Organic CHEM
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
why are carbon compounds so stable and numerous
tetravalent C atoms have no vacant orbitals or lone pairs
C bonds with many different atoms and forms double and triple bonds
C-H bonds have low polarity
what are newman projections
the way of drawing C atoms to show the direction of bonds
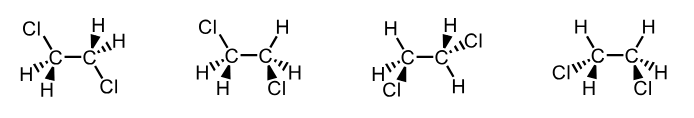
(know how to use IUPAC naming system)
what are the products of combustion of alkanes
CO2 & water
what is bond dissociation energy
a measure of how much energy is required to break a bond to give neutral fragments (radicals)
what is the heat of combustion
the heat released when a compound undergoes complete combustion in the presence of excess oxygen
put the following bonds into order of increasing bond dissociation energy, and give a reason for its placement
C≡C, C≡N, N≡N
C-C, C-N, C-O
C-H, N-H, O-H
C=C, C=N, C=O
N-N, N-O, O-O, O-Cl
N-N, N-O, O-O, O-Cl (all have lone pairs = weak)
C-C, C-N, C-O (single bonds)
C-H, N-H, O-H (stronger single bonds bc shorter bond length, bc bonded to smaller atom)
C=C, C=N, C=O (double bonds)
C≡C, C≡N, N≡N (triple bonds)
When is a reaction energetically favourable
(be able to estimate if reactions are energetically favourable using BDEs)
when it is exothermic it is favourable
what are constitutional isomers
molecules with the same molecular formula but different structural formulas
(normal isomers)
what are the staggered and eclipsed structures of ethane
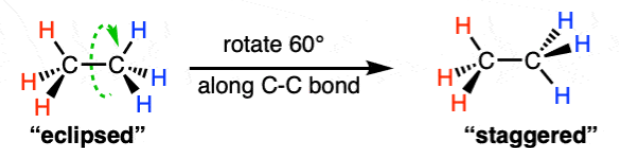
why are alkanes chemically unreactive
The C–C and C–H bonds are very strong.
The C & H have full valence shell configurations.
They do not have non-bonding pairs of electrons.
They do not have polarised bonds.
They are not acidic or basic
what is a functional group
Groups of atoms that include CC multiple bonds, or other elements
what are free radicals
species that have an unpaired electron
what is the difference between an ion and a radical
ions have a charge due to missing or having an extra electron
while radicals are a neutral species that simply has an unpaired electron
draw the mechanism of alcohol acting as a base in equilibrium
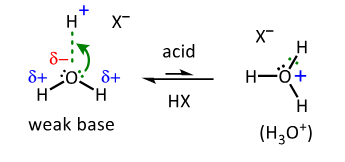
why are oxygen atoms in alcohols and ethers much less nucleophilic than N atoms in amines
the lone pair in O is held more tightly bc O is more electronegative than N, so O gives up its electron to the electrophile a lot less readily
draw the mechanism for alcohols acting as an acid in the presence of a strong base
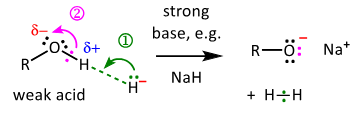
can N-H or C-H compounds act as acids in a similar way to alcohols? why / why not?
no, bc the N or C atoms are not electronegative enough to stabilise the negative charge that the reaction would cause them to have.
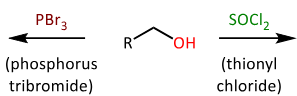
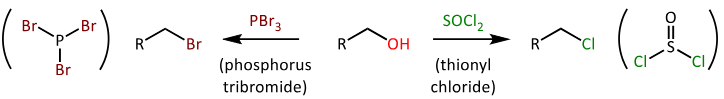
draw the EQUATIONs for the conversion of alcohols into alkylhalides
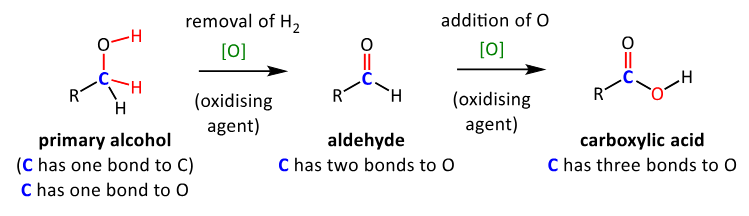
draw the equation of oxidation of a primary alcohol
define enantiomer
a non-superimposable mirror image of the molecule
what are diastereomers
stereoisomers, that are not mirror images of each other
What are the Cahn-Ingold-Prelog Rules, and know how to apply them
they are rules that tell you how to assign the letters R or S to the configuration of a chiral center of a molecule
what are the steps in cahn ingold prelog rules (lecture 5 pg14)
align the molecule so that the two dashed and wedged bonds are “zig-zag”
assign priorities to the bonded atoms in order of decreasing atomic number. If there are two of the same atom look at the next atoms in the chain until a difference is found. (in the chain, multiples of the same atom add to the priority, eg having 2 O vs 1. and multiple bonds are treated as the same number of single bonds)
orient the molecule so the lowest priority group is facing back
draw an arrow following the order of priority
if arrow is clockwise then = R (R for right)
if the arrow is anti-clockwise then = S
how do we detect chirality
polarimetry, chiral molecules rotate polarised light in different directions to each other (not related to the direction of the arc in the R S naming system)