QM 1 - translational motion
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
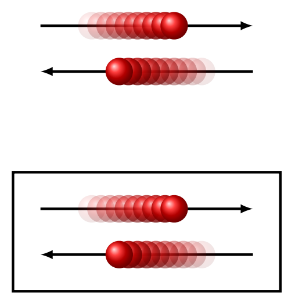
explain the difference between free translation and confined translation
free = constant speed, no interactions, any energy (not quantised)
confined = energy is restricted to certain values, interactions with wall
for free translational motion - what is potential and kinetic energy?
no interactions = nothing to slow it down, speed it up or change direction of motion
no potential energy, energy is purely kinetic
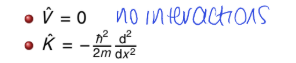
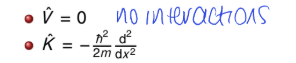
what is the Schrodinger equation for free translational motion?
what is the wavefunction? use Euler formula
values of k?
can be written as linear combination or as sine and cosine
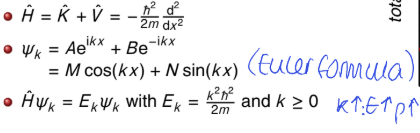
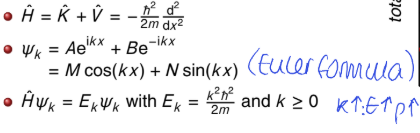
what does this mean?
there is no restriction, k can be any non negative real number
all energies are allowed -free translational motion is not quantised
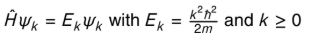
explain what it means if k = 0, and if k>0
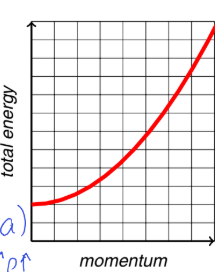
for free translational motion, show energy vs momentum graph
explain what graph shows
if k = 0 the particle is still
if k > 0 , the particle is moving and energy equals as shown
graph shows energy and momentum vary smoothly and continuously = shows no restrictions on k
what are the conditions of the box for particle in a box?
how are the walls described?
potential energy is 0 inside the box
box length is L
walls are infinitely repulsive (potential energy suddenly shoots up to ∞). this means no transfer of E to the wall
the wall is infinitely hard = no bounce back/deformation
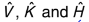
what are the values of these operators?
same as in free translation
purely kinetic
what does infinite repulsion mean about boundary conditions for particle in a box?
at the walls (x=0 and x=L), the wavefunction must equal 0
particle cannot be found there
this means that |ψ|2 = 0 and ψ = 0


what is the consequence of these boundary conditions?
not every wavelength fits properly between the walls
must be a sine function that starts at 0, oscillates and then returns to 0 at x=L
must be a whole number of half-wavelengths
this leads to quantisation
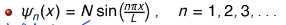
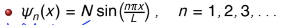
what is n? what is N?
n is the quantum number (can only take positive integer values)
the value of n is the same as the number of half wavelengths the wavefunction has
N is the normalisation constant
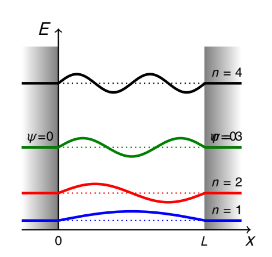
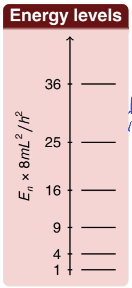
show diagram of particle in a box wavefunctions for each quantum number up to n = 4
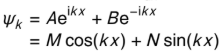
explain why cos term is not involved in particle in a box and how this is achieved
ψ=0 when x =0 but cos(0)=1
M = 0 to maintain this
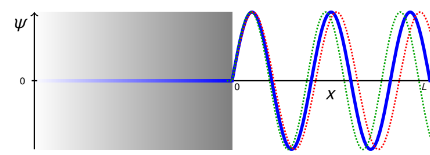
explain how the red and green wavelengths are incorrect
the wavelength of red is too long as it hasnt completed enough oscillations by the time it gets to x=L
there is a discontinuity at x=L as it doesnt hit zero
the wavelength of green is too short, it oscillates too quickly and doesnt land on 0 when x=L
this leads to a discontinuity
wavelengths must be continuous
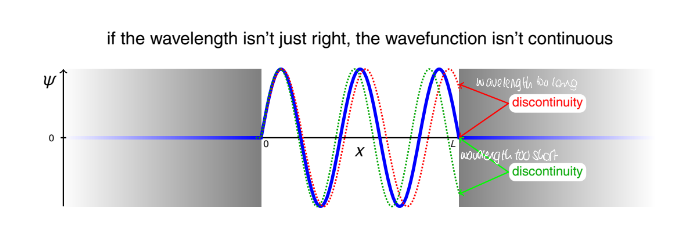
explain how the wavelengths not being continuous leads to quantisation of energy
hint: momentum
only some wavelengths being acceptable
since wavelength is connected to momentum through the de Broglie equation (p=h/λ)
momentum is connected to KE (K=p2/2m)
restricting wavelengths means restricting energy
what is the ground state for particle in a box
n =1
no nodes, single arch - 1 half wavelength
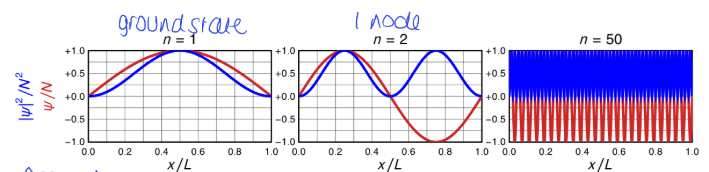
what happens as n increases? 3
number of nodes (n-1) increases
curvature of ψ increases, λ decreases, K increases
correspondence principle
why does KE increase as n increases in particle in a box?
the more curved the function is the more KE
the components are moving faster
what is the correspondence principle for particle in a box?
non uniformity of |ψ|2 becomes harder to detect
as you go macroscopic, there is constant probability of finding the particle anywhere in the box
quantum mechanics resembles classical - spends equal time everywhere
what is the zero point energy of particle in a box?
the lowest energy
confined particle can never stay still
why is the zero point energy not zero?
explain in terms of uncertainty and wavefunction shape
the uncertainty principle says that as the uncertainty of x is finite (confined to region of finite size), the uncertainty of p cannot be zero. if momentum isnt precisely zero, KE cant be zero either
the wavefunction must be zero at the walls but it cannot be zero everywhere (zero probability of finding the particle in the box)
wavefunction must be curved which means KE
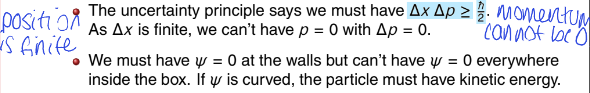
how do the energy gaps change as you go up in particle in a box? why?
what is gap proportional to?
energy gaps increase with n
gap between n and n+1 is proportional to 2n+1
the potential rises more steeply than the potential of a harmonic oscillator (which has constant gaps)
what is the energy gap and ZPE of particle in a box proportional to?
what about for macroscopic?
h2/mL2 (energy scale)
this gives energies that matter for electron in box the size of a chemical bond
for macroscopic, it is too small to be observed
dont notice quantisation when looking at macroscopic
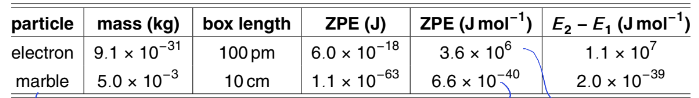
explain what this shows
electron in 100 pm box vs marble in 10 cm box
for electron, the ZPE is a few million joules per mole. the energy gap between 2 and 1 is 3x that - these are chemically significant
for the marble, the ZPE is much smaller (of the order of 10-40 Jmol-1) and undetectably small. the energy gap is also essentially zero. energies so small cannot be detected
quantisation is there in principle but for macroscopic, the ELs are so close together that they form what looks like a continuous spectrum
= correspondence principle
when is separation of variables applicable?
when the Hamiltonian is a sum of independent terms
coordinates that appear in one term do not appear in any other
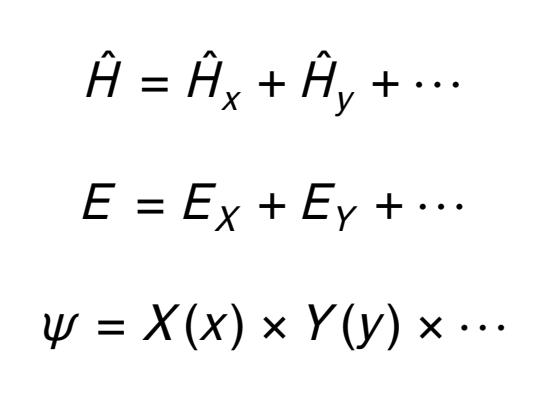
what is the wavefunction for a particle in a two or three dimensional box?
product of wavefunctions for a particle in a one dimensional box
what is the energy of a particle in a two or three dimensional box?
sum of the energies for each one dimensional component
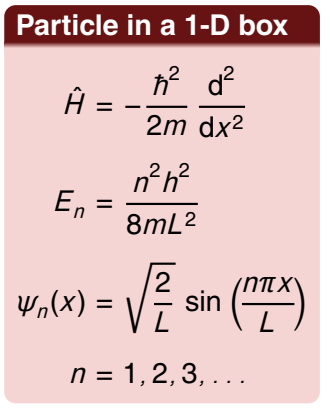
for a particle in a 1D box - what is the Hamiltonian? what does the energy depend on? and what is the wavefunction?
hamiltonian is the KE operator (second derivative wrt to x)
energy depends on a single quantum number n
wavefunction is a sin function with x as its variable

for a particle in a 2D box - what is the Hamiltonian? is the separation of variables applicable? what is the energy? what is the wavefunction? how many quantum numbers?
hamiltonian has two KE terms - one involving the second derivative wrt x and the other involving the second derivative wrt y
separation of variables is applicable
the two terms are independent and dont interfere with each other
2D can be split into a pair of 1D problems
since separation of variables is applicable, the total E is a sum of two independent 1D energies
wavefunction is a product of two independent 1D wavefunctions
there is now two quantum numbers, one for each dimension
show the Schrödinger equation for 3 dimensions
show particle in a 1D box wavefunctions (n=1 and n=2 overlayed) and explain positive and negative lobes
ground state has a single arch and is positive everywhere in the box
the wavefunction of the first excited state has a node in the middle and has one positive and one negative lobe
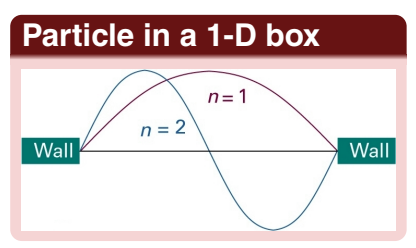
show particle in a 2D box wavefunctions for nx=1 or 2 and ny=1 or 2
explain the lobes
for ground state (nx=ny=1) there is a single positive region
nx=2 and ny=1 there is a quantum of excitation in x, there is a vertical nodal line at x=Lx/2
nx=1 and ny=2 there is a quantum of excitation in the y direction so there is a horizontal nodal line at y=Ly/2
for nx=ny=2 there are nodes in both directions which creates a pattern of 4 lobes with alternating signs
for a square box where Lx=Ly=L, what is the energy formula?
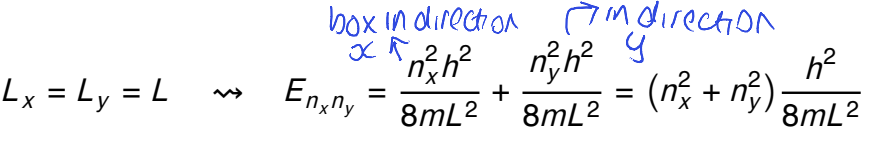
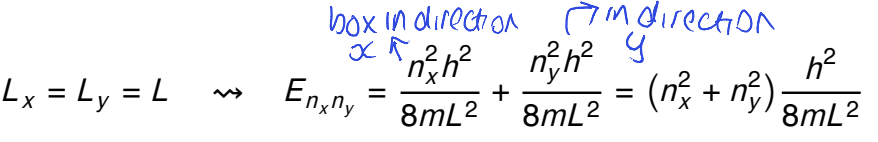
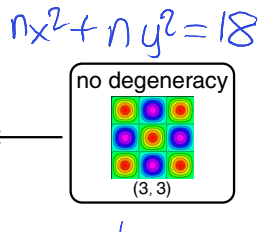
show whether (nx,ny)=(3,3) is degenerate or not
show diagram
there is only one state where you can get 18
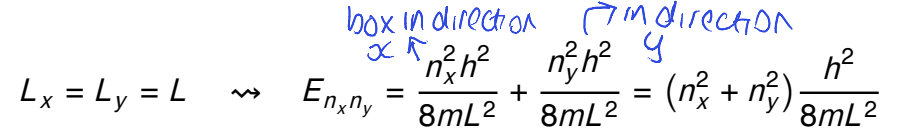
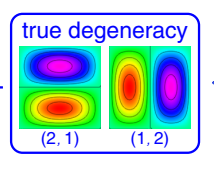
show whether (nx,ny)=(1,2) and (nx,ny)=(2,1) are degenerate or not
show diagram
nx2+ny2=5 in both cases
there is symmetry in the box so if you rotate by 90 degrees, you swap the x and y directions and the wavefunctions transform into each other
the wavefunctions have the same energy of excitation along x or y
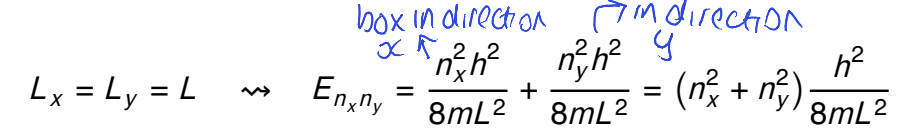
give an example of accidental degeneracy (for example use nx2+ny2=50)
show diagram
(7,1)/(1,7) and (5,5) are not related to each other by symmetry but they are degenerate as they have the same energy
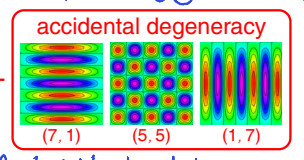
classically, how does a particle act when meeting a barrier? how is this different in quantum mechanics?
classic - either bounces back or goes through (depending on whether it has enough energy or not)
reflected or transmitted
quantum - both can happen
may undergo ‘tunnelling’
what is tunnelling?
penetration into or through a classically forbidden region
classically doesn’t have enough energy to get to
how does the probability of tunnelling change with height or width of the potential barrier?
probability decreases with increasing height or width of potential barrier
how does the probability of tunnelling change with particle mass?
probability decreases with increasing particle mass
what can graph of potential energy barrier be simplified to?
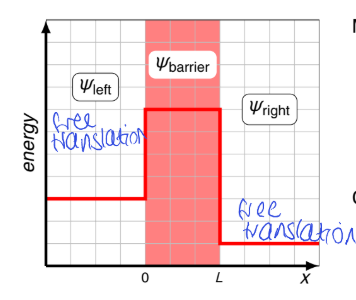
for a particle in a box with a finite barrier, what is the wavefunctions to the left, to the right and inside the barrier?
how does the wavefunction behave at x=0 and x=L (at each boundary)?
ψleft, ψright and ψbarrier
wavefunction must be continuous at each boundary and its slope (first derivative) must be continuous
for a particle in a finite box, what is are the values of x for ψleft, ψright and ψbarrier?
what are the boundary conditions of ψ and ψ’?

why must the wavefunctions be continuous at the boundaries for the particle in a finite box?
potential energy is finite everywhere
when it jumps to infinity as in an infinite box, we can tolerate the slope being discontinuous
what is the energy vs x graph for particle in a finite box?
what are the reflection and transmission probabilities?
R + T = 1
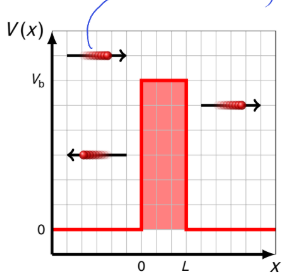
what happens if E>Vb? what are R and T? how does this differ from classical physics?
R>0 and therefore T<1
there is quantum reflection. classically we would expect R=0 and T=1
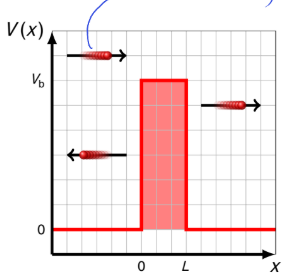
what happens if E<Vb? what are R and T? how does this differ from classical physics?
T>0 and therefore R<1
there is tunnelling into the classically forbidden region
classically we would expect T = 0 and R = 1
when does tunnelling take place (what values)?
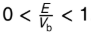
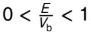
what happens as E/Vb → 0?
T → 0 and R → 1
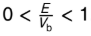
what happens as E/Vb → 1?
T increases and R decreases
for tunnelling, what happens as m increases?
T decreases and R increases
for tunnelling, what happens as L increases?
T decreases and R increases
(barrier becomes longer/wider)
when does tunnelling stop? (ie what size?)
heavier than a proton becomes classical
at the correspondence principle limit
show transmission probability vs E/Vb graph
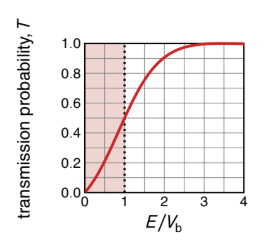
explain how it is that E/Vb <1
for tunnelling, E<Vb