chem 1: gases, liquids, and solids
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Ideal gas law
PV = nRT
Charles Law
V1/T1 = V2/T2
Gay Lussacs Law
P1/T1 = P2/T2
1 mol gas is equal to
22.4 L
Boyle’s Law
P1V1 = P2V2
Finish units of conversion: 1 atm =
1 atm = 760 torr = 760 mm Hg = 101.3 kPa = 14.7 PSI
What is STP?
Standard Temperature and Pressure
Kinetic Molecular Theory states that:
Gas particles are in constant, random motion, have negligible volume, and collisions are perfectly elastic
What is a gas?
A state of matter that has no fixed shape, takes the shape of its container, and has particles that are far apart and move freely.
Assumptions about gases
particles are points in random motion
don’t interact
have negligible volume and collisions are elastic.
moles represent how many ____ are in the container
particles
PV = nRT, what does each variable stand for?
P = pressure, V = volume, n = number of moles, R = ideal gas constant, T = temperature
What is the relationship between pressure and volume? They are _____ proportional
As pressure increases, volume decreases, and vice versa, demonstrating an inverse relationship, described by Boyle's Law.
Inversely
Volume are temperature are _____ proportional, as described by _____ Law.
directly, Charles’
0 Celcius =
273.15 Kelvin
combined gas law
(P1V1)/T1 = (P2V2)/T2
Avogrado’s Law
V1/n1 = V2/n2
1 mol gas =
22.4 L
R =
0.08206 L atm/mol K
What is root mean square velocity?
the measure of the speed of atoms in a gas
root mean square velocity formula and constants
V = sqrt(3RT/M), R = 8.3145 J/mol K , M = kg/mol
1 kg is how many g?
1000 g
1 L is how many mL?
1000 mL
velocity increases by the square root of the
temperature
According tot the RMS equation, when molarity goes up,
velocity goes down
If H2 has a speed of 200 m/s, what is the speed for O2?
50 m/s (yellow represents molar mass in screenshot)
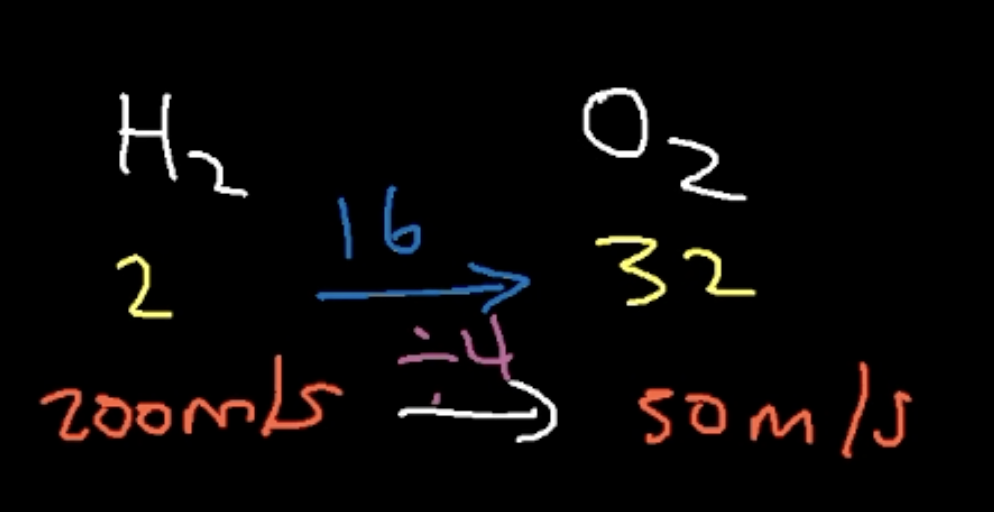
average of gas molecules is dependent on
temperature
rate of infusion is…
how fast a gas can escape from a container through a hole
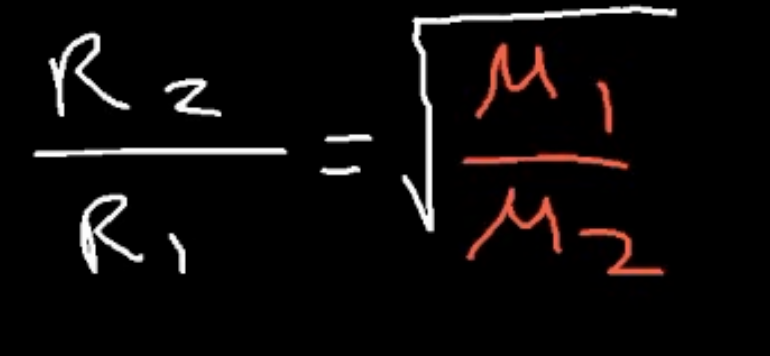
rate of effusion formula
refer to image
heavy gas molecules move____, a larger molar mass means a ____ rate of effusion
slower, slower M⬆V⬇
rate of diffusion with time
refer to image
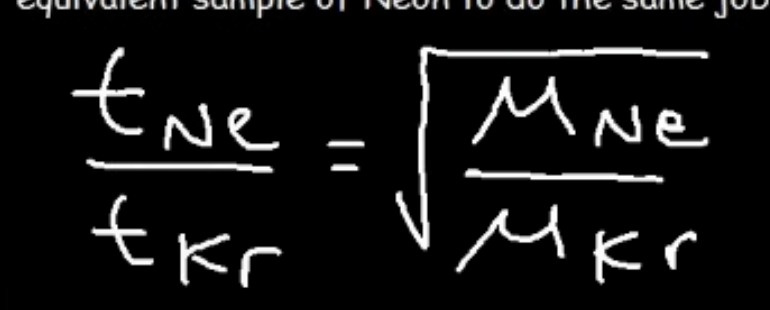
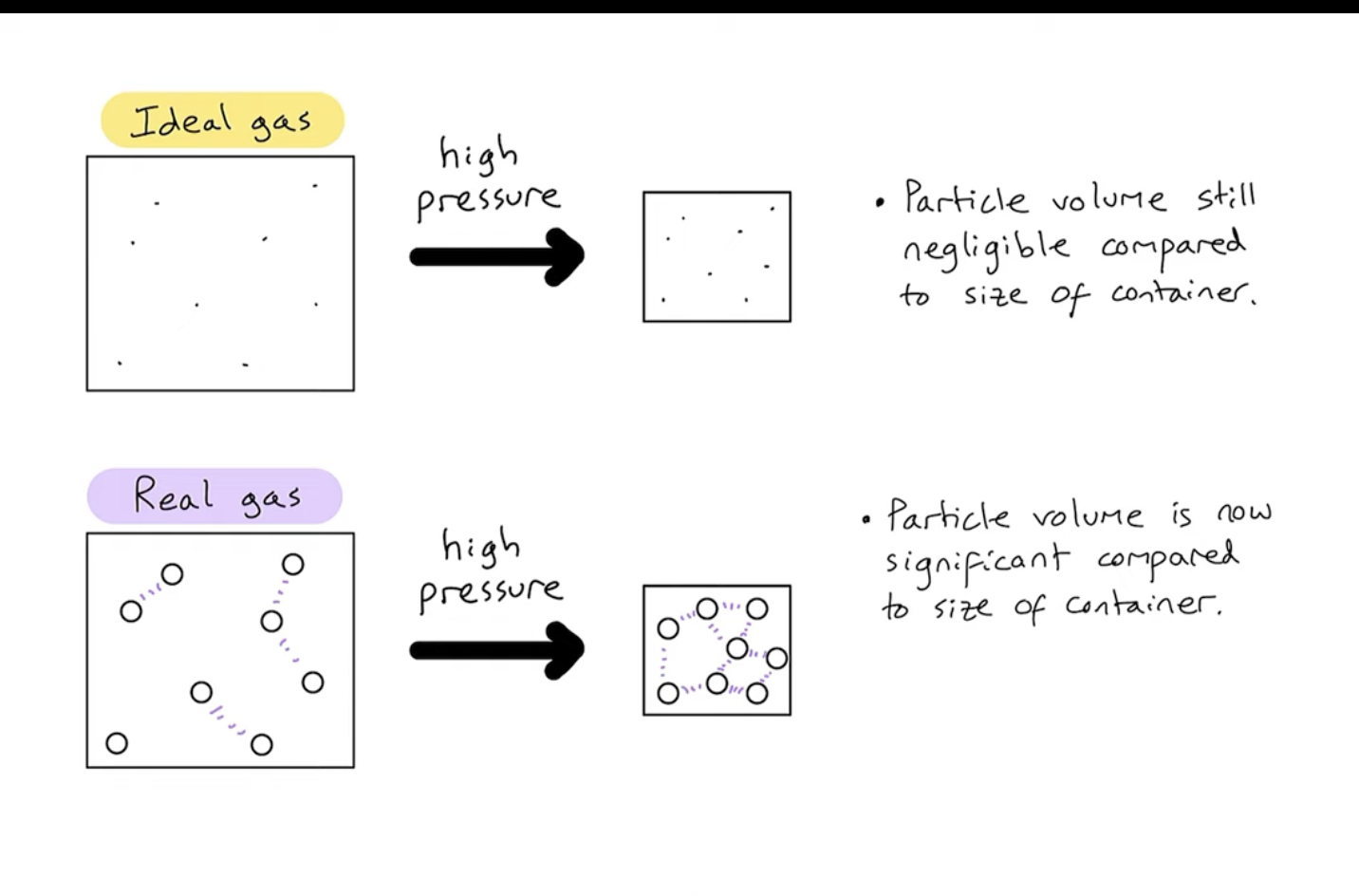
What makes a real gas closer to an ideal gas?
low pressure and high temperature
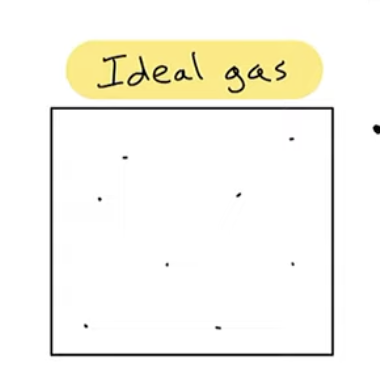
Assumptions about an ideal gas
particles have negligible volume (= 0)
particles have intermolecular forces
described by PV = nRT.
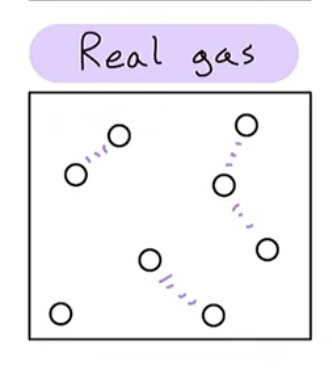
Assumptions about a real gas
particles have measurable volume
particles have intermolecular forces
at most conditions we can use PV=nRT
real gases tend to have:
high pressure and low temperature
Making a container smaller makes the pressure
higher