Classification of Matter
1/90
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
what is periodicity?
the trends observed across periods and down groups
atomic radius trend across a period
generally decreases
atomic number increases = increased nuclear charge
additional electrons added in same shell so shielding between nucleus and outer electrons roughly the same
increased nuclear charge = stronger attraction between nucleus and outer shell electrons = decreasing atomic radius
atomic radius trend down a group
generally increases
down a group the number of shells increases = outer shell electrons further from nucleus = weaker attraction
electrons in inner shells repel outer electrons = increased shielding
what occurs to the ionic radius of a cation
decreases compared to parent atom
loss of electrons, often loss of outer shell = fewer repulsions so greater attraction to nucleus
what occurs to the ionic radius of anions?
generally increases compared to parent ion
extra electrons gained = increased repulsion = decreased attraction to nucleus
what is the ionic radius trend across a period?
for cations it decreases
where anions form it sharply increases and then decreases again as nuclear charge increases
what is the ionic radius trend down a group?
increases
what is the ionization energy?
amount of energy required to remove one mole of electrons from one mole of atoms of an element in the gaseous state to form one mole of ions
what are the units of ionization energy?
kJmol-1
what is the ionization energy trend across a period?
generally increases
nuclear charge increases, shielding remains rougly constant
so stronger attraction to nucleus
so more energy needed to overcome stronger attraction
what happens to the ionization energy between the last element in one period and the first in the next?
rapid decrease due to increased distance between nucleus and outer shell electrons and increased shielding
what is the ionization energy trend going down a group?
generally decreases
number of energy levels increases so outer shell electrons are farther from nucleus
experience increased shielding
so attraction between nucleus and outer shell electrons decreases
so less energy required to remove outermost electron
what is electron affinity?
amount of energy released when one mole of electrons is gained by one mole of atoms of an element in the gaseous state to form one mole of ions
what are the units of electron affinity?
kJmol-1
what are the electron affinity trends across a period?
becomes more negative (more exothermic)
nuclear charge increases, atomic radius decreases
so stronger attraction to nucleus
what are the electron affinity trends down a group?
less negative (less exothermic)
number of shells increases, atomic radius increases, shielding increases
so electrons are further from the nucleus and have less attraction
what is electronegativity?
the ability of an atom to attract a pair of electrons towards itself in a covalent bond
what is the trend of electronegativity across a period?
increases
nuclear charge increases
shielding is constant
atomic radius decreases
so stronger attraction to nucleus
what is the trend of electronegativity down a group?
decreases
nuclear charge increases
increased shielding (more shells)
larger atomic radius
weaker attraction to nucleus
physical properties of group 1 metals
soft, easy to cut
shiny when cut
conduct heat and electricity
low melting points
low densities
chemical properties of group 1 metals
react readily with oxygen and water vapour
react vigorously with water to form an alkaline metal hydroxide solution and hydrogen gas
what is the equation for group 1 metal + water?
group 1 metal + water → group 1 metal hydroxide + hydrogen
what would be observed in a reaction between lithium and water?
floats and reacts slowly
bubbles of hydrogen gas
clear, colourless solution formed
what would be observed in a reaction between sodium and water?
reacts more vigorously than lithium but less than potassium
bubbles of hydrogen gas
sodium melts to form a silvery ball that moves on the surface
clear, colourless solution formed
what would be observed in a reaction between potassium and water?
reacts more violently than sodium and lithium
hydrogen gas released rapidly
potassium melts into a shiny ball that dashes around on the surface
produces a lilac flame
forms a clear, colourless solution
what is the trend in reactivity down group 1?
increases
atoms get larger
outermost electron further from the nucleus
more shielding
so weaker attraction
so outer shell electron lost more easily
what is the state and colour of fluorine at room temperature?
yellow gas
what is the state and colour of chlorine at room temperature?
pale green gas
what is the state and colour of bromine at room temperature?
orange-brown liquid, readily evaporates to form brown gas
what is the state and colour of iodine at room temperature?
grey-black solid that sublimes to form a purple vapour
what is the colour of chlorine in water?
green-blue
what is the colour of bromine in water?
orange
what is the colour of iodine in water?
dark brown
what is the trend in reactivity down group 17?
decreases
atomic radius increases
number of shells + shielding increases
electron affinity becomes less negative
attraction between nucleus and outer shell electrons decreases so less reactive
what occurs in displacement of halogens with halide ions?
a more reactive halogen will displace a less reactive halogen from an aqueous solution of its halide
what does amphoteric mean?
a substance can act as an acid and a base
what causes acid rain?
non-metal oxides dissolving in atmospheric water
how does ocean acidification occur?
CO2 dissolves in seawater to form carbonic acid, which dissociates to hydrogen carbonate ions and hydrogen ions, increased hydrogen ions lowers pH
what are the characteristic properties of transition elements?
variable oxidation states
high melting points
magnetic properties
catalytic behaviour
formation of coloured compounds
ability to form complex ions with ligands
how does transition metal magnetism arise?
unpaired electrons in d orbitals
each spinning electron generates a tiny magnetic dipole
paired electrons spin in opposite directions cancelling the dipole
where they are unpaired they can become aligned in an external magnetic field and produce magnetism
how do you form a permanent magnet?
by heating and cooling iron, nickel or cobalt in a magnetic field
magnetic dipoles align and the material retains its magnetism
why can transition elements catalyse redox reactions?
variable oxidation state means they can accept and lose electrons
they can be oxidised and reduced repeatedly so catalysing redox reactions
what is a hetergenous catalyst?
one in a different state to the reactants
what is a homogenous catalyst?
one in the same state as the reactants
what do catalytic converters do?
reduce air pollution in car exhausts
what is a ligand?
a molecule or ion that donates a lone pair of electrons
why do transition metal compounds appear coloured?
in an octahedral complex there is d-orbital splitting
a wavelength of visible light is absorbed
this excites an electron
and the complementary colour is observed
what does the structural formula show?
the spatial arrangement of atoms and bonds in a molecule
what is a skeletal formula?
simplified way of representing organic molecules
carbon-carbon bonds shown as straight lines
hydrogen atoms bonded to C omitted unless part of a functional group
what is a stereochemical formula?
shows the three dimensional arrangement of atoms or groups around a chiral carbon
what is a chiral carbon?
bonded to four different groups or atoms creating a tetrahedral shape with bond angles of 109.5º
what is the functional group name of an alkane?
alkyl
what is the functional group name and formula of an alkene?
alkenyl
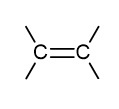
what is the functional group name and formula of an alkyne?
alkynyl
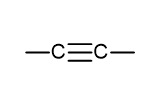
what is the functional group name and formula of an halogenoalkane?
halogeno
F-, Cl-, Br-, I-
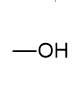
what is the functional group name and formula of an alcohol?
hydroxyl
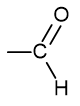
what is the functional group name and formula of an aldehyde?
carbonyl
what is the functional group name and formula of a ketone?
carbonyl (ketone)
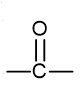
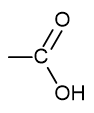
what is the functional group name and formula of a carboxylic acid?
carboxyl
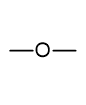
what is the functional group name and formula of an ether?
alkoxy
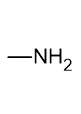
what is the functional group name and formula of an amine?
amino
what is the functional group name and formula of an amide?
amido
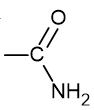
what is the functional group name and formula of an ester?
ester
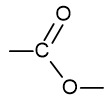
what is the functional group name and formula of an aromatic?
phenyl
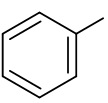
what is a homologous series?
a family of similar compounds with the same functional group and similar chemical properties
what are the characteristic features of a homologous series?
each member has the same functional group
same general formula
similar chemical properties
each member differs by -CH2-
gradually changing physical properties
what is the trend in melting point/boiling point for a straight chain homologous series?
as the number of carbon atoms increases so does the bp and mp
why does bp/mp increase in a straight chain homologous series?
each member differs by CH2
increases molecular size and area so stronger london dispersion forces
stronger forces = more energy required to separate molecules
what are structural isomers?
compounds with the same molecular formula but different structural formulae
what are the three types of structural isomerism?
functional group isomerism
positional isomerism
branched chain isomerism
what is functional group isomerism?
where different functional groups result from the same molecular formula
what is positional isomerism?
where isomers have the same molecular formula and the same functional group, but the functional group is in a different place in the chain
what is branched chain isomerism?
compounds have the same molecular formula but their longest hydrocarbon chain is not the same
what are stereoisomers?
compounds with the same atoms bonded to each other but arranged differently in space
what are the two types of stereoisomerism?
conformational
configurational
what is conformational isomerism?
compounds with the same atoms bonded to each other but different spatial arrangement due to free rotation around a single sigma bond
what is configurational isomerism?
compounds with the same atoms bonded to each other but different spatial arrangements that cannot be interconverted by rotation due to a pi bond
what is a cis isomer?
has two functional groups on the same side of a carbon-carbon double bond
what is a trans isomer?
has two functional groups on opposite sides of a carbon-carbon double bond
what are optical isomers?
chemical that contain a chiral carbon (a carbon with four different atoms or groups attached)
what are enantiomers?
a pair of chiral molecules that are non-superimposable mirror images of each other
what are diastereomers?
compounds with more than one chiral centre
what are the chemical properties of enantiomers?
optical isomers have different smells
what are the physical properties of optical isomers?
identical physical properties except
enantiomers can rotate the plane of polarised light (optically active)
diastereomers are not optically active
how can you determine between enantiomers?
by the rotation of plane polarised light
what is a racemic mixture?
mixture containing a 50:50 ratio of enantiomers
is a racemic mixture optically active?
no because each enantiomer will cancel the other’s effect on plane polarised light
what is detected on a mass spectrum?
only positively charged fragments
what does the highest peak of a mass spectrum show?
the parent ion or the Mr of the whole compound
what does IR spectroscopy measure?
changes in vibrations of atoms when they absorb IR
what must a molecule have to be IR active?
a permanent dipole