3.2.5.2 substitution reactions + 3.2.5.3 shapes of complex ions
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
ligands form what with the transition metal ion?
coordinate bonds
how many coordinate bonds are usually formed by the ligand to the transition metal?
usually 2, 4 or 6 bonds
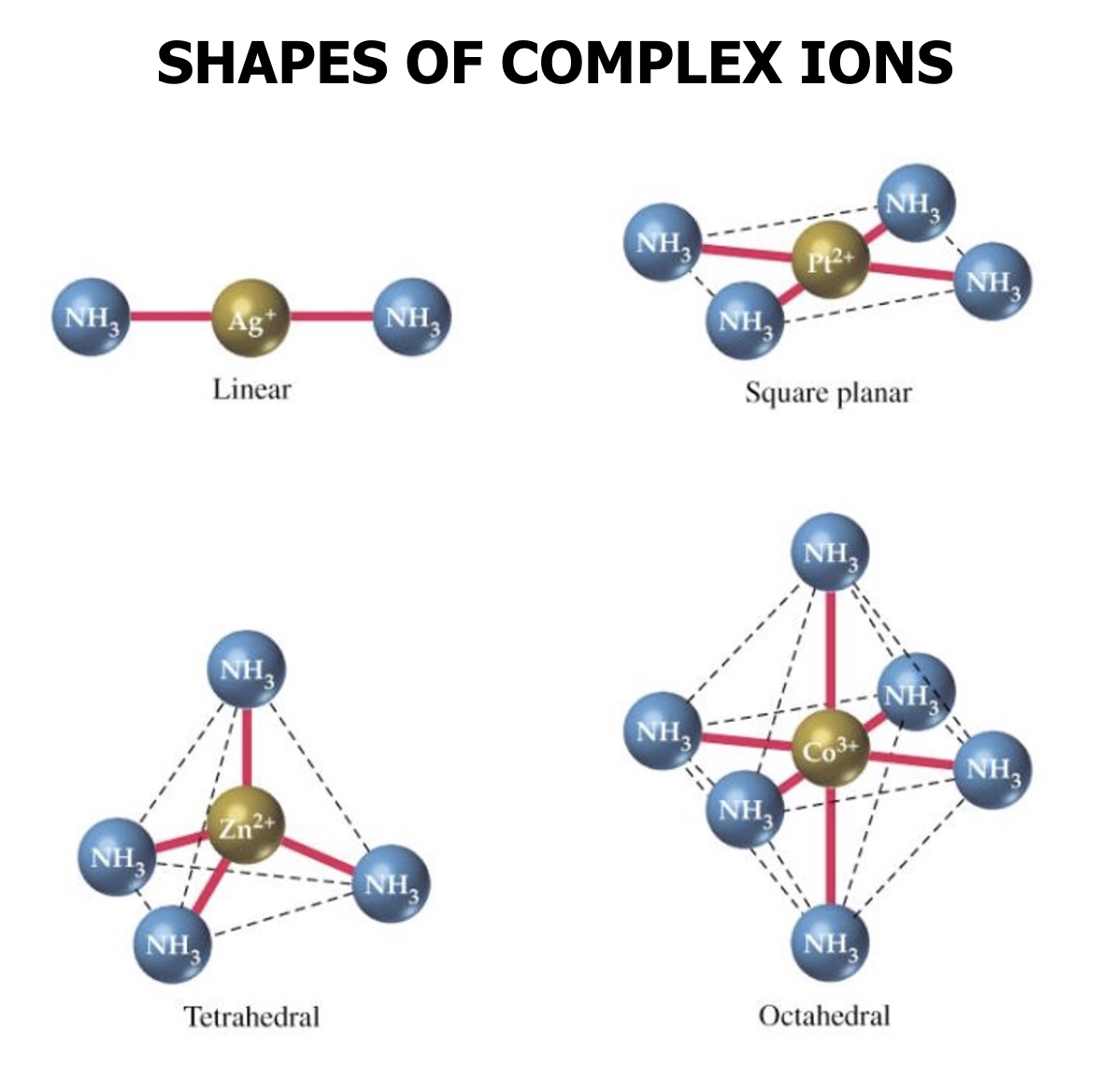
the value of the coordination number results in what?
in a 3d shape
what are the four complex structures transition metals can form?
linear
square planar
tetrahedral
octahedral
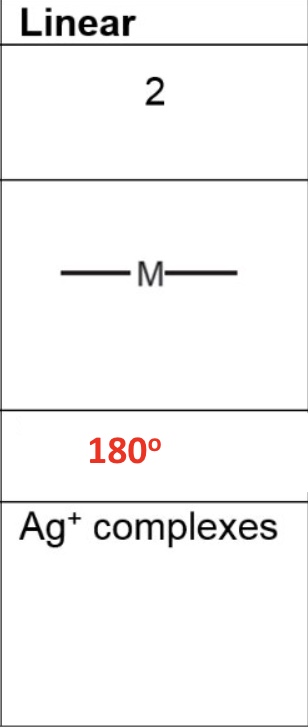
for linear, give the:
a. coordination number
b. shape
c. bond angle
d. occurence in the metals
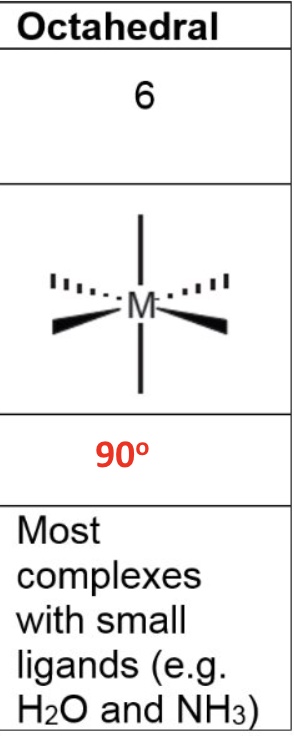
for square planar, give the:
a. coordination number
b. shape
c. bond angle
d. occurence in the metals
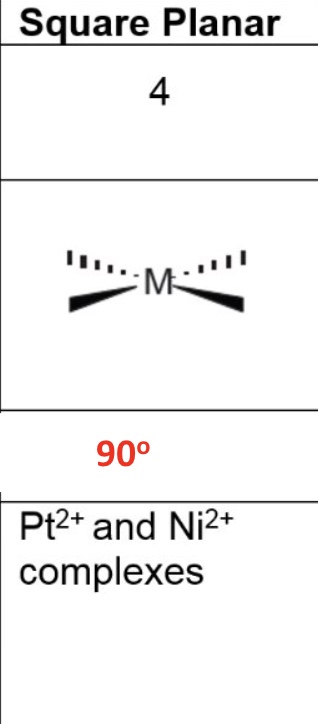
for tetrahedral, give the:
a. coordination number
b. shape
c. bond angle
d. occurence in the metals
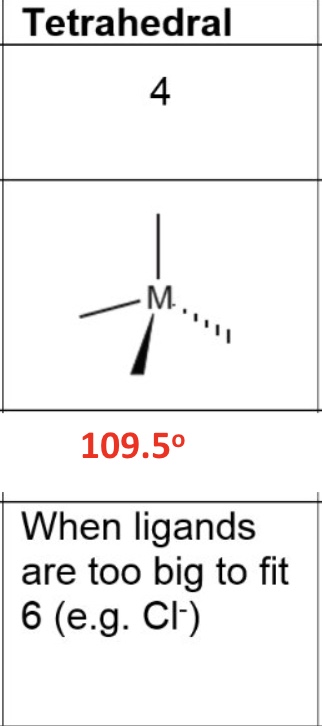
for octahedral, give the:
a. coordination number
b. shape
c. bond angle
d. occurence in the metals
how can ligands be described as? depending on what?
as monodentate, bidentate or multidentate
depending upon the number of coordinate bonds they form to the transition metal
define monodentate ligands
these form one coordinate bond to the transition metal ion
give 6 examples of monodentate ligands

how are H₂O and NH₃ similar?
they are very similar in size
and they are both neutral as well as CO
Cl⁻, CN⁻ and OH⁻ have negative charges, what is the effect of this?
a charge on the ligand will affect the charge on the complex
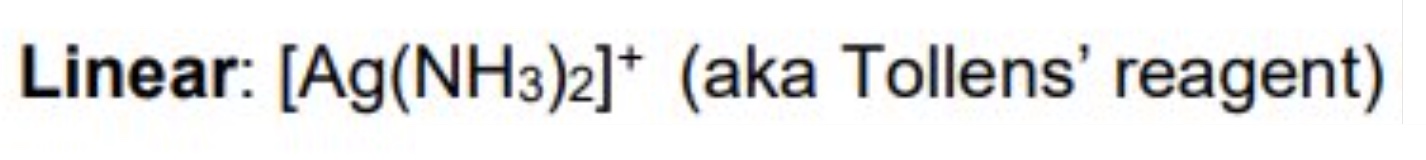
for this complex:
a. draw the shape
b. give the TM oxidation state
c. give the coordination number
b. +1
c. 2
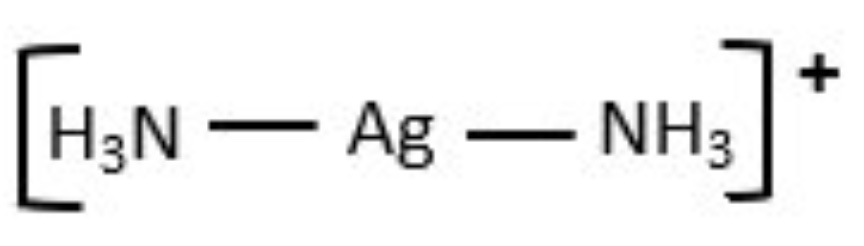

for this complex:
a. draw the shape
b. give the TM oxidation state
c. give the coordination number
b. +2
c. 4
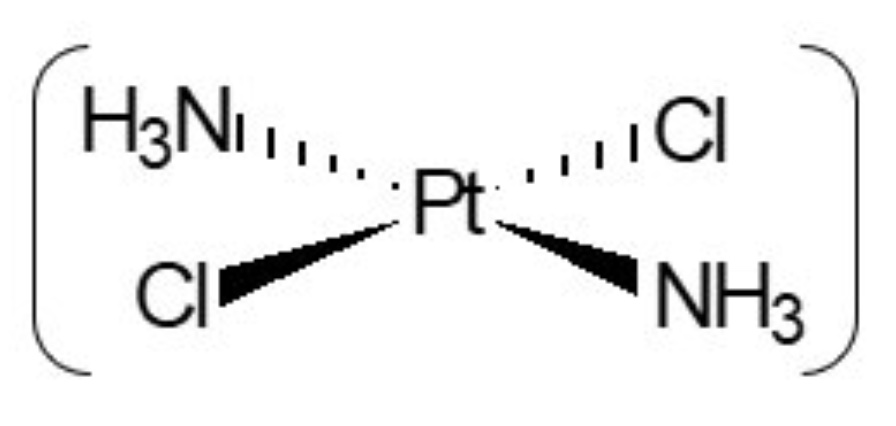
why does the complex ion of chloride ligands have a different shape to those of H₂O ligands?
chloride ligands are bigger than water ligands so only 4 ligands can fit around the TM ion
if all the ligands are neutral then what is the charge of the complex?
the charge will equal the charge of the central transition metal
when will the charge not equal the charge on the transition metal?
if some or all the ligands are charged
what type of isomerism can display in transition metal complexes?
cis-trans isomerism
in what complexes can cis-trans isomerism occur in?
in octahedral + square planar complexes where there are two ligands of a different type to the others
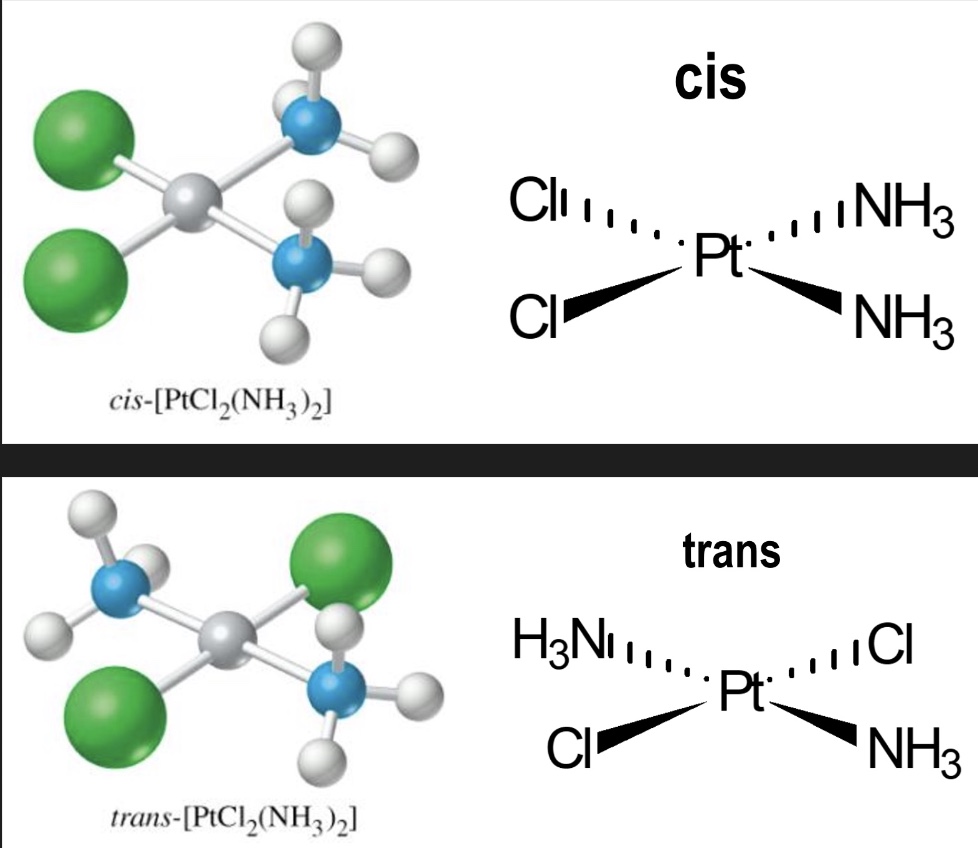
outline cis-trans isomerism
trans implies ‘opposite’ → ligands should be 180° apart (directly opposite)
cis implies ‘on the same side’ → eg next to each other, 90° apart
draw the cis and trans isomer of this complex - square planar
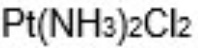
what is this complex?
its called cis-platin
it is an anti-cancer drug
give the advantages + disadvantages of cis-platin
advantages: it kills cancer cells
disadvantages: hair loss + fertility issues
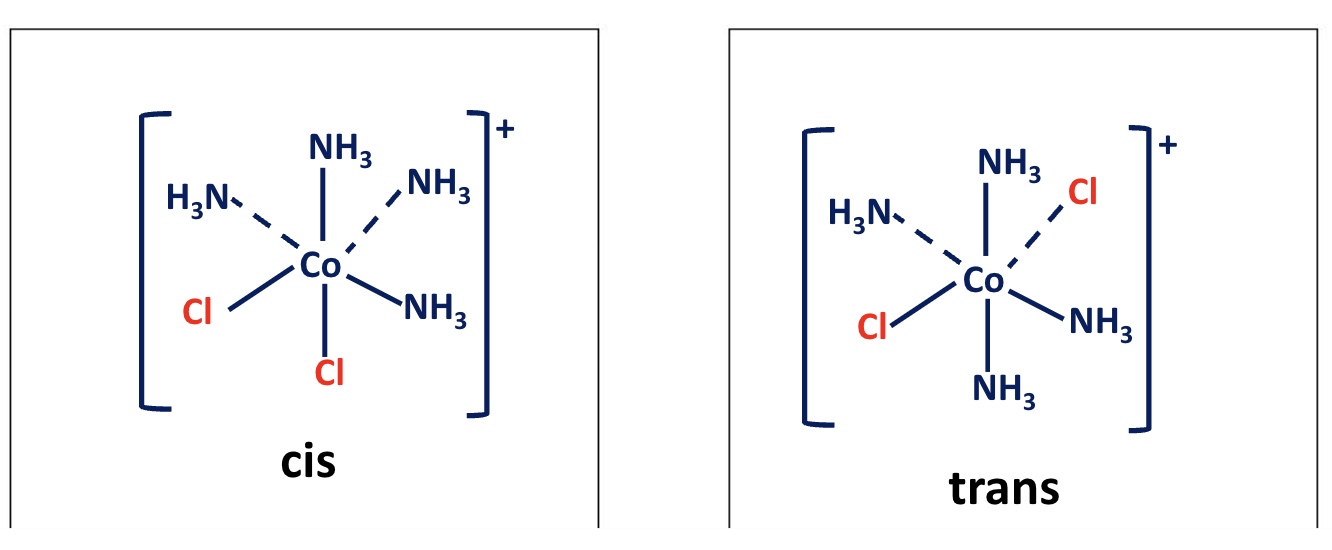
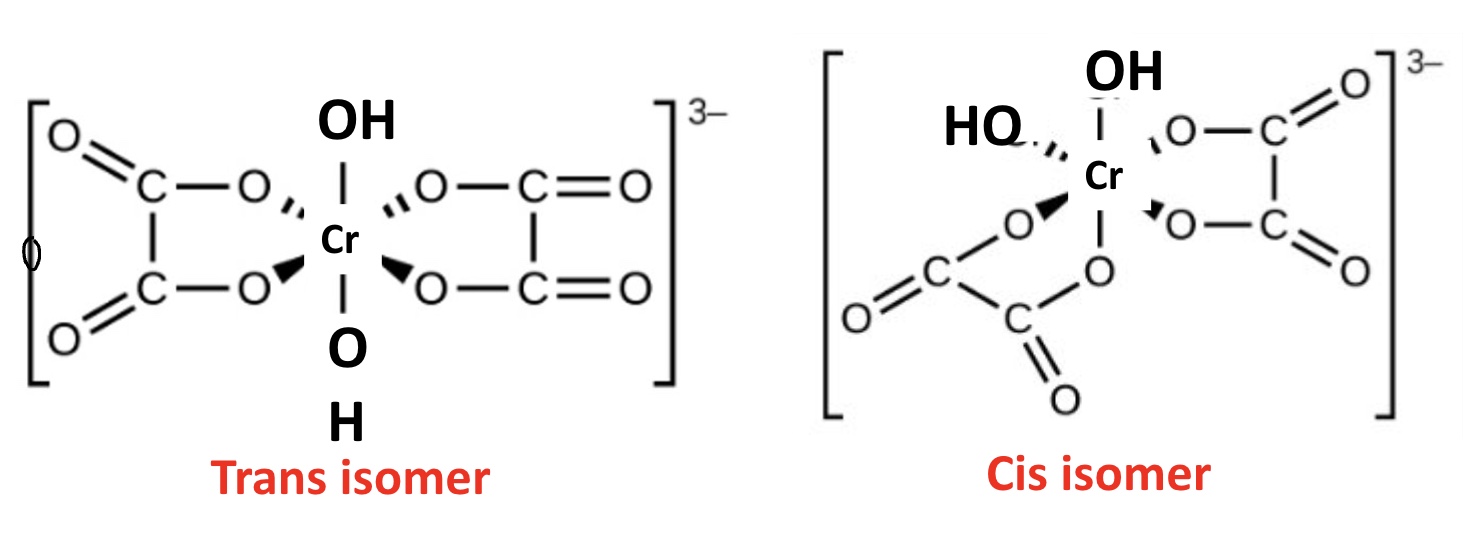
outline cis-trans isomerism in octahedral complexes
if two ligands in an octahedral complex are different from the other four, giving a MA₄B₂ complex, two isomers are possible
the two B ligands can be cis (90° to each other) or trans (180° to each other)
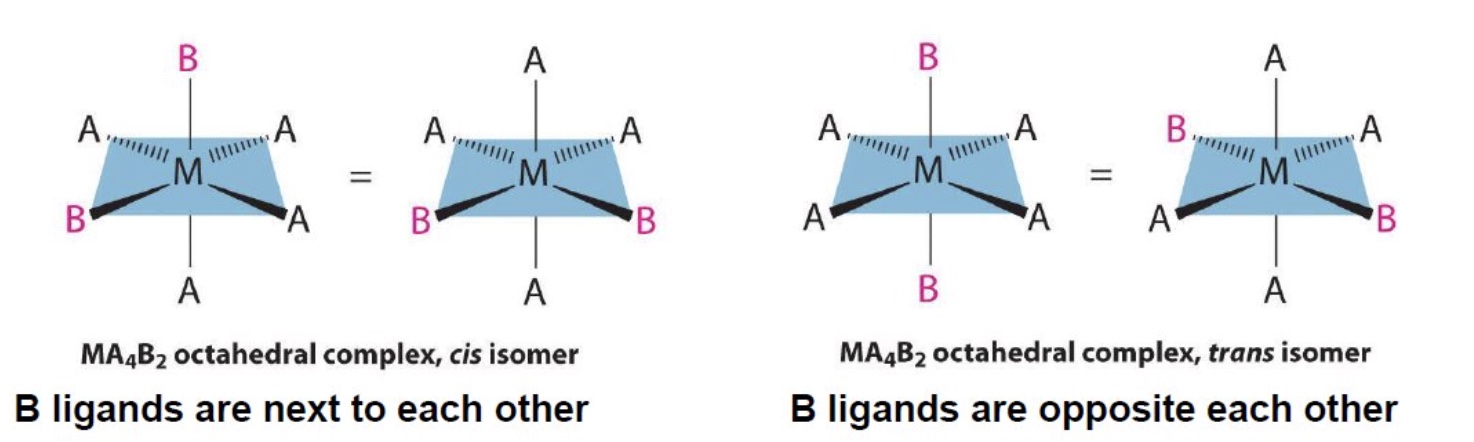
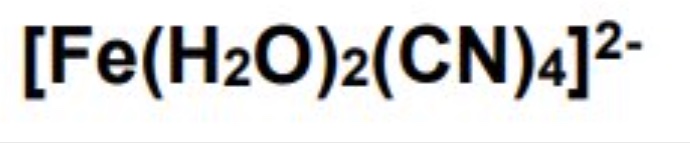
draw the cis and trans isomers for this complex
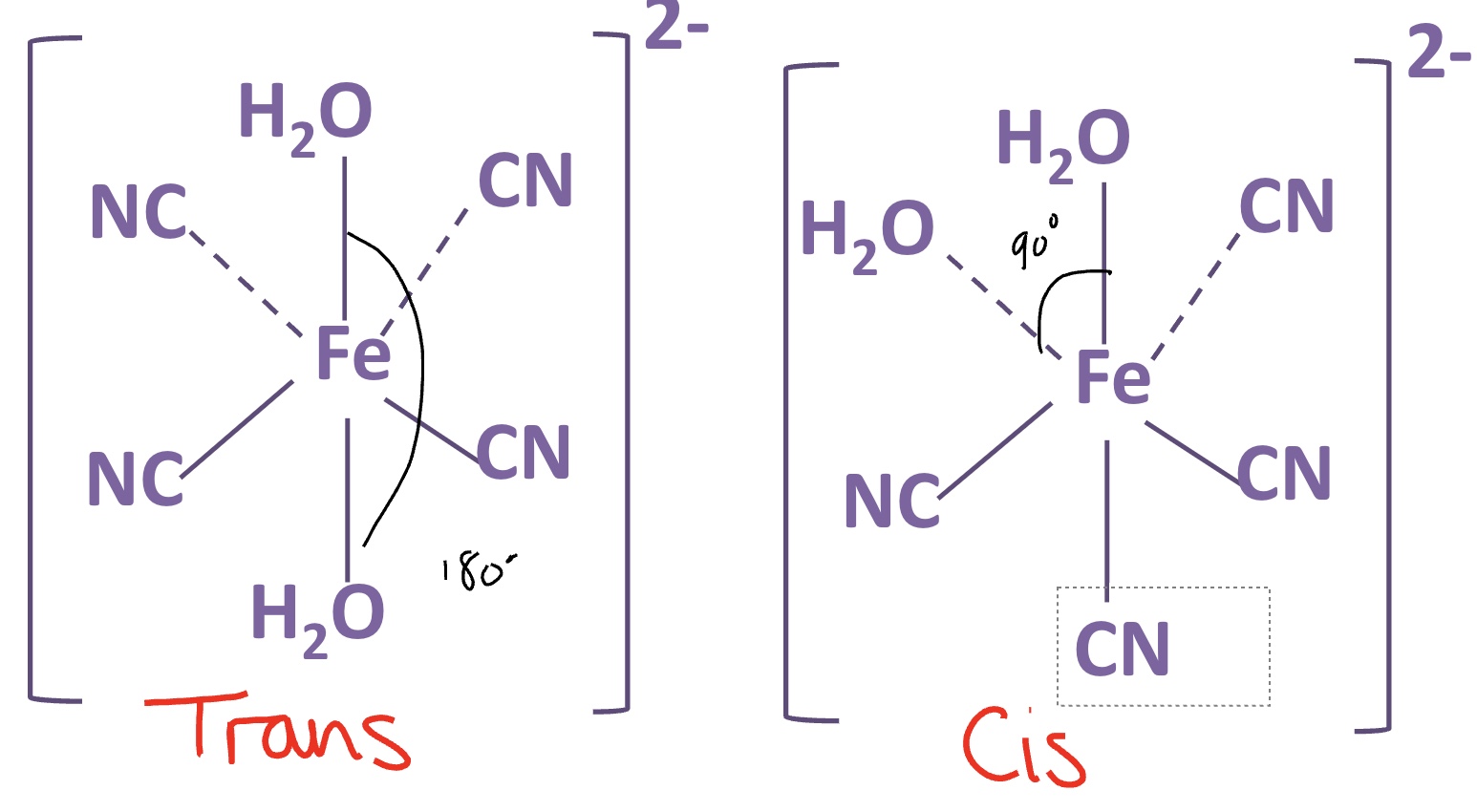
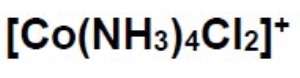
draw the cis and trans isomers for this complex
what is a bidentate ligand?
one which forms two coordinate bonds to a transition metal ion through two different atoms on the same ligand
where must the two lone pairs come from for bidentate ligands?
they must come from separate atoms (ie one oxygen cannot bond twice but 2 oxygens may bond once each)
give 2 examples of bidentate ligands
1,2-diaminoethane or ethyl diamine
ethanedioate ions
give the formula + structure of 1,2-diaminoethane
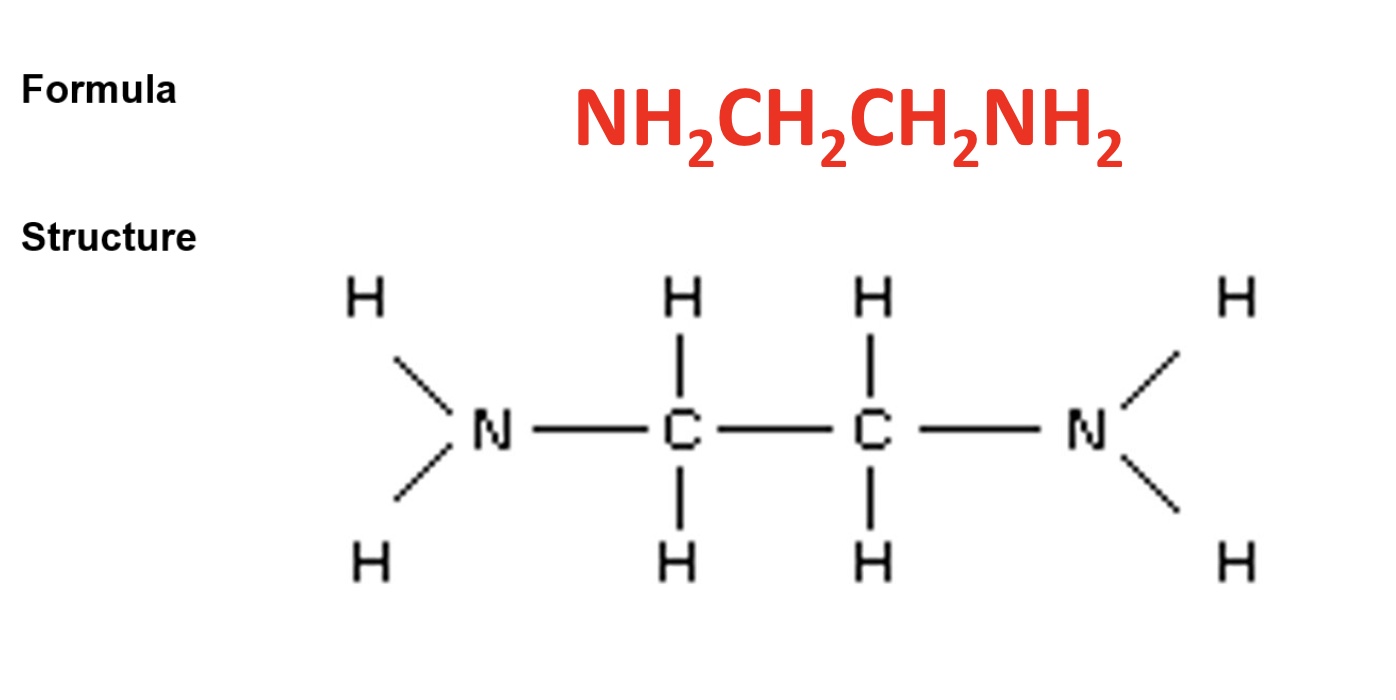
outline its charge + how 1,2-diaminoethane bonds as a ligand
the ligand is neutral
it contains 2 nitrogen atoms each with a lone pair that it donates to the transition metal when forming the coordinate bond
this means 3 bidentate ligands can fit around a TM atom or ion
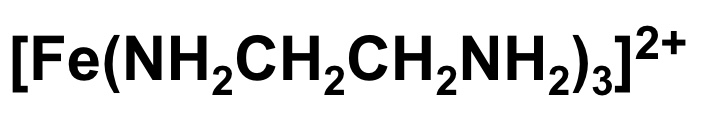
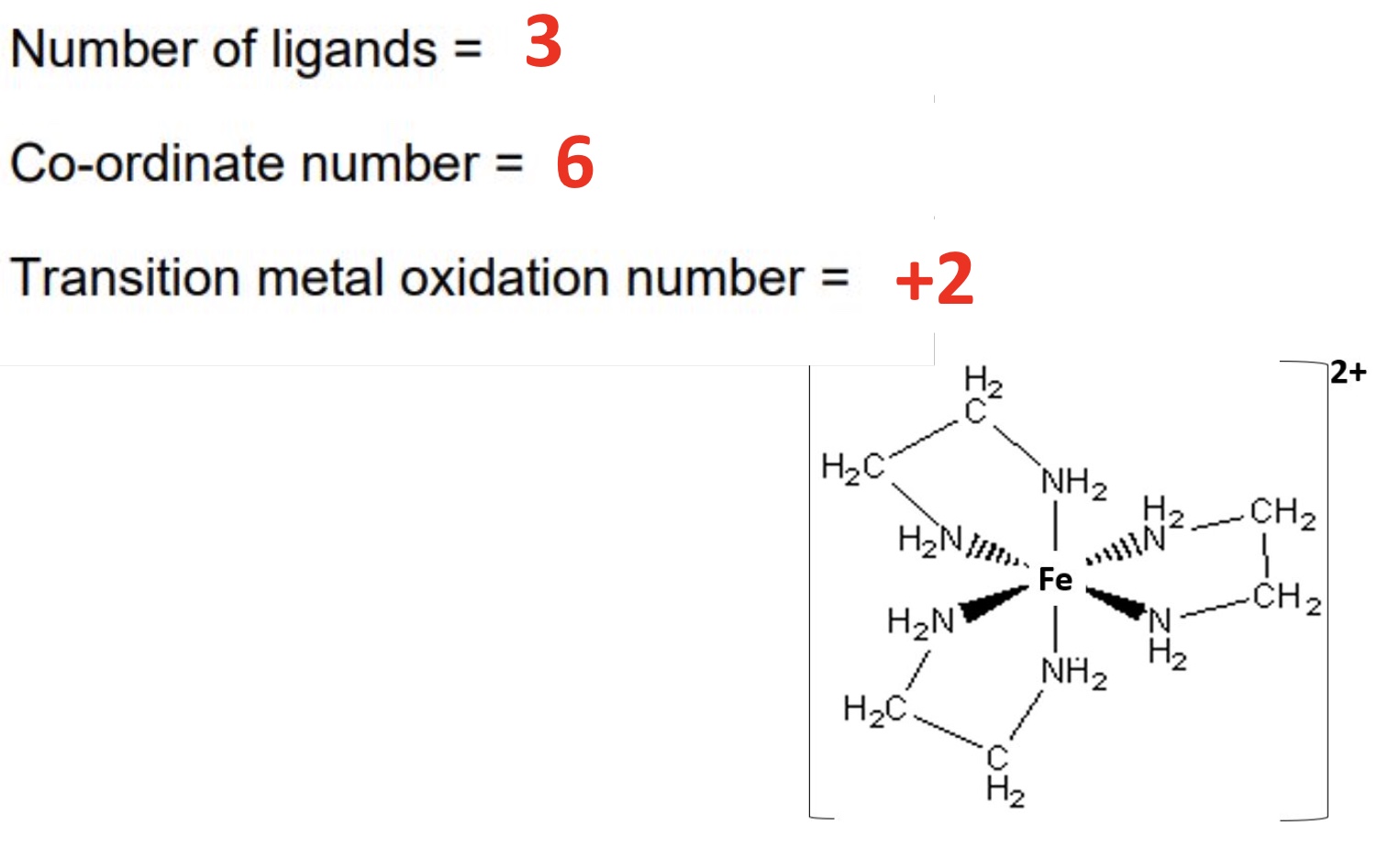
for this complex:
a. draw the shape
b. give the number of ligands
c. give the coordination number
d. give the TM oxidation state
give the formula + structures of ethanedioate ions (solid + in solution)
formula: C₂O₄²⁻
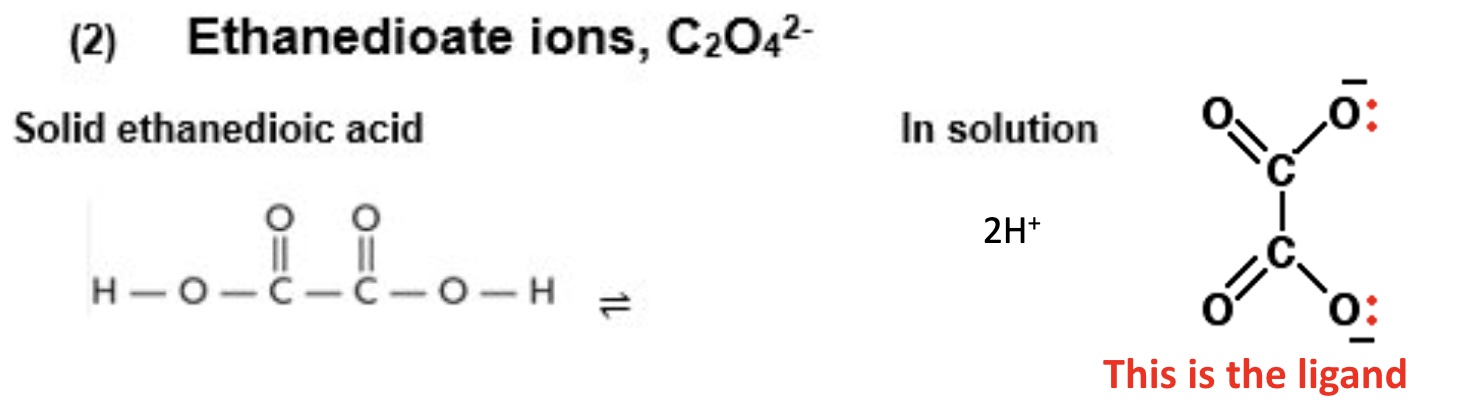
outline its charge + how ethanedioate ions bond as a ligand
the ligand has a 2- charge
it contains 2 oxygen atoms each with a lone pair that it donates to the transition metal when forming the coordinate bond
this means 3 ethanedioate ions can fit around a TM ion
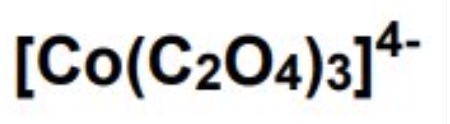
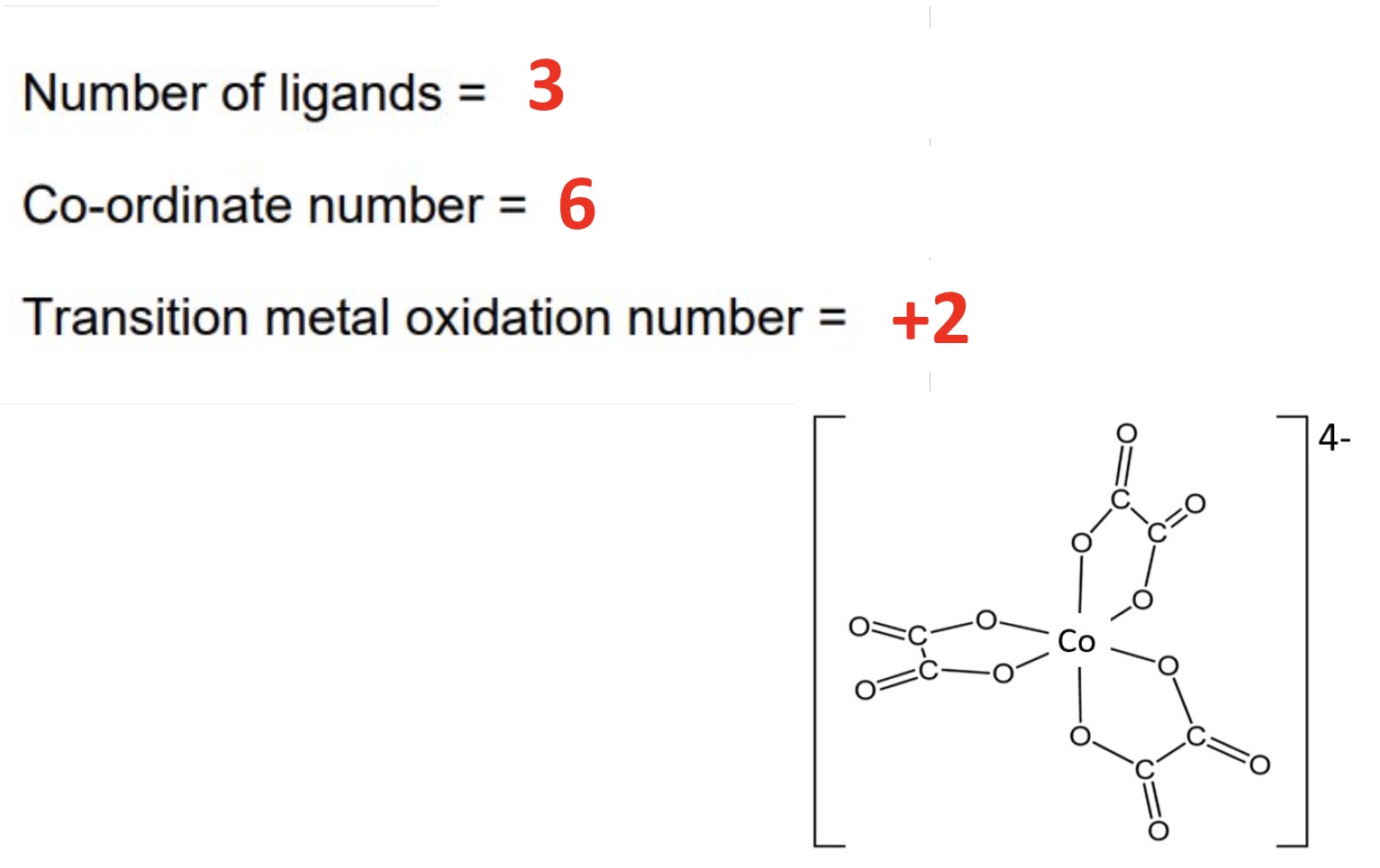
for this complex:
a. draw the shape
b. give the number of ligands
c. give the coordination number
d. give the TM oxidation state
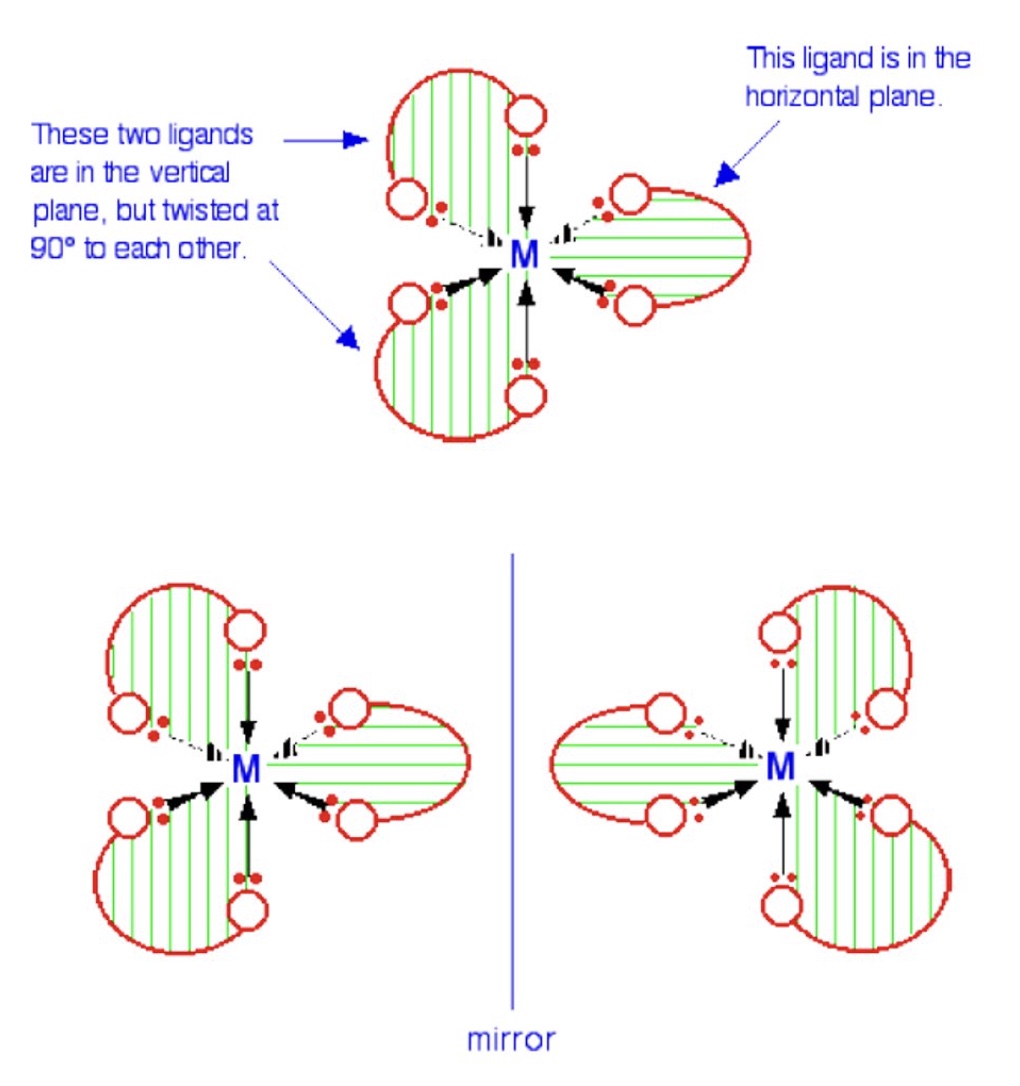
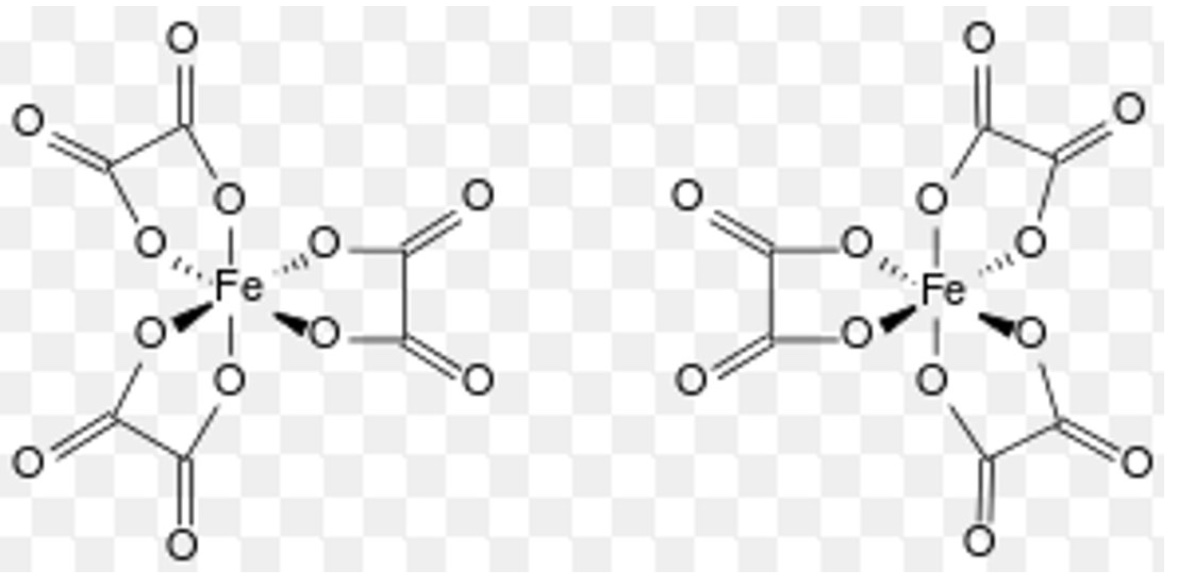
when does optical isomerism of complexes occur?
in octahedral complexes containing bidentate ligands
give 2 features of optical isomers
they have no plane of symmetry
they are non-superimposable mirror images of one another
how do optical isomers differ?
they differ in the direction with which they rotate plane-polarised light
what do octahedral complexes formed from bidentate ligands then form?
they form 2 isomers that are mirror images of one another + exhibit all the characteristics associated with optical isomers


what are multidentate ligands?
these are ligands that can form 2 or more coordinate bonds to a transition metal ion
what is the maximum number of bonds from one ligand?
6
what does the ligand need to have to make these 6 bonds?
6 lone pairs on different atoms within the one ligand
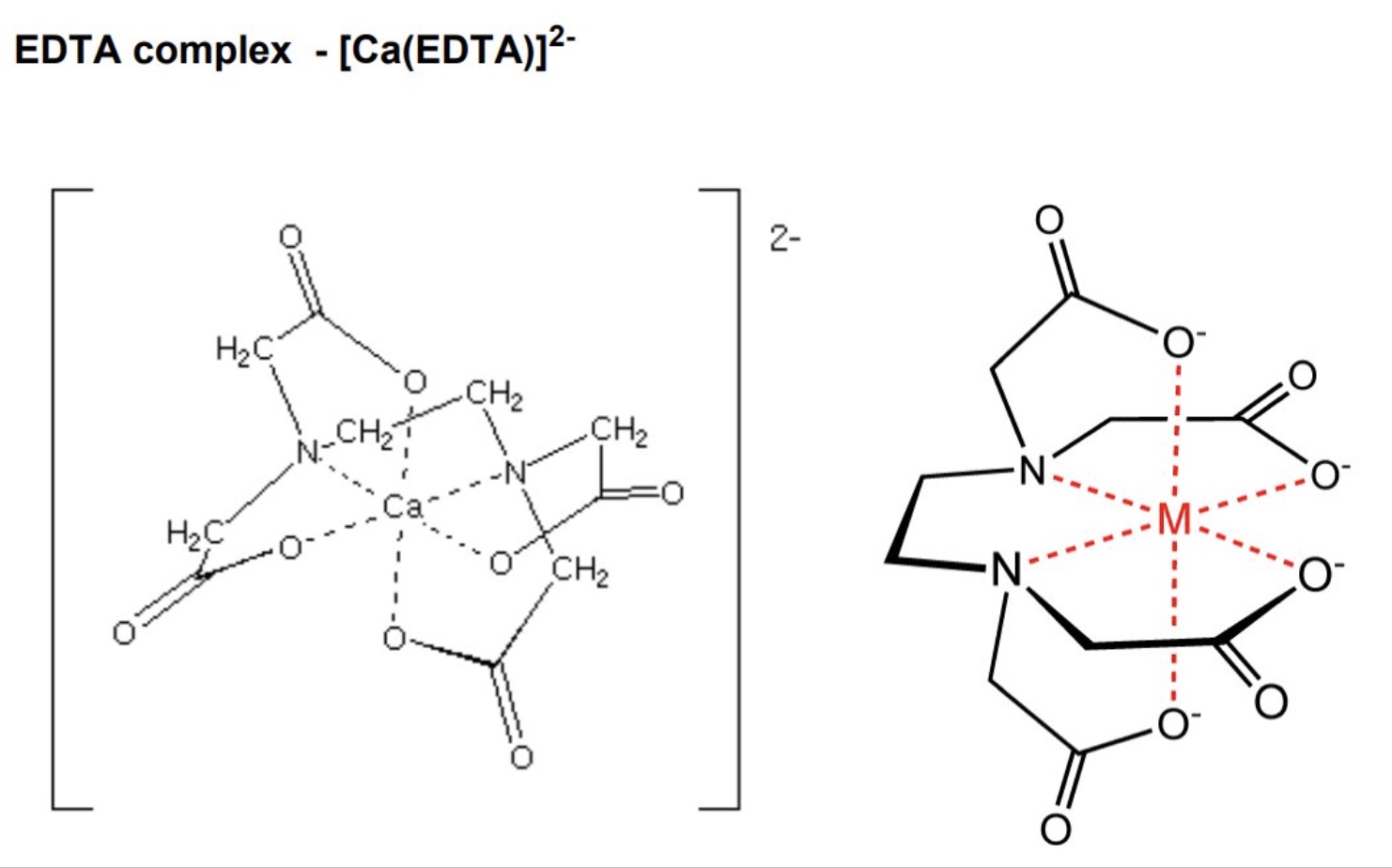
give an example of a multidentate ligand
EDTA⁴⁻
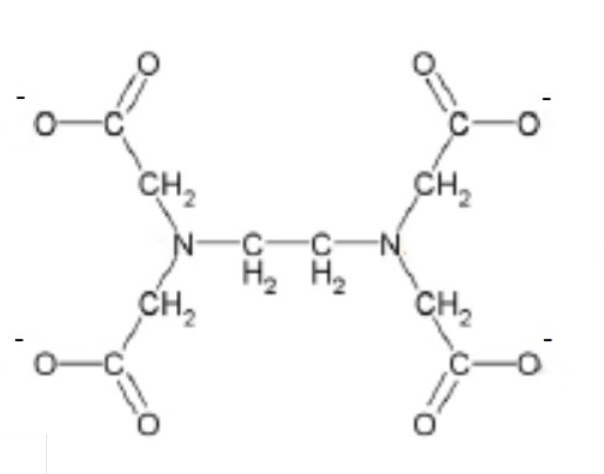
draw the lone pairs on the EDTA⁴⁻ ligand where they bond with the transition metal
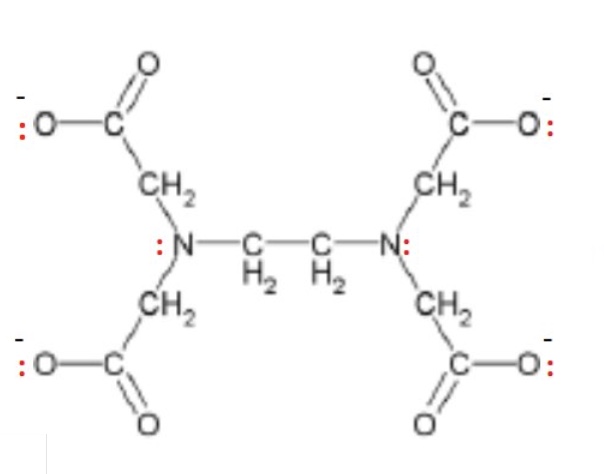
give the general formula for a complex ion containing EDTA⁴⁻ as well as the number of ligands + coordination number
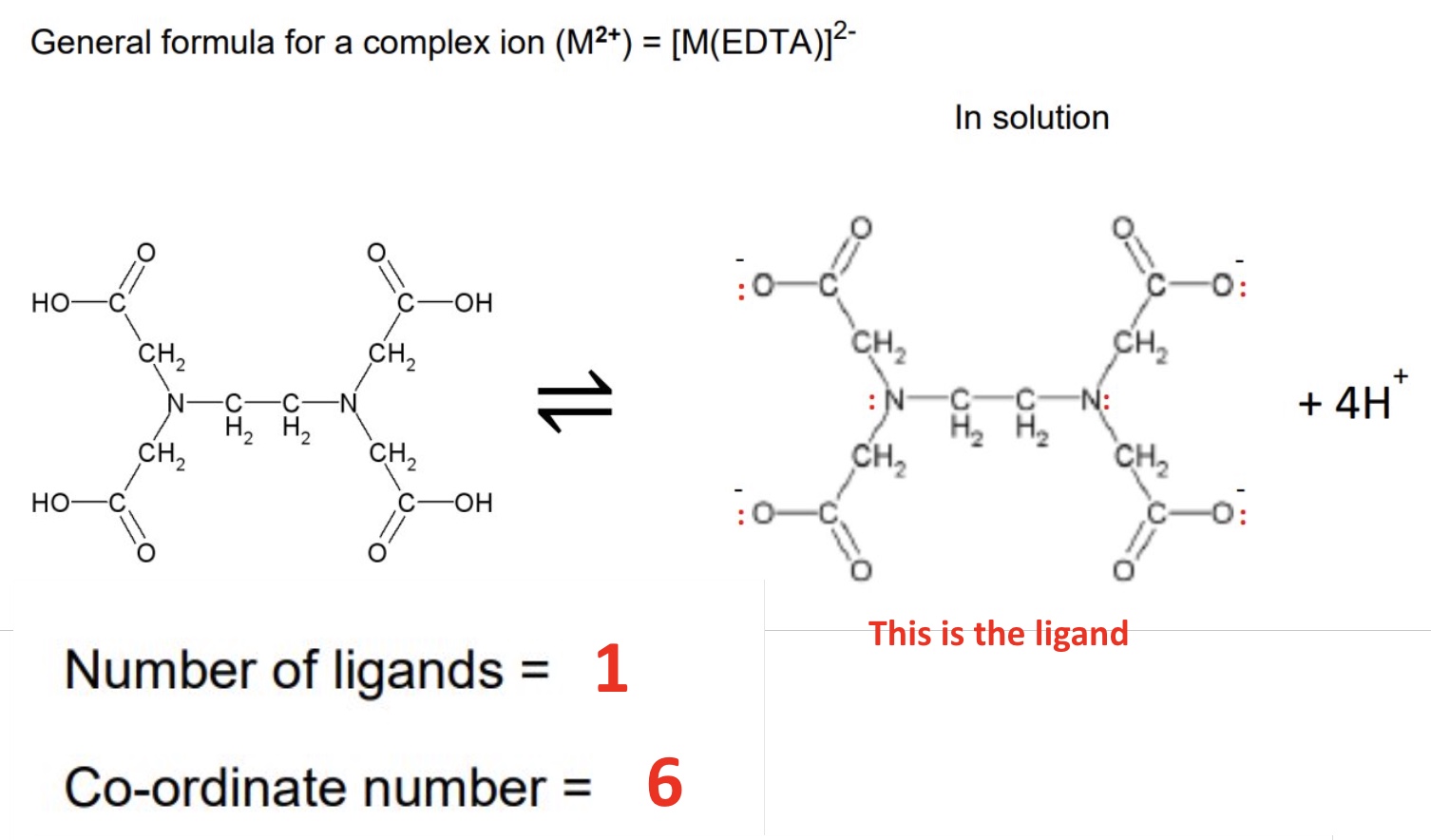
for EDTA to be most effective, it is used in alkaline conditions, outline why
the OH⁻ reacts with H⁺
equilibrium shifts to the right, therefore ensuing the EDTA can form 6 coordinate bonds
an EDTA complex forms a ______ shape with ____ isomerism
octahedral shape
no isomerism
give 2 uses of EDTA
medicinal use
complexometric titration
outline how EDTA has a medicinal use
EDTA is used to treat patients suffering from lead poisoning
this treatment is known as chelation therapy, in which EDTA makes the toxic ions present in the body harmless
describe how EDTA is used in a complexometric titration



what is haemoglobin?
an iron complex
used to transport oxygen around the body
*its structure is given on the data sheet
outline the central metal ion + ligand that haemoglobin contains
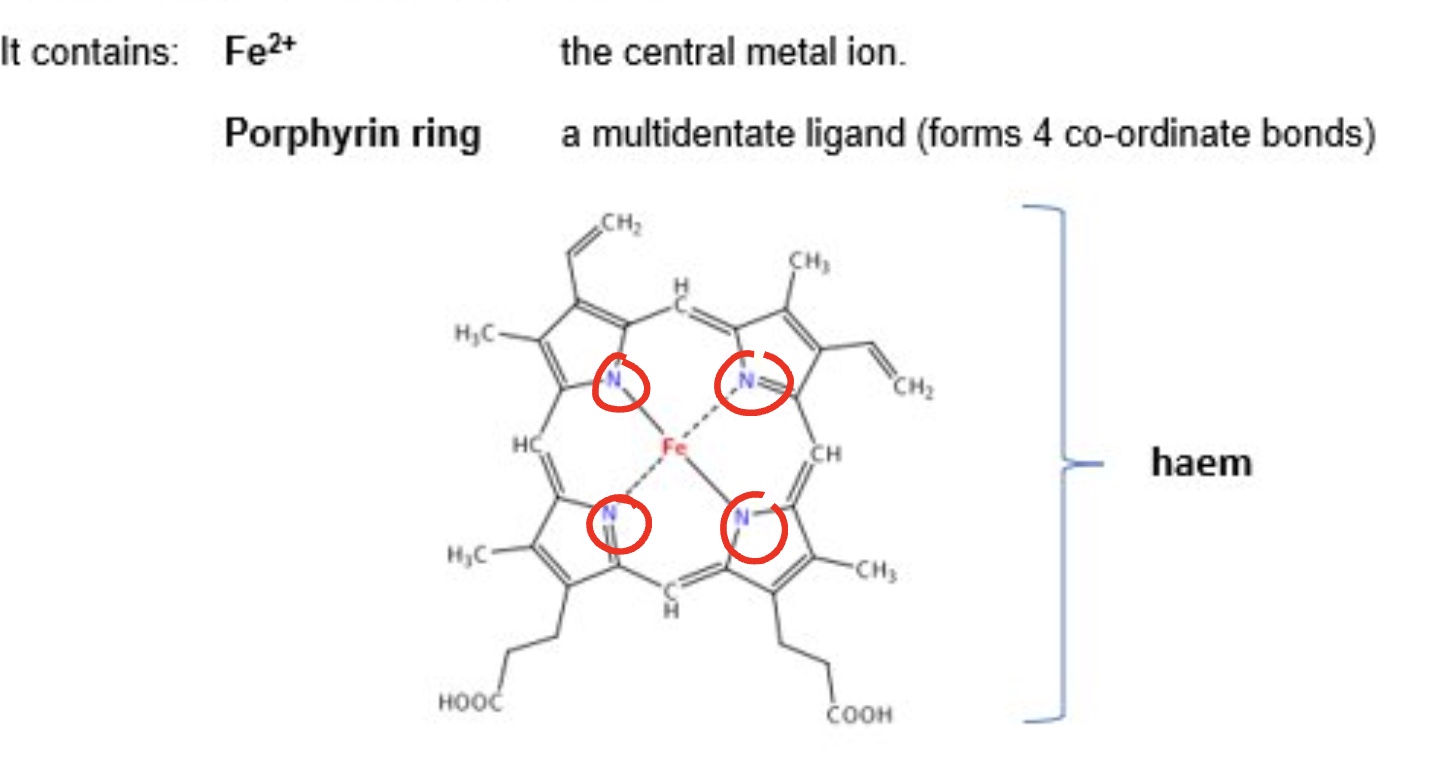
what is globin?
a protein acting as a monodentate ligand with a lone pair from an N atom
outline how haemoglobin transports oxygen
the porphyrin ring forms 4 coordinate bonds
this leaves one space to ‘carry’ a ligand
when oxygenated, O₂ is acting as a monodentate ligand + forms a coordinate bond to Fe → O₂ is easy to pick up in oxygen-rich lungs
explain why carbon monoxide is toxic
when it is inhaled it forms a coordinate bond with the Fe²⁺ ion in haemoglobin (taking up the spare space) instead of O₂
this prevents the transport of oxygen around the body
what can happen to the ligands on a complex?
they can be substituted for other ligands
example 1:
what can H₂O ligands be replaced with + why?
with NH₃ ligands
these are similar in size + both are neutral
sometimes all the ligands are replaced, sometimes just some
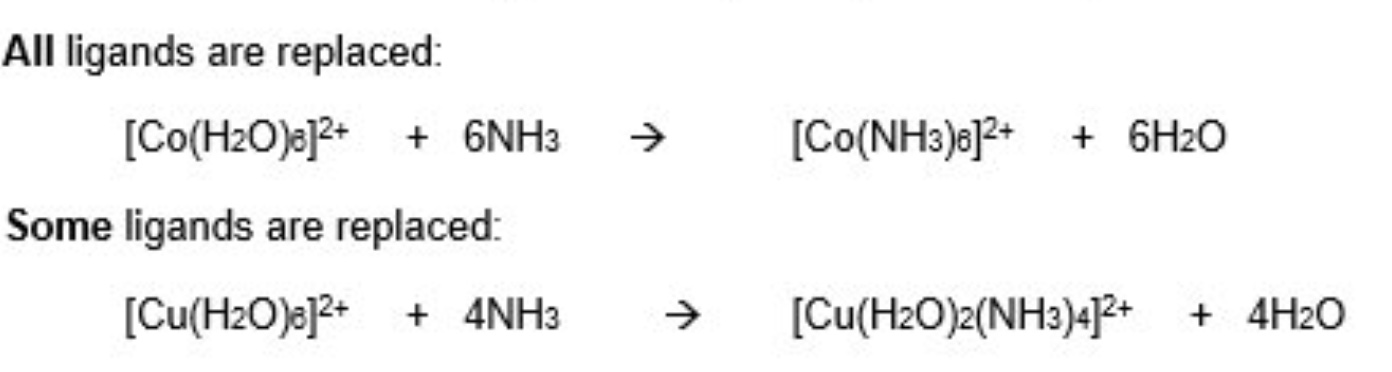
example 2:
outline another example how H₂O ligands can be substituted
they can be replaced with Cl- ligands
Cl- ligands are bigger + only 4 fit around the central metal ion so the complex formed will be tetrahedral
6 ligands are swapped for 4 ligands
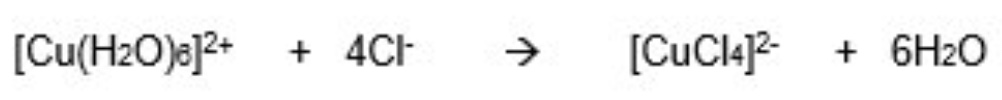
where do Cl- ligands come from?
HCl
example 3:
what can bidentate ligands substitute for?
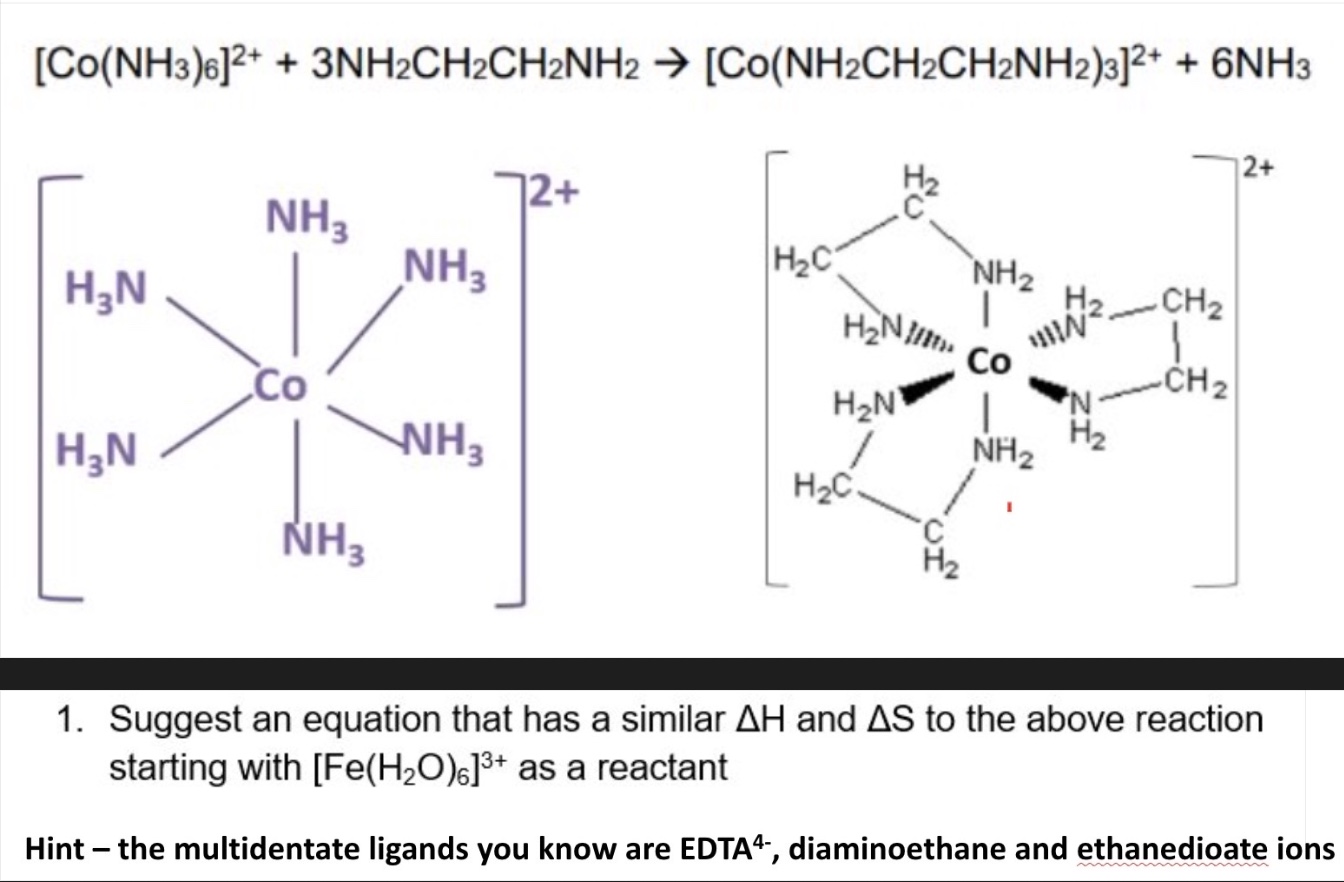
for monodentate ligands → remember only 3 bidentate ligands can fit around a TM ion
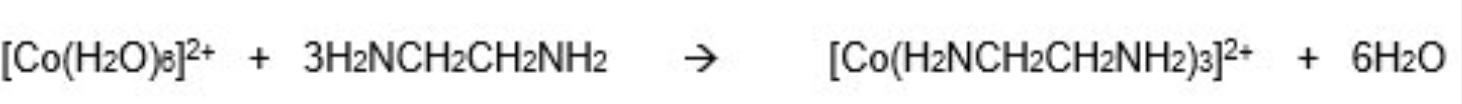

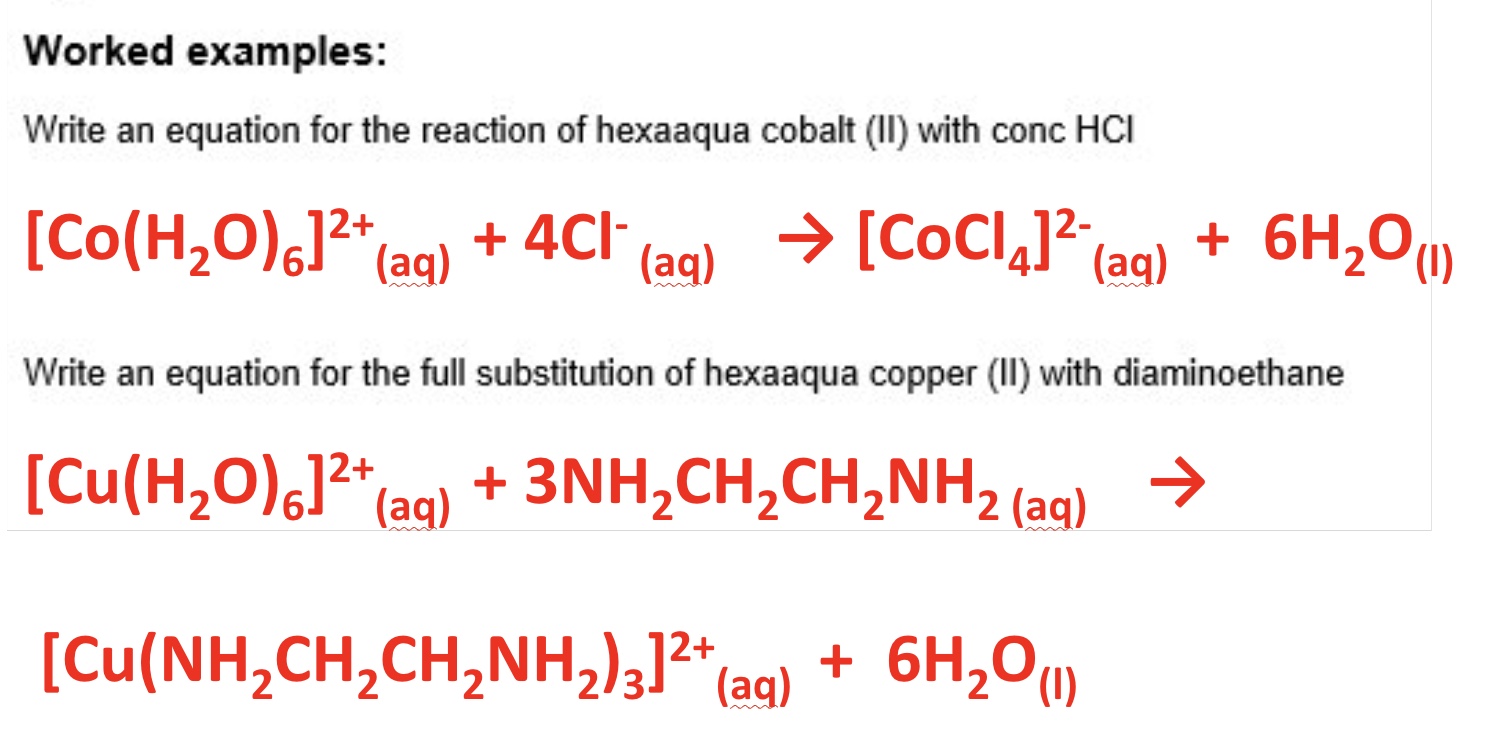
what is chelation?
the name given to the process whereby bidentate or multidentate ligands substitute monodentate ligands to create a more stable complex
what can the formation of the more stable multidentate ligand complex happen through?
through a ligand substitution reaction
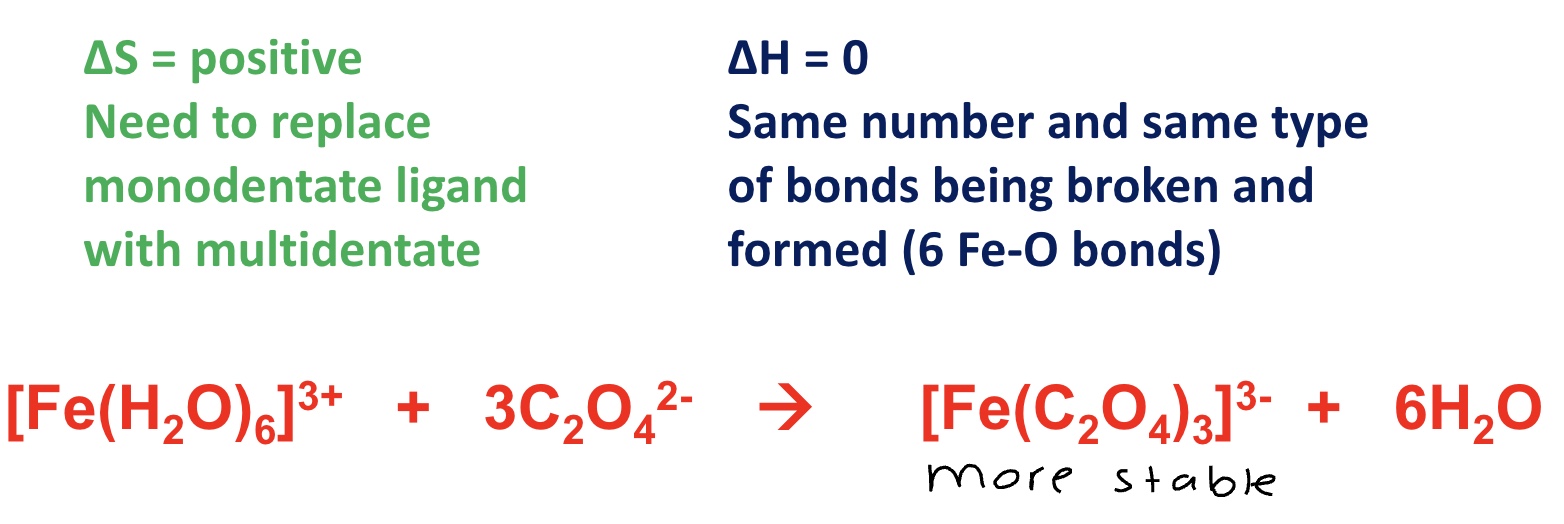
why is the product more stable?
as there has been a positive △S (more moles of product than reactant)
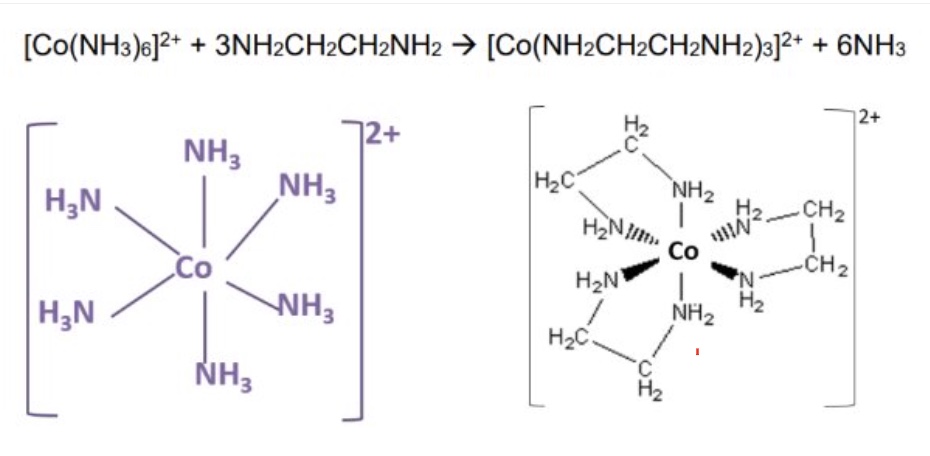
what are often used as chelating agents? to do what?
multidentate ligands
to remove harmful ions + render them useless
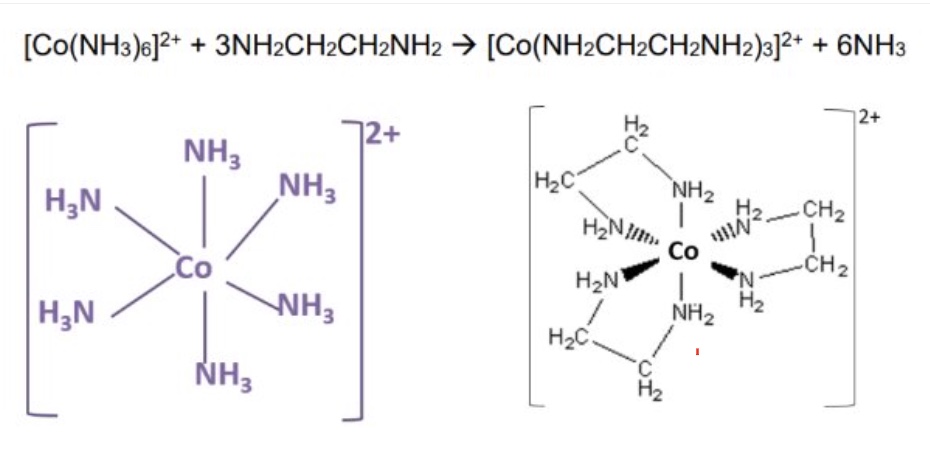
by considering entropy + enthalpy changes, explain why the free energy (△G) for this reaction is negative