Electronegativity - bond polarity
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is used to measure electronegativity?
Pauling scale (0-4)
What does electronegativity depend on? (3)
• The nuclear charge.
• The distance between the nucleus and the outer shell electrons.
• The shielding of the nuclear charge by electrons in inner shells
Why is electronegativity greater if the atom is smaller?
Because the nucleus is closer to the shared outer main level electrons
How does electronegativity change going across a period and why?
Increases because the nuclear charge increases, the number of principle energy levels remain the same and the atoms become smaller.
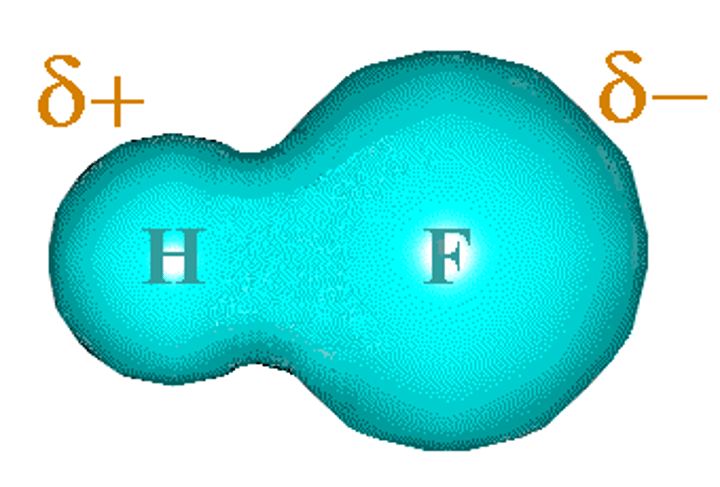
What is polarity?
The unequal sharing of electrons between atoms that are bonded covalently
What is electronegativity?
The ability of an atom to attract electrons in a covalent bond
How is electronegativity affected if the nuclear charge is larger?
Electronegativity is greater
How does electronegativity change going down a group and why?
Decreases because there is more shielding by electrons as the atomic radii increases
Where are the most electronegative atoms found on the perodic table?
At the top right hand corner of the periodic table, ignoring noble gases.
Which are the most electronegative atoms?
Flourine (4.0), oxygen (3.4), chlorine (3.2) and nitrogen (3.0)
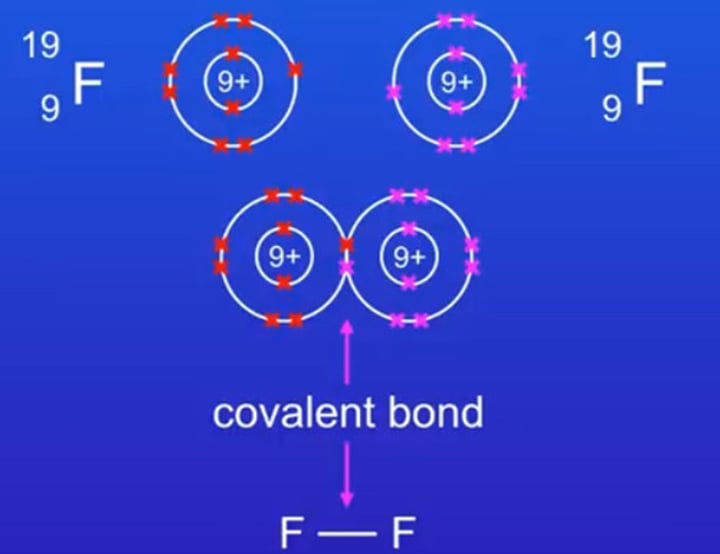
Electron diagram of fluorine molecule
Electron cloud around fluorine molecule
Even bubbles around both atoms
Is the covalent bond between fluorine molecules polar or non-polar?
Non-polar
Are C-H bonds polar or non-polar and why?
Non-polar because atoms have very similar electronegativities
Electorn diagram of hydrogen fluoride
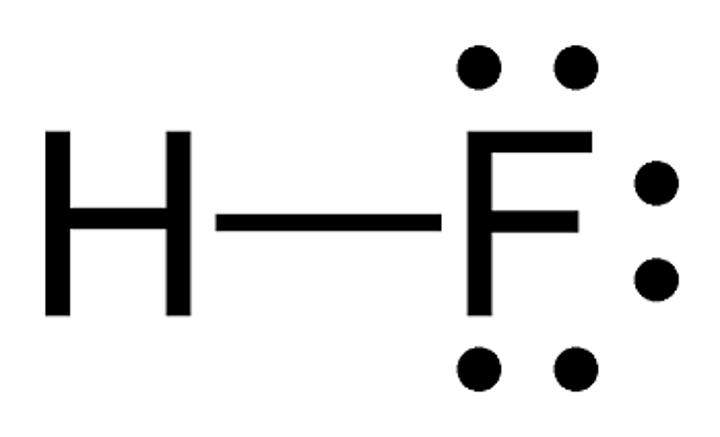
Electron cloud of hydrogen fluoride