Chem 161- exam 2
1/51
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Beta Decay
When an element loses a beta particle (0/-1e)- atomic # goes up by one (neutron→ proton)
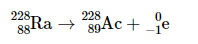
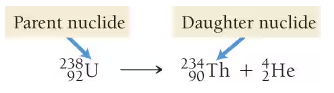
Alpha Decay
When an element emits a Helium nucleus (4/2 He)
Gamma emission
a form of em radiation usually emitted with other forms of decay with no mass and no charge
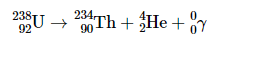
Positron emission
In positron emission, a proton emits a positron and becomes a neutron.
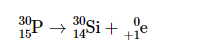
electron capture
when a nucleus assimilates an electron from an inner orbital of its electron cloud.
How to find energy from frequency? (v=f)
E=hv (h=planck’s constant)
What is planck’s constant?
6.626 × 10^-34 J
What is c?
Speed of light- 3 × 10^8
What is the relationship between speed of light, wavelength, and frequency?
c = λ ν
What is λ?
Lambda- wavelength
How to derive energy from wavelength?
E = h (c/ λ).
How to find energy put out or absorbed from n=levels? (orbitals)
change in energy-E = -2.178 x 10-18 J [1/nf^2 – 1/ ni^2 ]
What is the de Broglie equation?
λ = h/(mv)
Why is it called the the de Broglie equation?
The de Broglie equation is named after French physicist Louis de Broglie, who proposed in 1924 that electrons have both wave-like and particle-like properties, similar to light.
What is Heisenberg’s uncertainty principle?
“the product of Δx and mΔv must be greater than or equal to a finite number (h/4π). In other words, the more accurately you know the position of an electron (the smaller Δx), the less accurately you can know its velocity (the bigger Δv) and vice versa.”
Where to find the #n for quantum numbers?
Left side of the periodic table (period). ex: 5p
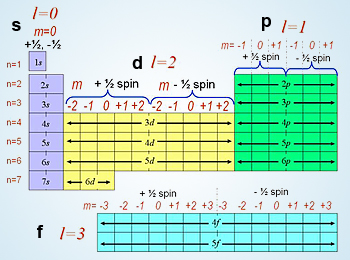
What is the rule for accepted l quantum numbers?
any integer between 0 and n - 1
What is the rule for ml quantum numbers?
ml can be between -l and +l (l=L, not 1)
What is the rule for ms quantum numbers?
ms = -1/2, +1/2
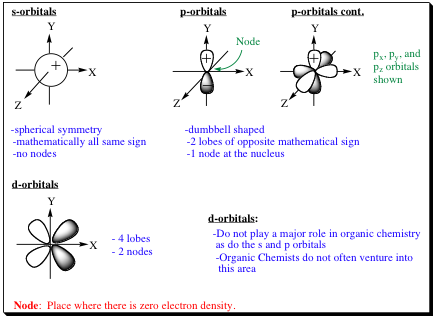
how does the orbital dictate shape?
s= sphere, p= dumbell, d= complex dumbell, f= more complex
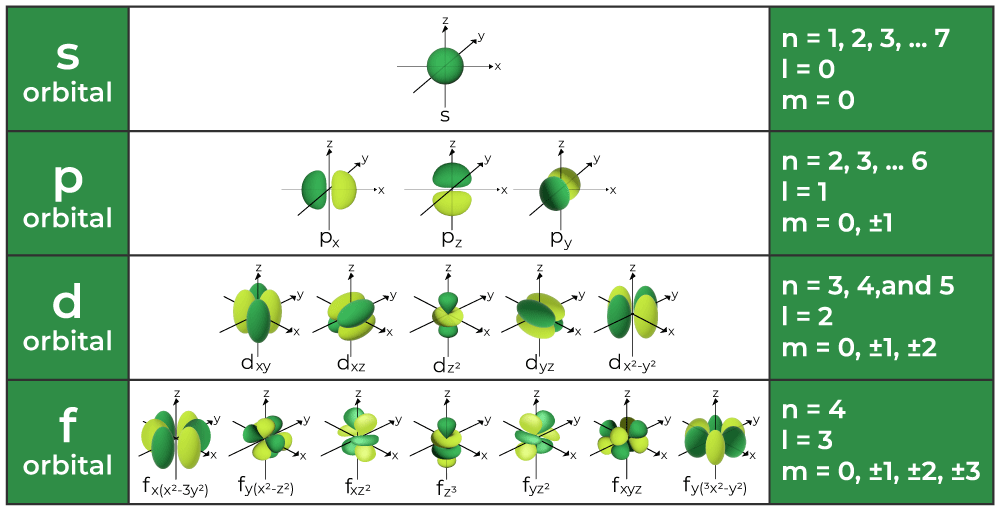
Orbitals vs bohr’s orbits?
Bohr predicted 1 orbital per energy level, There are actually more than that. Ex: 4 orbitals in n=2 (one 2s and three 2p orbitals)
What are energy levels?
1, 2, 3, 4 etc. (n=#) In chemistry, energy levels, also known as electron shells, are the fixed distances from an atom's nucleus where electrons can be found.
What are nodes?
The regions or spaces around the nucleus where the probability of finding an electron is zero are called nodes. (1/2 # of lobes)
How to predict type of radioactive decay?
If the atomic number is greater than 82, alpha decay is the most likely prediction.
If the atomic number is less than 82, calculate the neutron-to-proton ratio. If the energy ratio is too high, beta decay is the prediction.
If it is radioactive
Pauli’s Exclusion Principle
Can’t have the same 4 quantum numbers, they must have opposite spins (ms #’s)
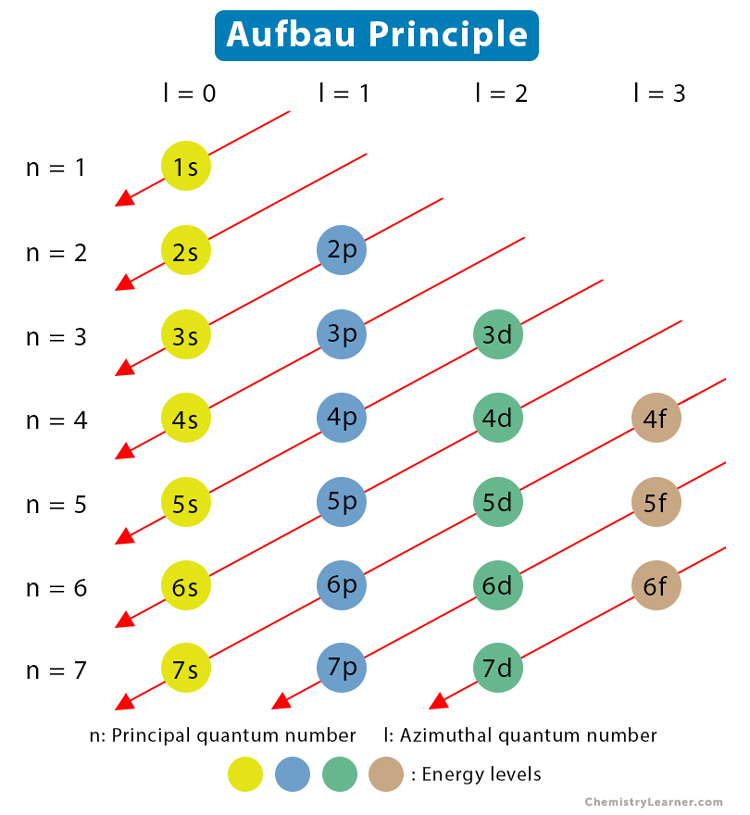
Aufbau principle
electrons in an atom's ground state fill atomic orbitals in order of increasing energy level.
Hund’s rule
Can’t start pairing until everything in an orbital is filled
Electron Spin concept
4th quantum number, dictates direction of electron spin.
How to write electron configs (long way)?
Start at 1s2 and use the chart to go until the exponents add up to the electron numbers (atomic numbers)
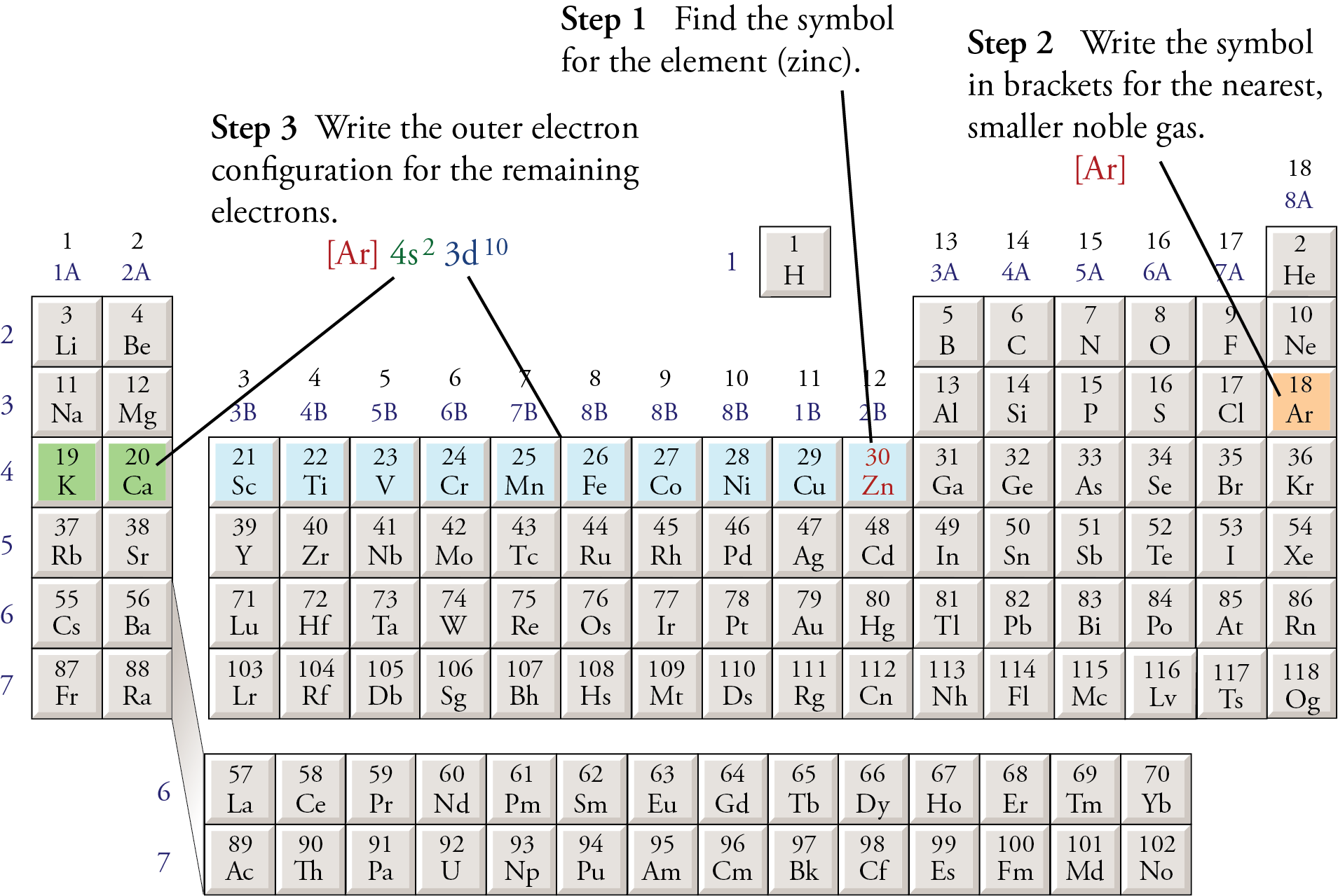
How to write condensed electron configs?
Use the closest noble gas config and start there
Then pick up where the noble gas ends
keep goings until exponents + electrons in the noble gas add up to the atomic number (IF THERE IS A + SOMETHING OR - SOMETHING PAY ATTENTION TO # OF ELECTRONS)
paramagnetic
has unpaired electrons
diamagnetic
everything paired
Difference between behavior of diamagnetic/paramagnetic?
Diamagnetic materials are repelled by magnetic fields, while paramagnetic materials are weakly attracted to them. When exposed to a magnetic field, diamagnetic materials create an induced magnetic field in the opposite direction, while paramagnetic materials create an induced magnetic field in the same direction.
Valence electrons vs core (in electron config)?
Electrons in the outer energy level vs everything else

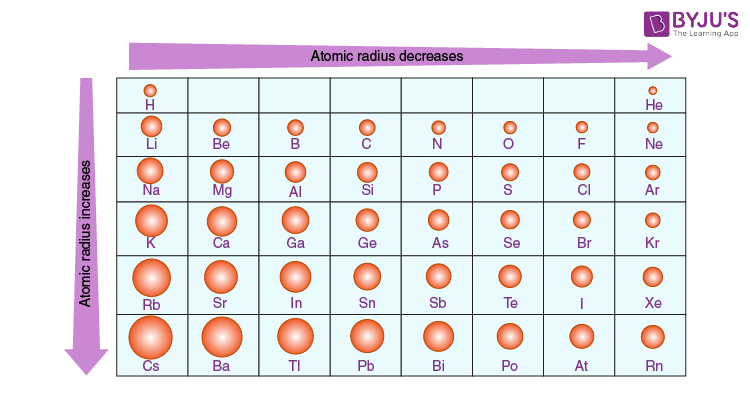
How to determine atomic size on periodic table?
Up/down= increases, left right=decreases; bottom left = biggest, top right=smallest
How to determine electron affinity periodic table?
down/up= increases, left right=increases; bottom left = least, top right=most (from group 3-17) (greater electron affinity = more negative)
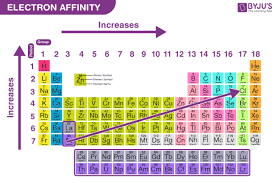
Electron affinity noble gasses?
Noble gases on the periodic table have an electron affinity of zero or less than zero.
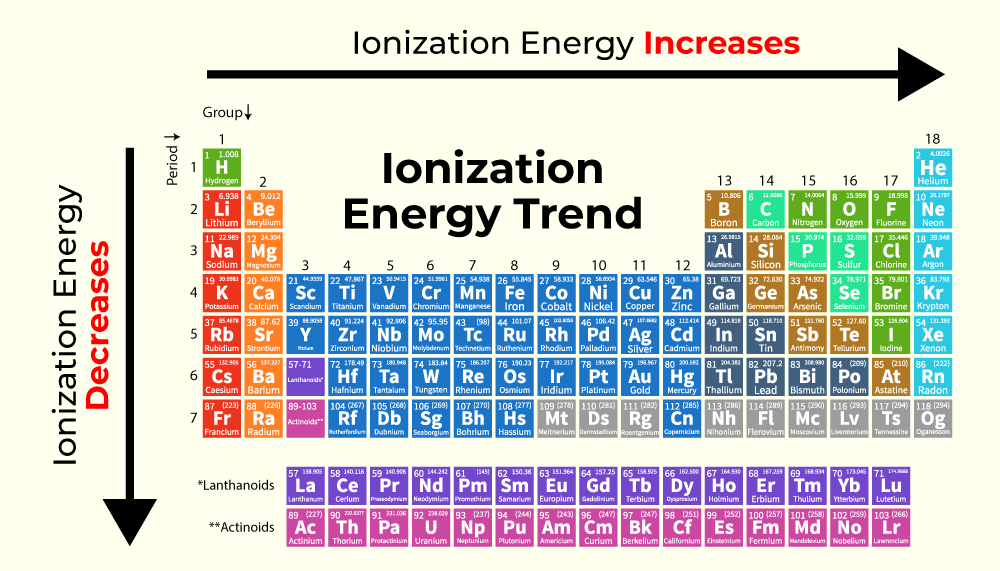
How to determine ionization energy periodic table?
the minimum amount of energy needed to remove an electron from an isolated atom, molecule, or positive ion. Decreases up/down, increases left/right.
Metals- identify
Metals are mostly located in groups 1-13- silvery - metallic luster
Non-metals identify
and nonmetals are mostly starting from group 14 to 18.- brittle
Metalloids Identify
on the steps of the periodic table, cross between identities of metals/nonmetals. Boron to Astatine
Exo/Endo reactions
We can tell if the reaction is exo or endo depending if the reaction is forming bonds or breaking. In an exothermic reaction, heat is being released and bonds are being formed. But in endothermic reactions, bonds are being broken because energy is being put in to break the bonds.
Would n=1- n=3 be exo or endo ?
Endo- requires energy to be absorbed
Would n=5- n=1 be exo or endo ?
Exo- requires energy to be released
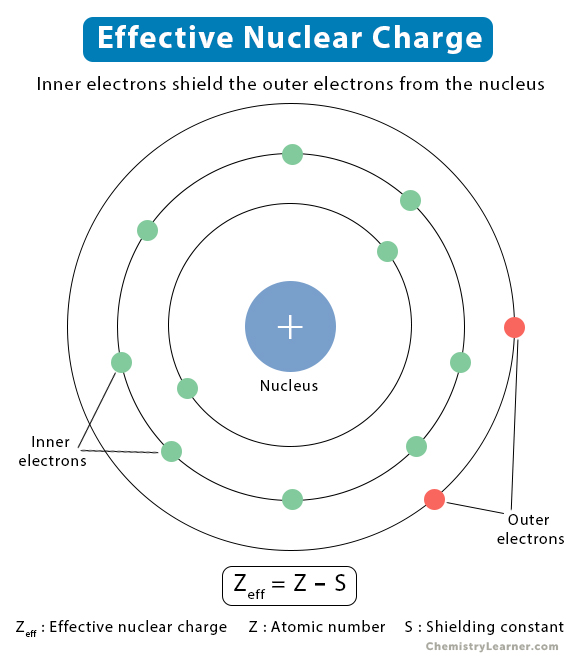
Zeff
Effective Nuclear Charge- The actual nuclear charge experienced by an electron, defined as the charge of the nucleus plus the charge of the shielding electrons. (The pos charge that pulls electrons to the nucleus)
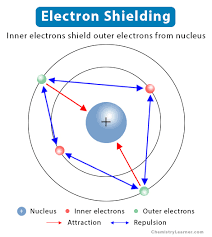
Shielding
The attraction between an electron and the nucleus of an atom with more than one electron. Shielding occurs when the core electrons repel the outer electrons, which reduces the nucleus' effective charge on the outer electrons. This means that the nucleus has "less grip" on the outer electrons.
penetration of electrons
electron penetration is the ability of an electron to get close to the nucleus by penetrating other shells and subshells. The extent of penetration depends on the subshell (l) and shell (n) of the electron. s orbital has max penetration effect
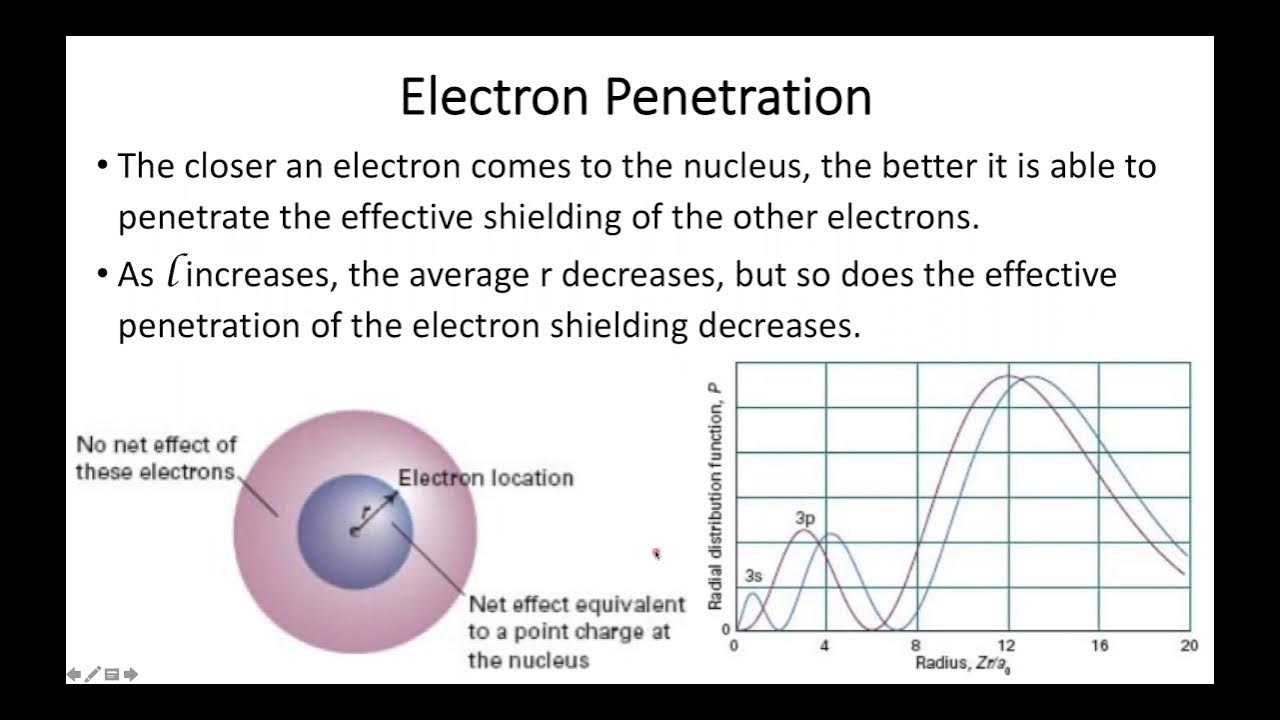
Electron-electron repulsions
Electron-electron repulsion occurs when the negative charge of one electron repels other electrons, similar to magnets with the same poles facing each other. This repulsion is caused by classical Coulomb forces and when electrons have parallel spins.
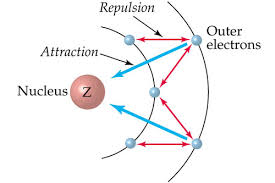
Octet Rule
The octet rule is a chemical rule of thumb that states that atoms are most stable when their valence shells have eight electrons. This gives the atom the same electronic configuration as a noble gas.
isoelectronic configurations
Isoelectronic configurations are when two or more atoms or ions have the same electronic configuration. Isoelectronic species are expected to have similar chemical properties and predictability in their reactions.
For example:
Re²+ and Hf^- both have 73 electrons (same config)
coloumb’s law
The closer two charges are, the stronger the force between them
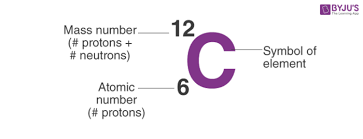
How to write an element
Mass #/Atomic # Symbol