Formulas and Definations- 4
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Energy (Defination)
Energy means the ability to do work.
Principle of conservation of energy (Defination)
the principle of conservation of energy states that energy cannot be created nor destroyed; if energy disappears in one form. it re-appears in another.
Kinetic Energy
Eκ = ½×m×v²
Kinetic Energy= ½× mass× velocity ²
Eκ= Kinetic Energy (J)
m= mass (kg)
v= velocity (ms⁻¹)
Units= kgm²s⁻² or J
Potiential Energy
Ep= m×g×h
Potiential energy= Mass× gravity × height
Ep= Potiential Energy ( J)
m= mass (kg )
h= height ( m)
g= acceleration due to gravity (ms⁻2)
Types of energy
Nuclear
Thermal
Light
Kinetic
Potiential
Sound
Mechanical
Chemical
Heat and matter thoeries
Caloric theory and kinetic theory
Kinetic Theory
All matter is made up of small particles in continual state of motion. As a result of this motion heat is formed. Friction causes this heat due to mechanical energy being converted to kinetic energy in the substance.
Caloric Theory
Caloric Theory states that heat was fluid called caloric. Calcoric was created from friction between particles and this fluid moved from hot bodies to cold bodies when in contact
Kinetic theory examples (2)
Eg.
1) Brownian Motion- The erratic movement of particles because of thier interaction with other molecules
2) Diffusion in liquids- movement of perticles from a place of high concentration to a low concentration along a concentration gradient.
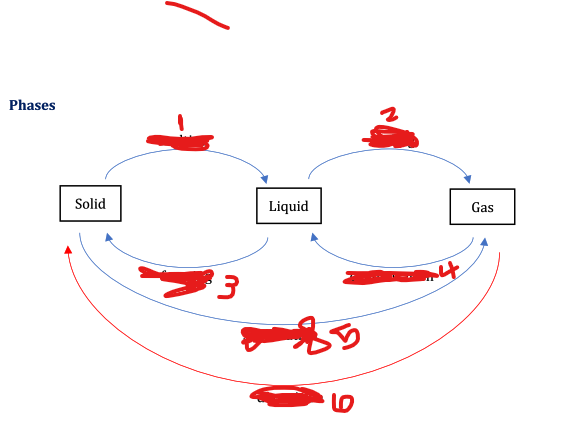
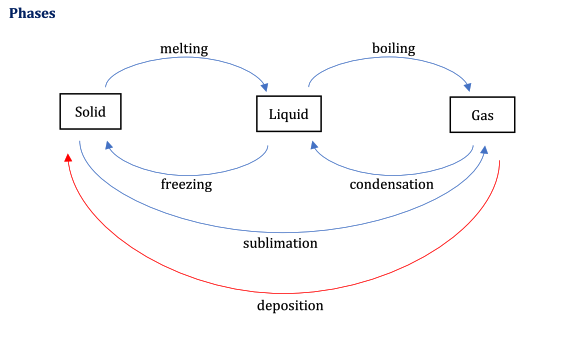
States of matter ( Label 1 to 6 be sure to mention the changes in state)
1) Melting- Solid to liquid
2) Boiling- Liquid to gas
3) Freezing- Liquid to solid
4) Condensation- Gas to liquid
5) Sublimation- Solid to gas
6) Deposition- Gas to solid
Types of thermometers
1) Laboratory
2) Mercury
3) Clinical/ Medical
4) Theromocouple
5) Alcohol
Laboratory Thermometer ( 3 points- Use, range, why its used and how it works, restriction)
Laboratory theromometers are used in scientific experiments and have a range of −10 celsius to 110 celsius.
Its used to measure the temputure of liquids during experiments because it has no constriction meaning the liquid moves up and down freely.
You must read the theromometer while still in the substance because the liquid within the thermometer falls immediately when removed.
Mercury Thermometer
Mercury Thermometers uses mecury, a metal liquid because its shing and easy to see and expands evenly making the reading more accurate. It has a range of −39 celsius to 357 celsius and are used in labs or medicine.
Alcohol Thermometer ( usual features, use and why, range)
Alcohol theromometers use alcohol that is usually dyed. These thermometers are used to measure very low temputures because alcohol freezes lower than mercury and its range is below -100 celsius.
Celsius to Kelvin
Celsius value+ 273 = Kelvin value
fahrenheit to kelvin
Fahrenheit value +255 = Kelvin value