Properties of period 3 elements and their oxides
1/57
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
Which is more reactive: sodium or magnesium? Explain your answer.
Sodium is more reactive than magnesium. This is because sodium forms Na^+ ions whereas magnesium forms Mg²+ ions, it takes more energy to remove two electrons than one so more energy is required for magnesium to react, making sodium more reactive.
Give the balanced symbol equation for the reaction between sodium and water.
2Na (s) + 2H2O (l) → 2NaOH (aq) + H2 (g)
Describe and explain any observations made during the reaction between sodium and water.
Vigorous reaction, fizzing (hydrogen gas being produced), ball (unreacted sodium).
Give the balanced symbol equation for the reaction between magnesium and cold water.
Mg (s) + 2H2O (l) → Mg(OH)2 (aq) + H2 (g)
Why is magnesium hydroxide weakly alkaline?
Magnesium hydroxide is sparingly soluble so there aren’t many OH^- ions when it is in a solution.
Describe and explain any observations made in the reaction between magnesium and cold water.
Slow.
True or false? Magnesium reacts much faster with steam than with cold water.
True.
Give the balanced symbol equation for the reaction between magnesium and steam.
Mg (s) + H2O (g) → MgO (s) + H2 (g)
What is one difference between the products of the reaction between magnesium and cold water and magnesium and steam?
The reaction between magnesium and cold water produces magnesium hydroxide whereas the reaction between magnesium and steam produced magnesium oxide.
What do period 3 elements form (in general) when they react with oxygen?
Period 3 oxides.
Give the balanced symbol equation for the reaction between sodium and oxygen. What is the name of the product?
2Na (s) + ½ O2 (g) → Na2O (s)
Sodium oxide.
Give the balanced symbol equation for the reaction between magnesium and oxygen. What is the name of the product?
Mg (s) + ½ O2 (g) → MgO (s)
Magnesium oxide.
Give the balanced symbol equation for the reaction between aluminium and oxygen. What is the name of the product?
2Al (s) + 1 ½ O2 (g) → Al2O3 (s)
Aluminium oxide.
Give the balanced symbol equation for the reaction between silicon and oxygen. What is the name of the product?
Si (s) + O2 (g) → SiO2 (s)
Silicon dioxide.
Give the balanced symbol equation for the reaction between phosphorous and oxygen. What is the name of the product?
P4 (s) + 5O2 (g) → P4O10 (s)
Phosphorous (V) oxide.
Give the balanced symbol equation for the reaction between sulfur and oxygen (standard conditions). What is the name of the product?
S (s) + O2 (g) → SO2 (g)
Sulfur dioxide.
What is the product of the reaction between sulfur and oxygen at higher temperatures and with a catalyst? Give the balanced symbol equation for how this is formed from sulfur dioxide.
SO3 (sulfur trioxide).
2SO2 (g) + O2 (g) → ← 2SO3 (g)
Give the general trend of the reactivity of the period 3 oxides with air (oxygen) going across period 3.
Reactivity decreases.
Describe how sodium oxide reacts with air.
Very fast.
Describe how magnesium oxide reacts with air.
Very fast.
Describe how aluminium oxide reacts with air generally.
Slowly.
How can you make the reaction between aluminium oxide and air faster?
Turn the aluminium oxide into powder (increase its surface area).
Describe how silicon dioxide reacts with air.
Slowly.
Describe how phosphorous (V) oxide reacts with air.
It spontaneously combusts.
Describe how sulfur dioxide reacts with air.
It steadily burns.
Describe the general trend in the melting points of period 3 oxides across period 3.
Melting points of period 3 oxides decreases across period 3.
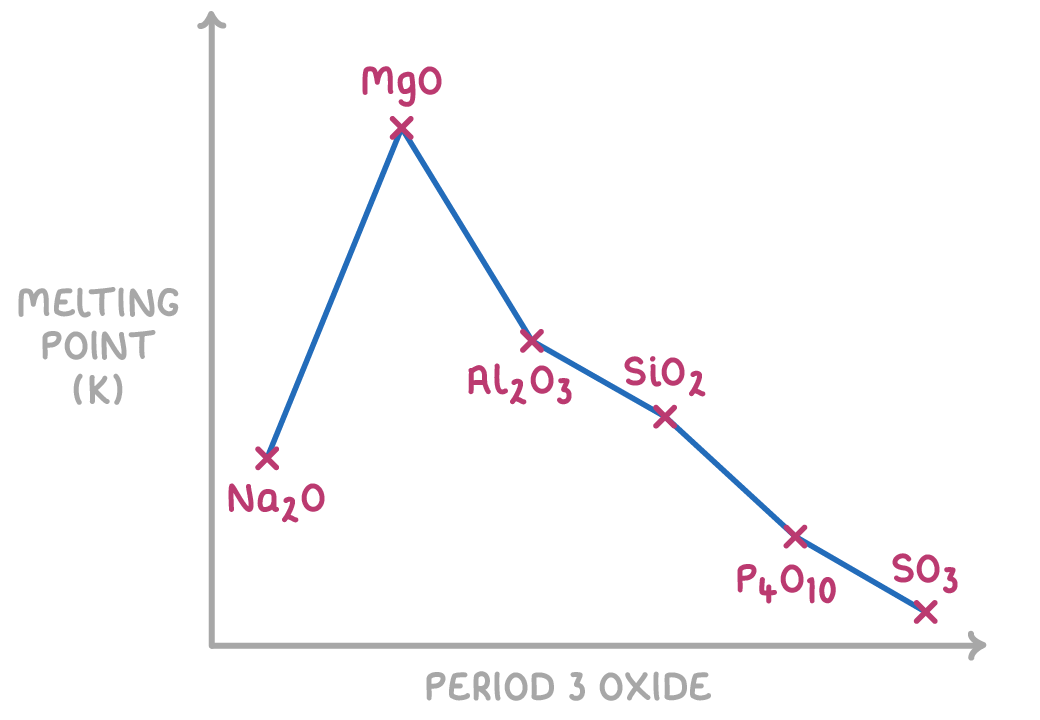
Which period 3 oxides form ionic lattices?
Sodium oxide, magnesium oxide, aluminium oxide.
Why do sodium oxide, magnesium oxide, and aluminium oxide have the highest melting points out of all the period 3 oxides?
They are ionic lattices meaning they contain lots of strong attractive forces that require lots of energy (high temperatures) to break.
Why does magnesium oxide have a greater melting point than sodium oxide?
Magnesium forms 2+ ions which are attracted more strongly to (2-) oxygen, whereas sodium forms 1+ ions, so more energy is required to break the bonds in magnesium.
Why does aluminium oxide have a lower melting point than magnesium oxide?
The Al³+ ions distort the electron cloud meaning there is some covalent character as well as the expected ionic interactions (interferes with ionic bonding), so less energy is required to break the bonds.
What is covalent character?
When there is a partial sharing of electrons in an ionic bond (somewhat in between an ionic and covalent bond).
Why does silicon dioxide have the highest melting point out of all the non-metal period 3 oxides?
It is macromolecular (giant covalent structure), many strong covalent bonds require lots of energy to break.
Why do phosphorus pentoxide (P4O10) and sulfur dioxide have the lowest melting points out of period 3 oxides?
They are simple molecular substances meaning only weak intermolecular forces have to be broken, which doesn’t require much energy, to melt them.
True or false? Period 3 ionic oxides form acidic solutions when added to water.
False. PERIOD 3 IONIC OXIDES FORM ALKALINE SOLUTIONS WHEN ADDED TO WATER.
Why do ionic oxides form alkaline solutions when added to water?
The O²- ion in them accepts H^+ ions (protons) from water, forming OH^- ions.
Give the balanced symbol equation for the reaction between sodium oxide and water. What is the pH of the solution formed?
Na2O (s) + H2O (l) → 2NaOH (aq)
pH = 12-14
Does sodium oxide dissolve readily in water?
Yes.
Does magnesium oxide dissolve readily in water?
No, magnesium oxide dissolves sparingly in water.
Give the balanced symbol equation for the reaction between magnesium oxide and water. What is the pH of the solution formed?
MgO (s) + H2O (l) → Mg(OH)2 (aq)
pH = 9-10
True or false? Period 3 simple covalent oxides dissolve in water to form acidic solutions.
True.
Give two balanced symbol equations to show how phosphorus pentoxide reacts with water to form an acidic solution. What is the pH of the solution formed?
P4O10 (s) + 6H2O (l) → 4H3PO4 (aq) (forms phosphoric acid)
H3PO4 (aq) → 3H^+ (aq) + PO4³- (aq) (phosphoric acid dissociates)
pH = 0-2
Give two balanced symbol equations to show how sulfur dioxide reacts with water to form an acidic solution. What is the pH of the solution formed?
SO2 (g) + H2O (l) → H2SO3 (aq) (sulfurous acid)
H2SO3 (aq) → 2H^+ (aq) + SO3²- (aq) (sulfurous acid dissociates)
pH = 0-2
Give two balanced symbol equations to show how sulfur trioxide reacts with water to form an acidic solution. What is the pH of the solution formed?
SO3 (l) + H2O (l) → H2SO4 (aq) (sulfuric acid)
H2SO4 (aq) → 2H^+ (aq) + SO4²- (aq) (sulfuric acid dissociates)
pH = 0-2
True or false? Silicon dioxide is water soluble.
False. SILICON DIOXIDE IS INSOLUBLE IN WATER.
Why is silicon dioxide insoluble in water?
It is a giant covalent structure so lots of energy is required to break the strong covalent bonds in it.
True or false? Aluminium oxide is insoluble in water.
True.
True or false? Aluminium oxide is amphoteric.
True.
True or false? Silicon dioxide reacts with an acid to form a salt so is therefore classed as a base.
False. SILICON DIOXIDE REACTS WITH A BASE TO FORM A SALT SO IS THEREFORE CLASSED AS AN ACID.
What is the general word equation for the reaction between an acid and a base?
Acid + base → water + salt
Give the balanced symbol equation for the reaction between hydrochloric acid and magnesium oxide.
2HCl (aq) + MgO (s) → MgCl2 (aq) + H2O (l)
Give the balanced symbol equation for the reaction between sodium oxide and sulfuric acid.
H2SO4 (aq) + Na2O (s) → Na2SO4 (aq) + H2O (l)
Give the balanced symbol equation for the reaction between silicon dioxide and sodium hydroxide.
SiO2 (s) + 2NaOH (aq) → Na2SiO3 (aq) + H2O (l)
Give the balanced symbol equation for the reaction between phosphorus pentoxide and sodium hydroxide.
P4O10 (s) + 12NaOH (aq) → 4Na3PO4 (aq) + 6H2O (l)
Give the balanced symbol equation for the reaction between sulfur dioxide and sodium hydroxide.
SO2 (g) + 2NaOH (aq) → Na2SO3 (aq) + H2O (l)
Give the balanced symbol equation for the reaction between sulfur trioxide and sodium hydroxide.
SO3 (g) + 2NaOH (aq) → Na2SO4 (aq) + H2O (l)
What is unusual about the reaction between aluminium oxide and a base?
No water is produced, only a salt.
Give the balanced symbol equation for the reaction between aluminium oxide and sodium hydroxide.
Al2O3 (s) + 2NaOH (aq) + 3H2O (l) → 2NaAl(OH)4 (aq)
Give the balanced symbol equation for the reaction between aluminium oxide and sulfuric acid.
Al2O3 (s) + 3H2SO4 (aq) → Al2(SO4)3 (aq) + 3H2O (l)