Geochemistry Exam 2
1/122
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
123 Terms
isotope
An element with a different number of neutrons than expected.
more
Isotopes always have ____ neutrons than protons.
stable isotopes
Isotopes that are not radioactive and do not decay.
both
Some elements, like carbon can have _____ stable and radiogenic isotopes.
less, more
Light isotopes have ___ neutrons than a known standard and heavy isotopes have ___ neutrons.
Pure water with no salt or other chemicals.
What is Vsmow?
Pee dee Belemnite; a geologic formation in the Carolinas that has carbon and a marine deposit of fossils
What is PDB?
equilibrium fractionation
When isotopes of an element are redistributed among chemical substances or phases in a dosed, well-mixed system at equilibrium.
Water evaporation; gas becomes lighter
What is an example of equilibrium fractionation?
heavy/light
The fractionation ratio is..?
heavier, higher, positive
Enriched = ?
lighter, lower, negative
Depleted = ?
((Rsample/Rstandard) - 1) x 1,000
How do you solve for fractionation in change per mil?
fractionation
The distribution of heavy and light isotopes in a thing changes due to some process.
more
Covalent bonds cause fractionation as heavier isotopes form a ____ stable bond than light isotopes.
Ionic
____ bonds have a minimal effect of fractionation.
higher
When it comes to fractionation, heavier isotopes prefer to bond to elements of ______ oxidation states.
solid, liquid
When it comes to fractionation, heavier isotopes form a _____ first so the _____ becomes depleted.
greatest
Fractionation is ________ at lowest temperature.
Dissolves CaCO3
CaCO3 + CO2 > Ca(2+) + H2CO3*
Precipitates stalactite
Ca(2+) + H2CO3* > CaCO3 + CO2
kinetic fractionation
When isotope reacts, diffuses, evaporates faster than another due to the process and catalysts.
lighter
Kinetic fractionation results in the ______ isotope accumulating in the product.
faster
Lighter molecules react ______.
Rayleigh fractionation
Evaporation/precipitation/condensation cycles on global scale; occurs when a material changes phase
fractionation ratio
How fractionation is changing between two substances relative to each other.
Temperature, latitude, continentality, and humidity
What factors influence isotope ratios?
Formation of rocks, climate (CO2), various photosynthesis pathways, etc.
How can isotopes impact everyday life?
radiogenic isotopes
Isotopes that spontaneously decay into daughter isotopes.
alpha radiation
The type of radioactive decay when a particle is lost with 2 protons, 2 neutrons, and a +2 charge.
alpha radiation
This is an example of what kind of radioactive decay?

beta decay
The type of radioactive decay when an electron is lost; occurs in series with alpha decay.
beta decay
This is an example of what kind of radioactive decay?
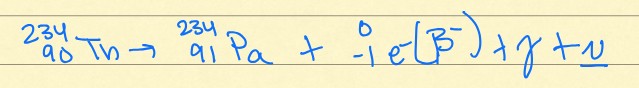
positron decay
The types of radioactive decay when there is an emission of positron (positively charged electron).
positron decay
This is an example of what kind of radioactive decay?
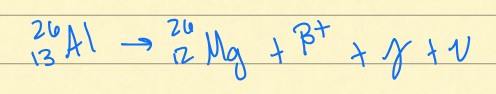
positrons
________ form when a proton converts to a neutral state and releases a positive particle.
gamma
In alpha, beta, and positron decay - ______ radiation is released, stabilizes the nucleus to a lower energy, and a more stable state.
fission decay
Nucleus splits into two or more nuclei; produces an alpha particle.
half-life
The amount of time it takes for some substance to decay to half its original mass.
Pt = P0 x e^-kt
What is the equation for half-life?
zero-order
Do these graphs represent a zero, first, or second-order rate?
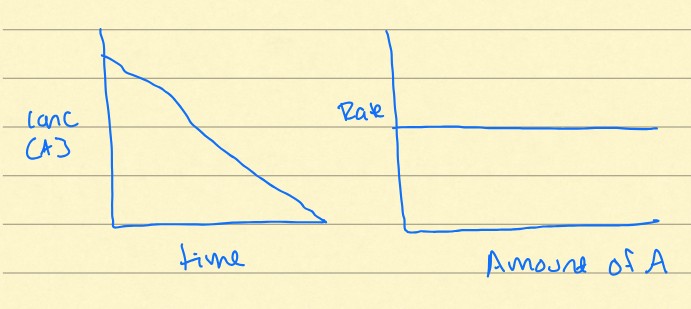
Dissolution of some salts
What kind of chemical reactions follow a zero order rate?
First-order
Do these graphs represent a zero, first, or second-order rate?
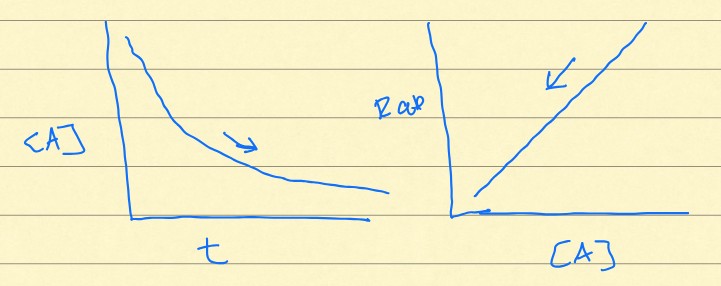
Population growth, radioactive, hill slope
What things could be represented by a first-order rate?
Carbon-14
What isotope is used in carbon dating?
Burning of fossil fuels and atomic bomb tests
Radiocarbon dating must be corrected for “old carbon” in the form of..?
Acids have H+; bases have OH-; limited to aqueous solution
Arrhenius definition?
Acid is proton donor (gives H+); base is a proton acceptor (accepts H+); conjugate acids and bases
Bronsted-Lowry definition?
Acid dissociation constant (Ka)
When the conjugate pair dissociates, we describe this as..?
-log(Ka)
pka = ?
[H+][OH-]
kw = ?
completely
Strong acids _________ dissociate in water.
large
Strong acids have a ______ Ka.
small
Weak acids have a _____ Ka.
don’t
Weak acids ____ fully dissociate in water.
polyprotic acids
Acids with multiple hydrogens
acid dissociation constant
For every deprotonation step, there is a separate..?
pH
At different __ values, different species are dominant.
base, acid
In each step of a phosphate bjerrum plot, in each step the phosphorus and hydrogen goes from being ____ to an ____.
amphoteric substances
Something that can be an acid or a base depending on pH and the system.
CO2
H2CO3 (carbonic acid) is present everywhere due to the partial pressure of ___ in the atmosphere.
H2CO3
_______ is critical to controlling the pH of water in a system.
Encompasses the CO2(g) > CO2(aq); CO2(aq) + H2O > H2CO3
What is H2CO3*
Sum of CO2, H2CO3, HCO3, and CO3 in an aqueous system
dissolve inorganic carbon (DIC)
alkalinity
Ability of a solution to accept acidity and not change pH.
titration
Alkalinity is operationally defined by _______, a process that adds acid slowly to a solution while pH is measured after each addition.
meq/L
What are the units for alkalinity?
buffer
A solution or solid that resists change (pH) when something is added.
buffer capacity
Measure of buffering in a solution.
buffered
Karst waters are well..?
charge
Alkalinity is measured in meq because titrations are just a ______ balance.
buffer zone 1
Very low pH; lots of [H+]; addition of acid is negligible; OH- becomes neutralized because of high [H+]
buffer zone 2
Between pk1 and pk2; add acid (HCO3>H2CO3*); add base (H2CO3>HCO3-); species transfer back and forth and buffer between the two species
buffer zone 3
High pH; lots of [OH-]; well buffered; transfer of species between HCO3- and CO3(2-); any addition of acid is quickly neutralized
constituents
Chemical reactions add __________ or stuff to H2O.
mineral dissolution/precipitation; weathering; reactions in solution
What are some ways that more constituents can be added to water?
water cycle, dissolved vs particulate, mineral solubility/reaction types, water measurements, Eh-pH diagrams
What are the five big picture controls on water chemistry?
total dissolved solids
Measure of all dissolved components in water - organics, inorganics, ions, colloids, in ppm
Non aqueous phase liquids (NAPLs); both dense and light non-aqueous phase liquids (DNAPLs & LNAPLs)
What things in H2O don’t get dissolved?
colloids
Small solids with surface chemistry interactions.
suspended items
Particulates, bacteria, viruses, some colloids
Na+, Ca(2+), Mg(2+), K+
What are the monoatomic ions that can be found in water?
HCO3-, OH-, SO4(2-), PO4(3-)
What are the polyatomic ions that can be found in water?
sorbed species
Things attached to mineral surfaces
ionic charge (z) / ionic radius (r)
Ionic potential is defined by the ratio of electric charge to radius of the ion:
ionic potential
How strongly or weakly the ion will be electrostatically attracted to ions of opposite charge; how strongly or weakly the ion will repel ions of the same charge
strong, high, small
High z/r is associated with _____ bonding, ____ charge, and ____ radius
weaker
Lower z/r generally means ______ bonding.
solubility
Cations with high z/r can form stable complexes which impacts __________ and geological reactions/processes.
oxygen
Low z/r has weak bonds with _______ if they can form at all. It makes them soluble and enter into solids at relatively low temperatures.
high ionic potential
Charge is more focused; strong bonds with oxygen and repel other cations
K+, Na+, Sr+
What are some examples of low ionic potential monoatomic ions?
NO3-, SO4(2-), PO4(3-)
What are some examples of high ionic potential polyatomic ions?
Al, Ti, Fe, Mn (multiple oxidation states)
What are some examples of intermediate ionic potential transition metals?
transition metals, oxides, and hydroxides
What molecules/atoms have intermediate ionic potential?
solubility
pH controls mineral..?
pH
Not all minerals have the same solubility pattern relative to..?
karst
Consists of specific surface and subsurface features formed by the dissolution of soluble rocks containing adequate porosity with exposure to chemically aggressive water over appropriate time.
Px = nx/ntotal
How do you calculate partial pressure?
acid mine drainage
Generates SO4 from the oxidation of pyrite.