AP Chemistry Chapter 11 - Kinetics
1/50
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Zero order reaction
A chemical reaction in which the rate remains constant as the concentration of the reactants rises or falls.
First order reaction
A chemical reaction in which the reaction rate is linearly dependent on the concentration of only one reactant.
Second order reaction
One whose rate is proportional to the square of the concentration of one reactant.
Half-life
The amount of time it takes for an initial concentration to drop to half of its original value.
Activation energy
The minimum amount of extra energy required by a reacting molecule to get converted into a product.
Reaction mechanism
The sequence of elementary steps by which a reaction occurs.
Unimolecular
Only one reactant molecule is necessary for the reaction to occur.
Bimolecular
Two reactant molecules must collide for the reaction to occur.
Termolecular
Three reactant molecules must collide for the reaction to occur.
Reactive intermediate
A substance that is formed in one step of a mechanism and then used up in a later step.
Rate-determining step
The slowest step of a reaction that determines the speed at which the overall reaction occurs. This step CANNOT have an intermediate as a reactant.
Enzyme
A biological catalyst.
what is the equation for average rates of reaction in kinetics
rate= -1/a Δ[A]/ ΔT….. reactants at -, products are +
5 factors affecting reactant rates
chemical nature of reactants
surface area of reactants
temperature
concentration of reactants
presence of a catalyst
k and m need to be deduced from _____________ data
experimental
k is dependent on
nature of reaction
Temperature
K is independent of
concentration
zero order is ____________ of concentration
independent
in first order, rates are ___________ to concentration
porportional
in second order, rates are ___________ concentration
porportional to the square of the concentration
to find overall order
sum of m and n
if half lives decrease with time _____ order
zero-order
if half lives are constant ______ order
first order
if half lives get longer
second order
Higher activation energy means faster or slower reaction?
slower
which step controls the reaction in an elementary reaction
the slowest
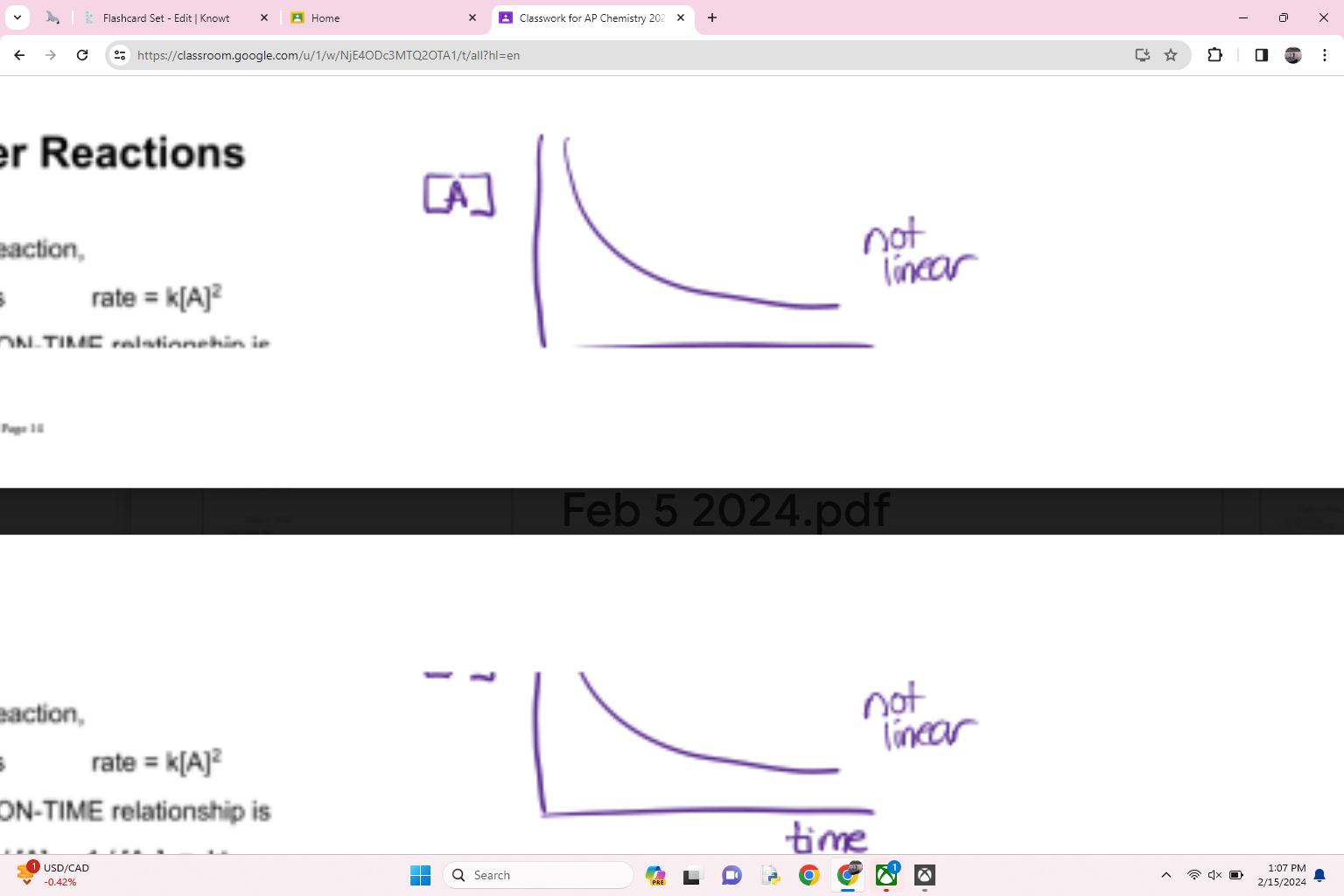
what order is this
zero order
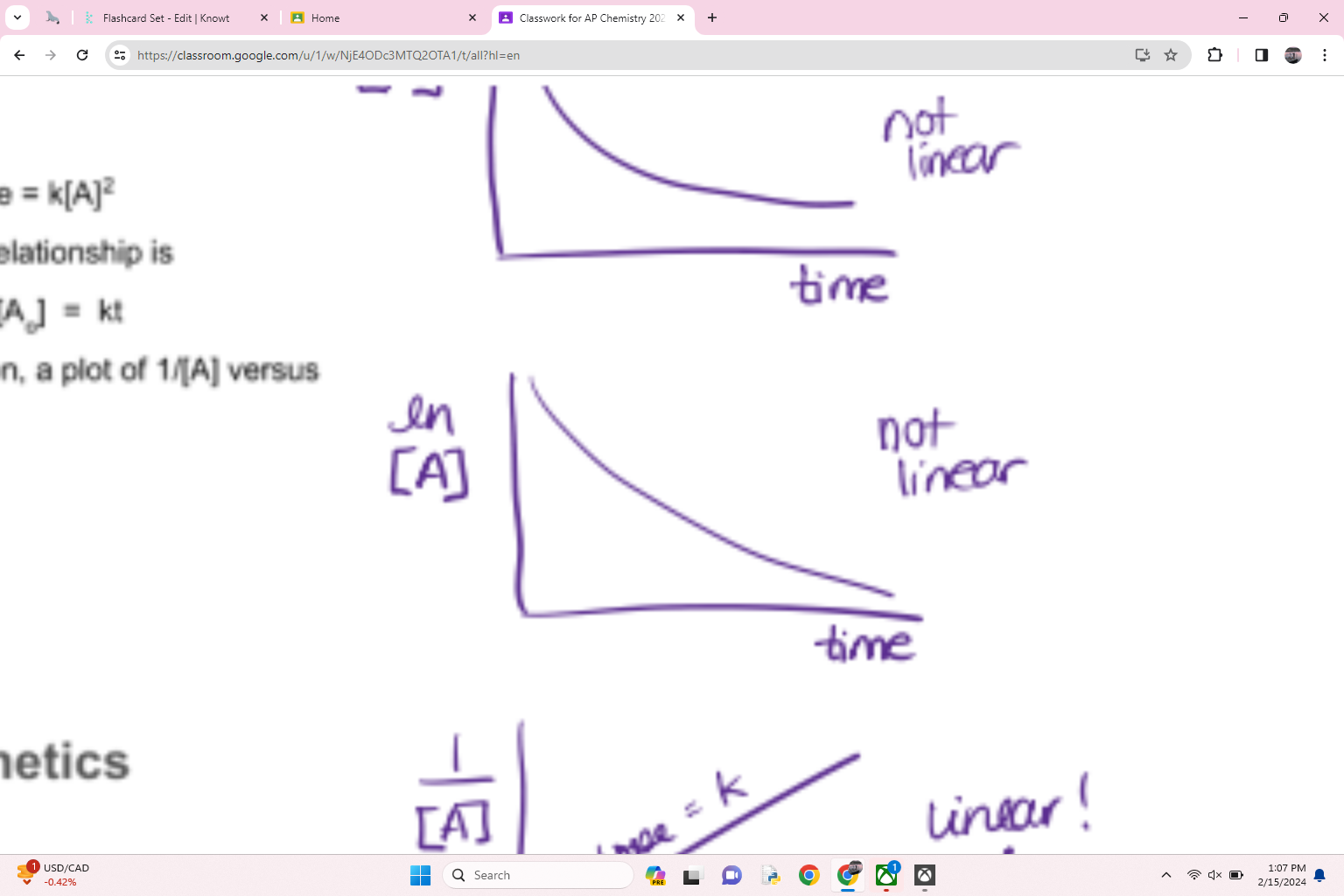
what order is this
first order
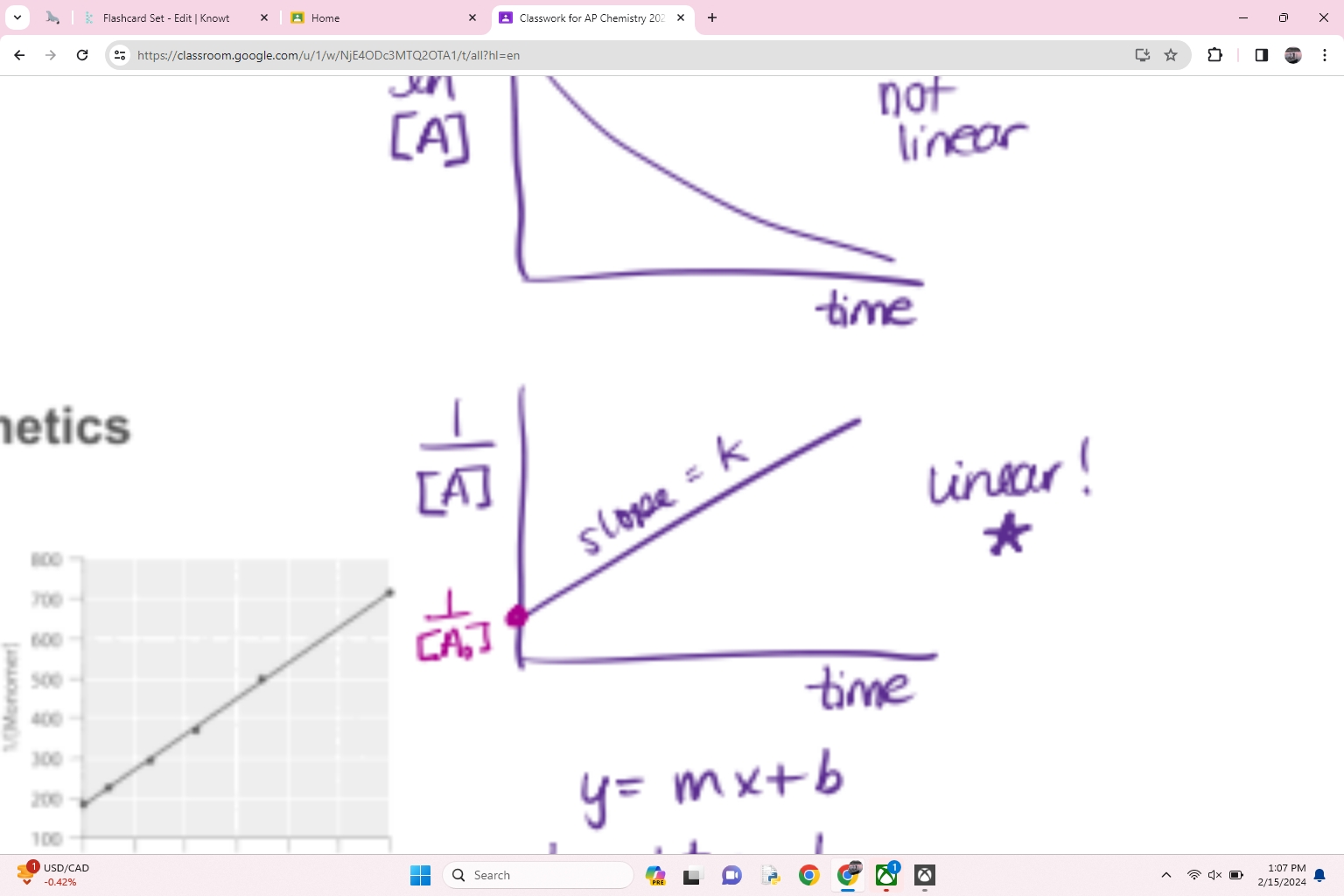
what order is this
second
variable that changes half life
starting molarity
what is p
steric factor
is p temperature independent or dependent
independent
what is z
collision frequency
is collision frequency temperature independent or dependent
relatively temperature independent
why does temp affect reaction rates
as temp increases, more collision have sufficient energy to achieve Ea
every 10 degrees what happens to the reaction rate
doubles!
does the slowest step in a mechanism the lowest or highest activation energy
highest
what does a catalyst do
offers a second path of lower energy
what kind of catalysis is in different phases
heterogenious
what kind of catalysis is dispersed in a single phase, ususally liquid
homogeneous
what kind of catalysis are acid and base catalysis
homogeneous
what happens to the rate of a 0 order reaction over time
it stays constant
what happens to the rate of a 1st order reaction over time
it decreases
what happens to the rate of a 2nd order reaction over time
it decrease
what are the units of K for a second order reaction
mol²/L²-time
what is the unit for k in a first order reaction
time^-1
in a second order reaction what happens if more reactant is added
k and rate increase
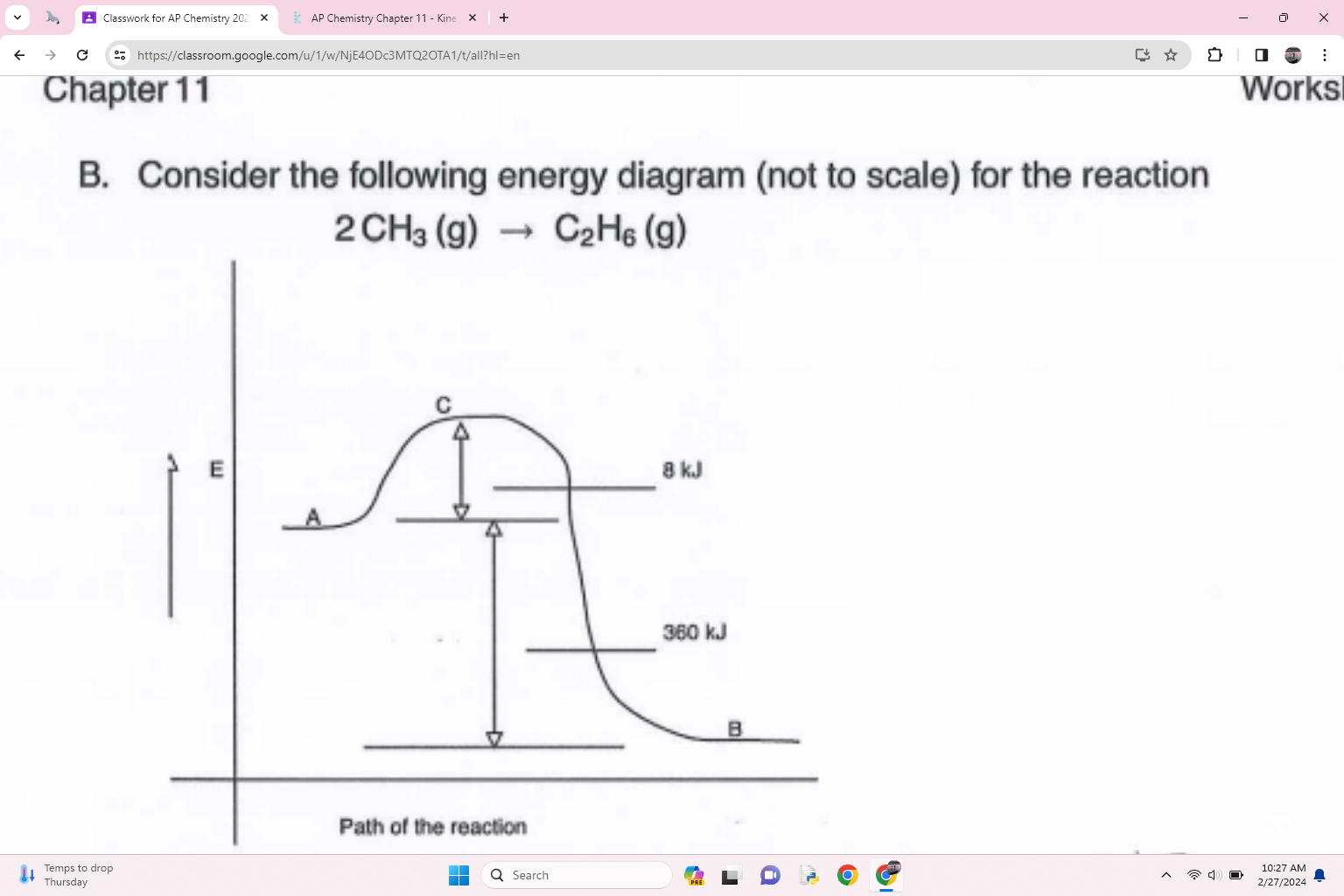
what is the activation energy of the forward reaction
8 Kj
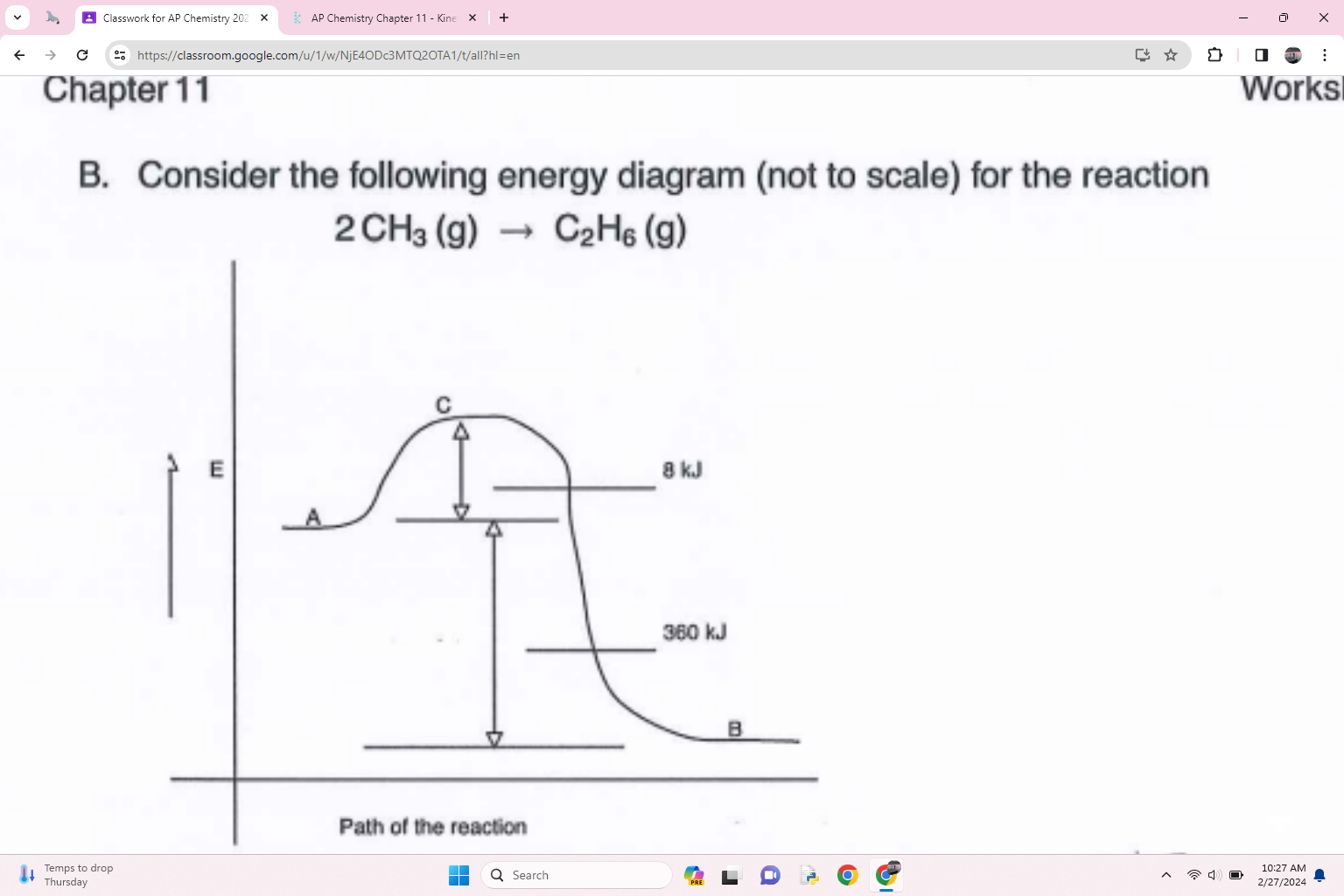
what is the delta h for the reaction
360 kJ
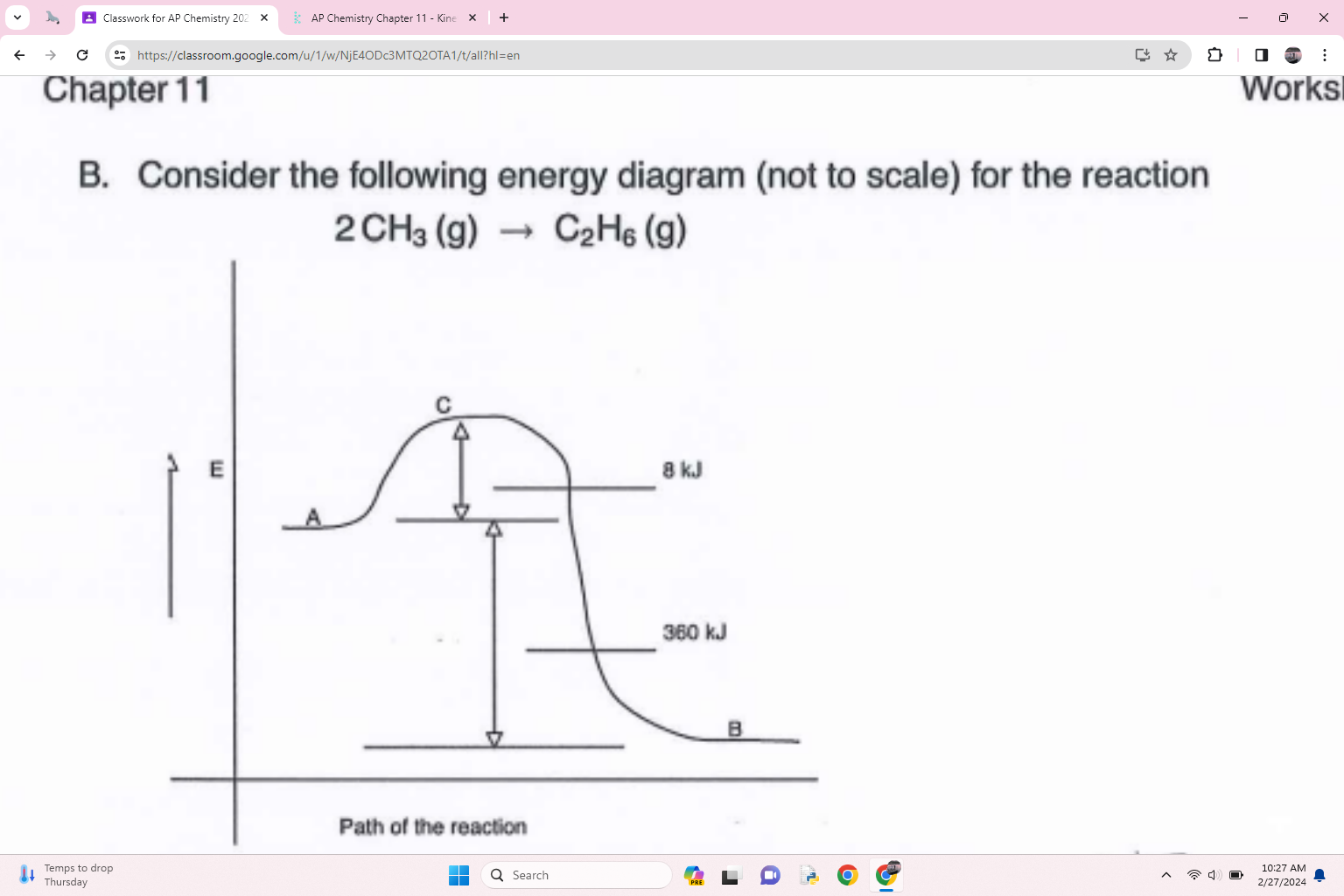
What is the activation energy for the reverse reaction
368 kJ
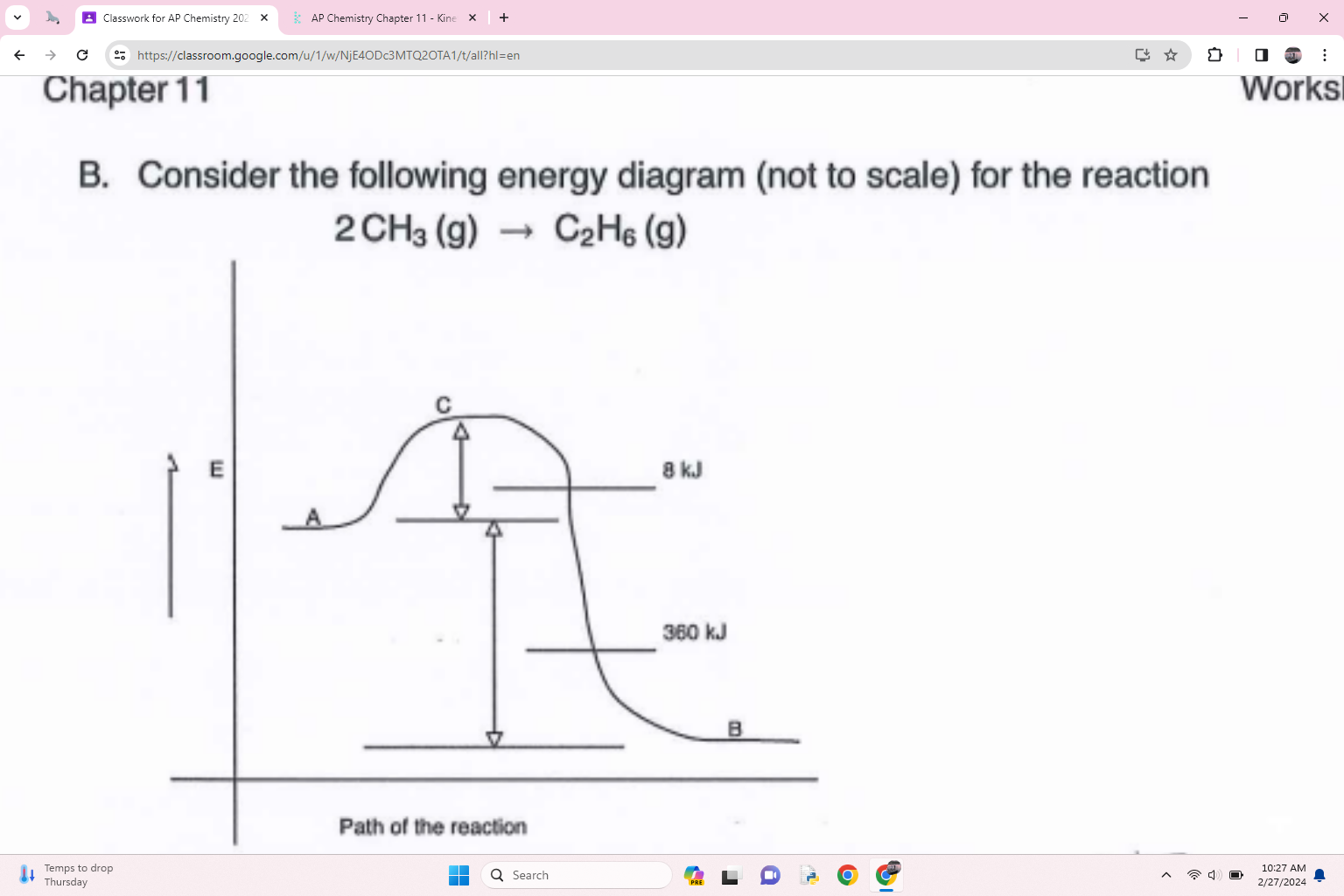
what is an species at point C called
activated complex