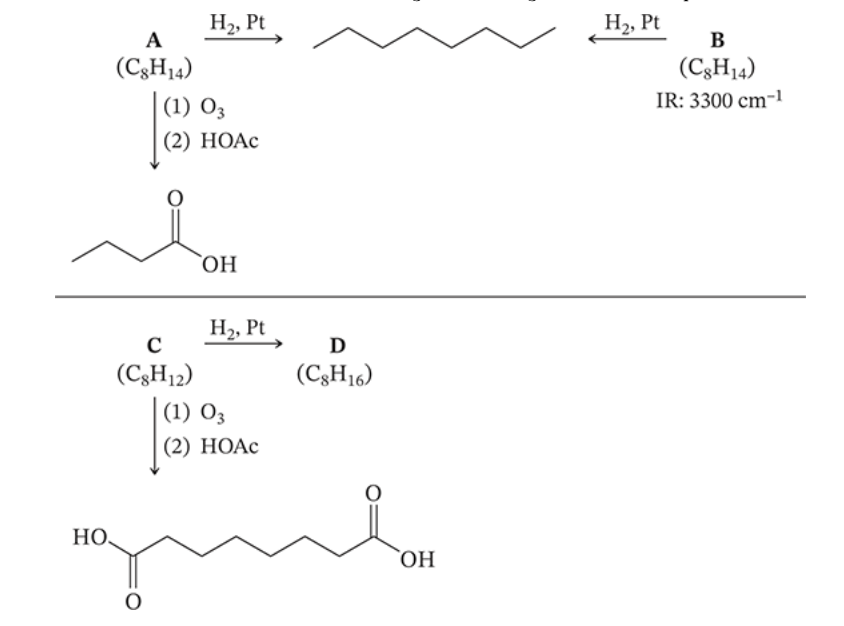
8.24
A, B, and C are alkynes. Elucidate their structures and that of D using the following reaction roadmap.
8.25
How would you modify the procedure given in Solved Problem 8.8 so as to synthesize a racemic form of (3R,4R)- and (3S,4S)-3,4-dibromohexane?
8.26
Write structural formulas for the products that form when 1-butene reacts with each of the following reagents:
a. HI
b. H2, Pt
c. H2O, cat. H2SO4
d. HBr
e. Br2
f. Br2 in H2O
g. HCl
h. O3, then Me2S
i. OsO4, then NaHSO3/H2O
j. KMnO4, HO−, heat, then H3O+
k. Hg(OAc)2 in THF and H2O, then NaBH4, HO−
l. BH3:THF, then H2O2, HO−
8.27
Repeat Exercise 8.26 using 1-methylcyclopentene instead of 1-butene.
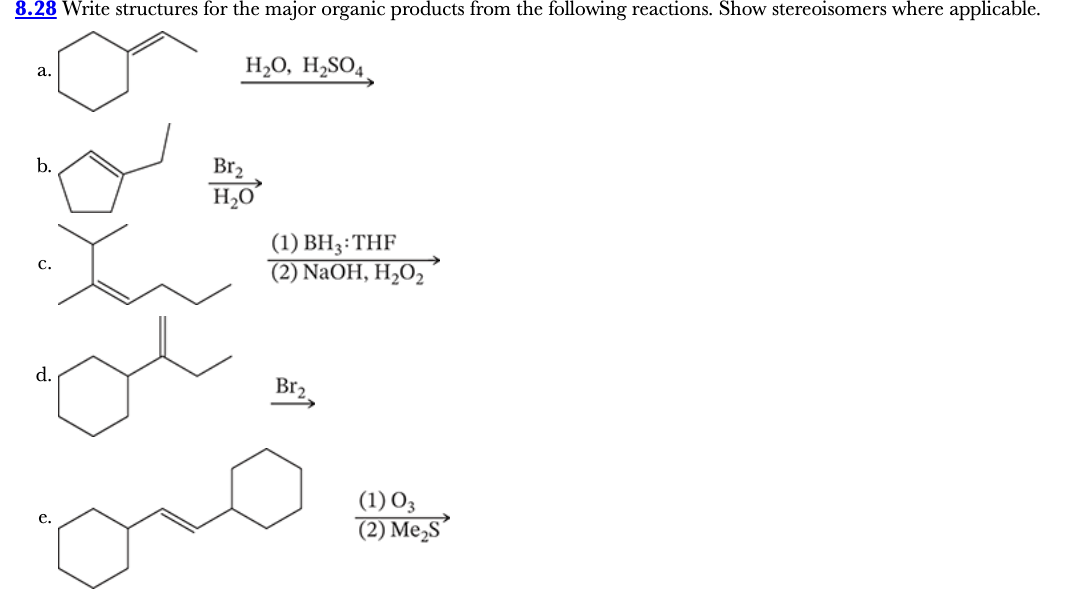
8.28
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable.
8.29
Give the structure of the products that you would expect from the reaction of 1-butyne with:
a. One molar equivalent of Br2
b. One molar equivalent of HBr
c. Two molar equivalents of HBr
d. H2 (in excess)/Pt
e. H2, Ni2B (P-2)
f. NaNH2 in liquid NH3, then CH3I
g. NaNH2 in liquid NH3, then (CH3)3CBr
8.30
Give the structure of the products you would expect from the reaction (if any) of 2-butyne with:
a. One molar equivalent of HBr
b. Two molar equivalents of HBr
c. One molar equivalent of Br2
d. Two molar equivalents of Br2
e. H2, Ni2B (P-2)
f. One molar equivalent of HCl
g. Li/liquid NH3
h. H2 (in excess), Pt
i. Two molar equivalents of H2, Pt
j. Hot KMnO4, HO−, then H3O+
k. O3, then HOAc
l. NaNH2, liquid NH3
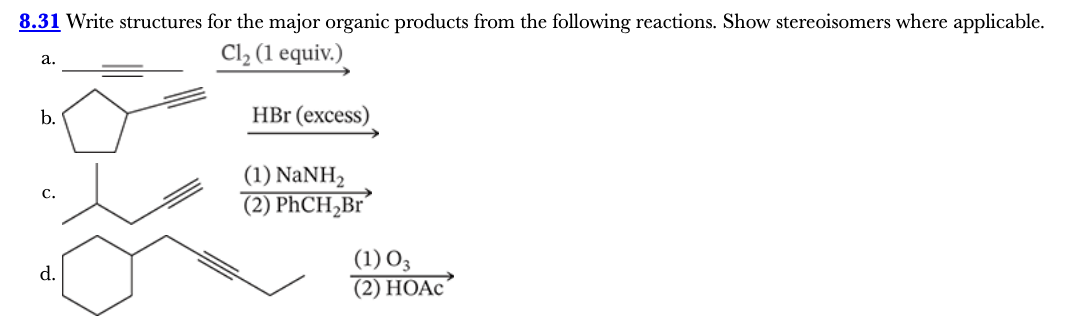
8.31
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable.

8.39
Write stereochemical formulas for all of the products that you would expect from each of the following reactions. (You may find models helpful.)
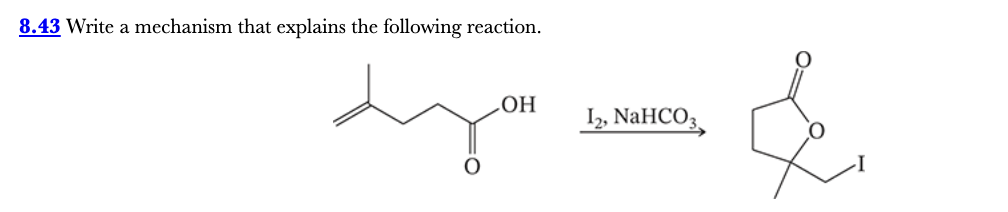
8.43
Write a mechanism that explains the following reaction.
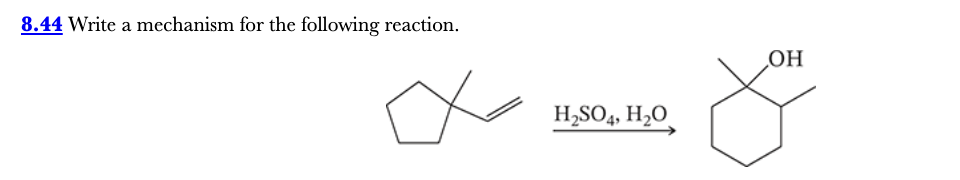
8.44
Write a mechanism for the following reaction.

8.59
For each of the following, identify the product (represented by A, B, and C) that would be formed through the indicated sequence of steps from the given starting material.
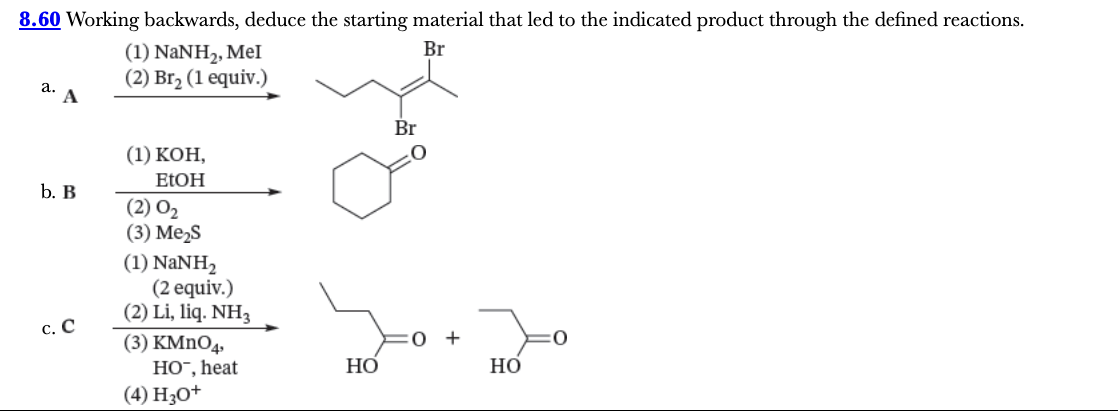
8.60
Working backwards, deduce the starting material that led to the indicated product through the defined reactions.
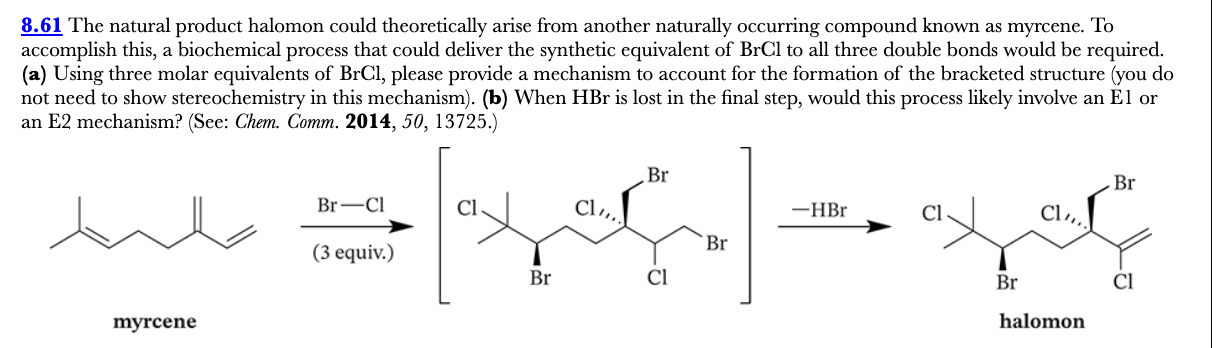
8.61