Benzene Structure and Azo Dyes
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Draw the kekule’s structure of benzene and the accepted structure and list the simularities and differences
Both
All bond angle are 120
Planar structure
3 sets of electrons repelled
Kekule Structure
Alternating single and double bonds (suggesting different bond lengths)
Two dimethyl isomers expected
Hydrogenation enthalpy 3x that on cyclohexene due 3 double bonds instead of 1
Undergoes addition reactions
Should decolourise bromine water
Actual Benzene
C-C all the same length
Only 1 isomer of dimethyl
Hydrogenation enthalpy is less than 3x that of cyclohexene - less energy released so more stable due to delocalisation
Undergoes substitution reactions
Won’t decolourise bromine water
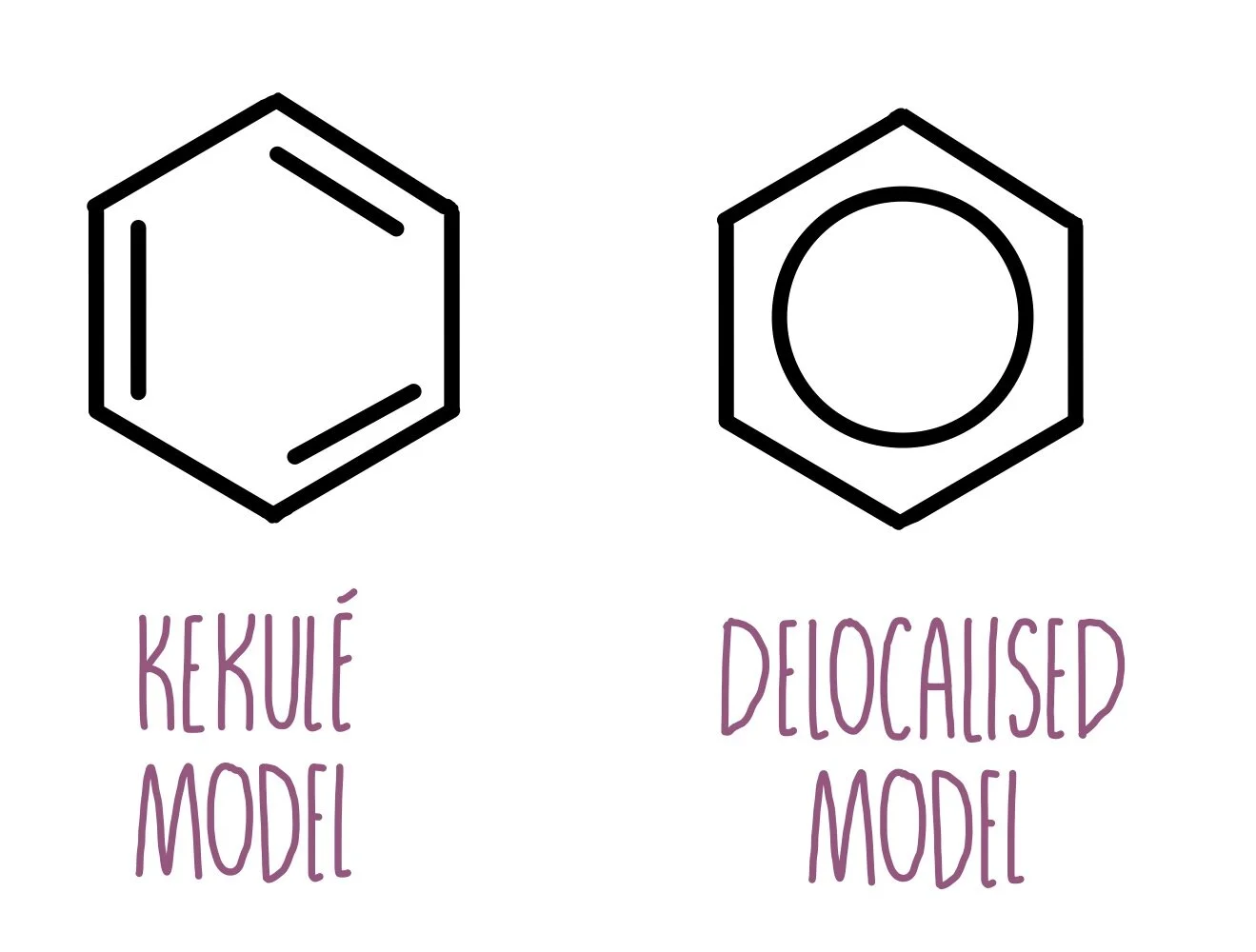
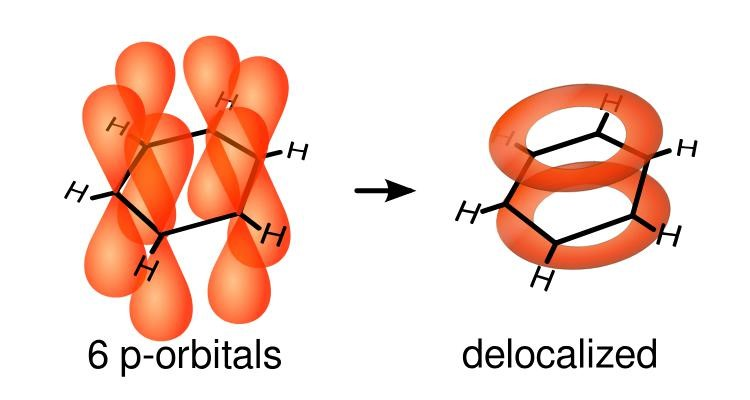
Explain, using a diagram, how benzene is delocalised
Each carbon atom forms 3 sigma bonds
This leaves the 4th electron in the p orbital of each carbon
The 6 p orbitals overlap forming a pi cloud of electron density
This is two rings of electrons one above and below the plane of the carbon atoms
The electrons move freely within this pi system. They are delocalised
What is the difference between an aromatic compound and an arene
Aromatic compounds - contain a ring of delocalised electrons
Arenes - contain a benzene ring
Show the general mechanism for the electrophilic subsitution of benzene with elctrophile, E+

Give the reagents, conditions, electrophile, equations (generation of electrophile and regeneration at catalyst) and mechanism for the halogenation of benzene
Reagents - Cl2 or Br2
Conditions - Anhydrous AlFe3/AlBr3/FeCl3/FeBr3 Catalyst
Electrophile - Cl+ or Br+
Generation of electrophile - AlCl3 + Cl2 → AlCl4- + Cl+
Regeneration of catalyst - AlCl4- + H+ → AlCl3 + HCl

Give the reagents, conditions, electrophile, equations (generation of electrophile and regeneration at catalyst) and mechanism for the nitration of benzene
Reagents - Concentrated HNO3
Conditions - Concentrated H2SO4 (Catalyst and dehydrating agent) and less than 55C
Electrophile - NO2+

Give the reagents, conditions, electrophile and equation for the sulphonation of benzene
Reagents - Concentrated H2SO4
Conditions - Heat under reflux
Electrophile - SO3H+

Give the reagents, conditions, electrophile and equation (including the generation of electrophile) for the Friedel-Crafts Reaction (adding carbon chains to a benzene ring)
Reagents - haloalkane with correct R group
Conditions - Anhydrous AlCl3 catalyst, reflux
Electrophile - CH3+ (R+)
Generation of electrophile - AlCl3 + CH3Cl → CH3+ + AlCl4-

Give the reagents, conditions, electrophile and equation (including the generation of electrophile) for the Friedel-Crafts Acetylation Reaction
Reagents - Acyl chloride
Conditions - Anhydrous AlCl3 catalyst
Electrophile - e.g. CH3CO+
Generation of electrophile - AlCl3 + CH3COCl → CH3CO+ + AlCl4-

Show how benzene can be halogenated using halogen carrier
Reagents - Polarised halogen molecule, AlCl3 catalyst

Azo dyes contain an azo functional group. Draw the general structure of an azo functional group
R—N=N—R
Explain, using equations, how azo dyes form
Step 1: Formation of diazonium compound
Reagents - Aromatic amine, sodium (III) nitrate, dilute HCl
Conditions - <5C
Step 2: Coupling Reaction
Reagents - diazonium compound, phenol (or phenolamine), NaOH
Conditions - alkaline (NaOH), <5C
Observations - coloured ppt of azo dye formed

Explain how colour arises in organic molecules and what colour depend on
Molecules must have an extended delocalised system (alternating single and double bonds)
Radiation (usually visible light) is absorbed by electrons according to the energy gap (E=hv)
Electrons in extended delocalised system move to higher energy levels
Complementary colour is transmitted
The energy gap and so frequencies depend upon
- Amount of delocalisation
-Size of chromophore
-Bonding and functional groups on chromophore
Smaller delocalised system = larger energy gap and so higher frequency absorbed (e.g. UV light which will transmit all visible colours)
What is a chromophore and what are the impacts of its side groups on organic colour and solubility
Chromophore - the part of the molecule that gives it its colour i.e. all the parts with alternating double and single bonds
Side groups can affect the colour
Groups with lone pairs (e.g. OH) - delocalise in the pi system changing the energy gap and therefore the frequency absorbed
Acyl groups and nitro groups contain double bonds and so extend the delocalised system
Ionic groups like sulfonate groups allow ion-dipole bonds to form with water, increasing solubility as these are stronger than hydrogen bonds in water
How can dyes stick to fabric
Ionic bonding - ionic bonds form between NH3+ o the fibres and the sulfonate group on dye molecules
Covalent bonding
- Mordanting - Functional groups with lone pairs form a complex ion by dative bonding with a metal ion
- Fibre reactive dyes - Functional groups added to the dye molecules that will allow it to react with the fibre, e.g. condensation reaction
Intermolecular bonding
- Hydrogen bonds form between OH on fibres and NH2 or OH groups of dye
- ID-IDs
How can aldehydes be prepared from primary alcohols
Reagents - primary alcohol and K2Cr2O7/H+(aq)
Conditions - Heat under distilation
Observations - Orange → Green
Equation - CH3CH2CH2OH + [O] → CH3CH2CHO + H2O
How can ketones be prepared from secondary alcohols
Reagents - secondary alcohol and K2Cr2O7/H+(aq)
Conditions - Heat under Reflux
Observations - Orange → Green
Equation - CH3CH(OH)CH3 + [O] → CH3COCH3 + H2O
How can aldehydes and ketones be distinguished using K2Cr2O7/H+
Warm with K2Cr2O7/H+
Aldehyde - Orange → Green
Ketone - Stays orange as ketones can’t be oxidised further
How can aldehydes and ketones be distinguished using Fehlings Solution
Warm with Fehling’s solution
Aldehyde - Blue solution forms brick red precipitate (CuO)
Ketones - Solution stays blue
How can aldehydes and ketones be distinguished using Tollens Reagent
Warm with Tollens Reagent
Aldehyde - Silver mirror forms
Ketones - No change
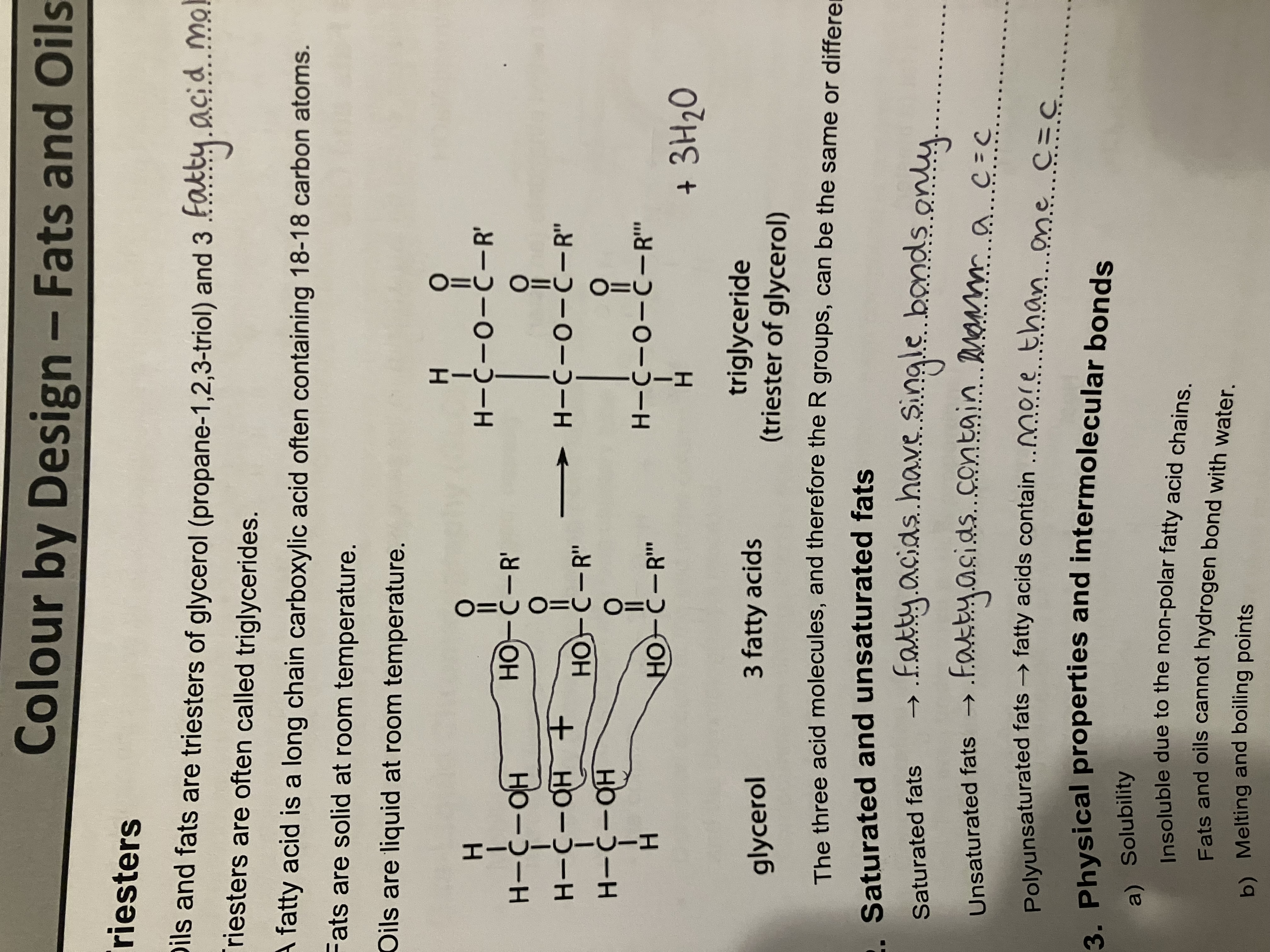
Show the mechanism for the reaction between an aldehyde and HCN/KCN. Give the type of reaction, reagents and equation
Nucleophillic Addition
Reagents - acidified HCN or KCN
Equation - CH3CH2CHO + HCN → CH3CH2CH(OH)CN
Hydroxynitrile formed
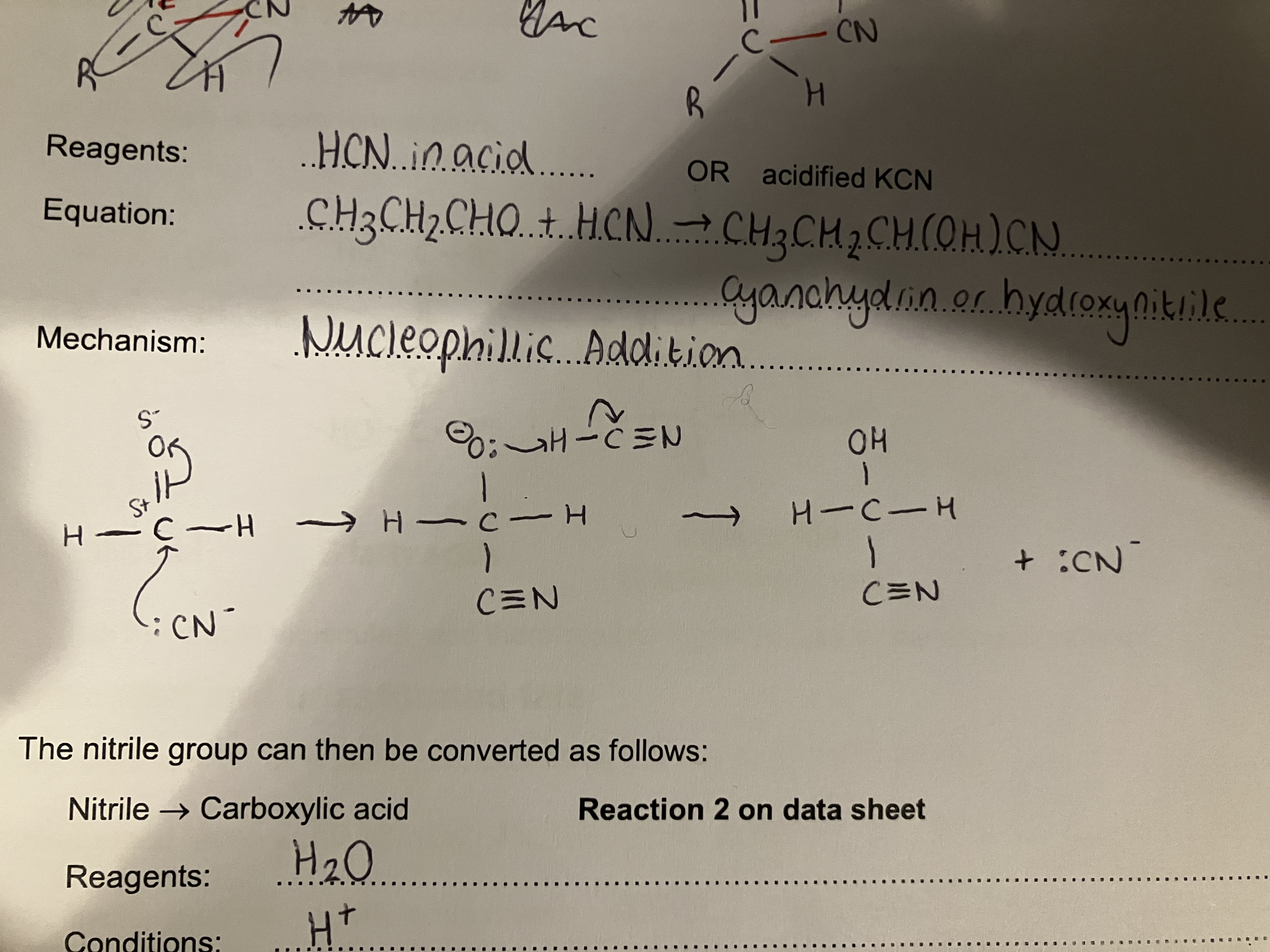
Show how triesters (triglycerides) from glycerol and 3x fatty acids
What are saturated and unsaturated fatty acids
Saturated - single bonds only
Unsaturated - contains C=C
Unsaturated fatty acids can be cis (z) or trans (E), what does this mean
Cis (Z) - adjacent parts of chain are next to each other across the double bond
Trans (E) - adjacent parts of chain are on opposite sides across the double bond
Show how a triglyceride can be hydrolysed, give the reagents and conditions
Reagents - Concentrated NaOH
Conditions - heat
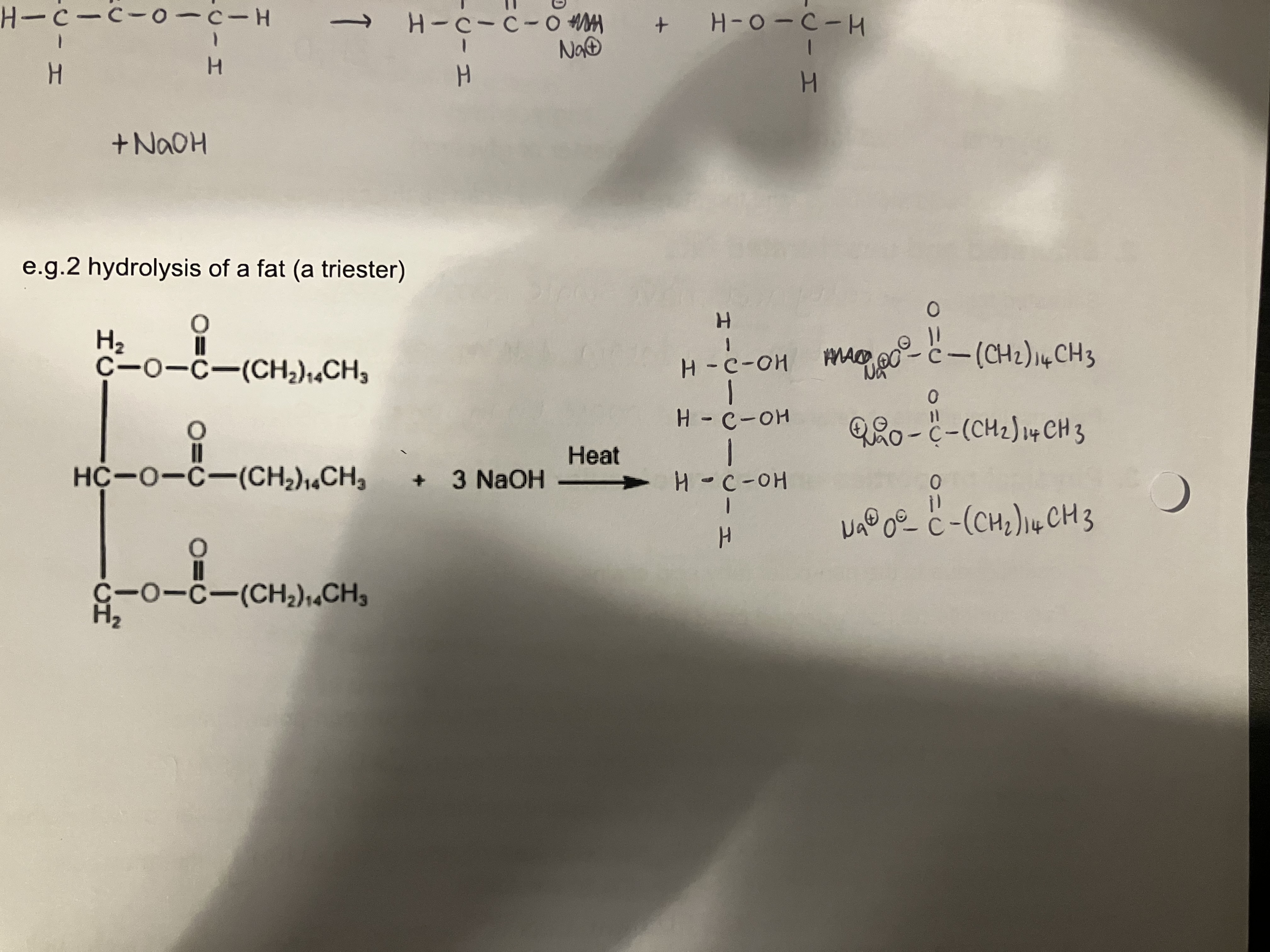
What is the mobile phase and stationary phase of Gas-Liquid Chromatography
Mobile - inert carrier gas, usually nitrogen
Stationary phase - high boiling point liquid on a porous support in a long capillary tube
How can GLC be used to find the concentration of components
The area under each peak is proportional to the amount/conc of that component
This can be compared with a calibration curve
What is retention time (GLC)
The time taken from the injection of the sample, for the component to leave to column - can be used to identify a component
What types of intermolecular bonds form between triglycerides and where do they form
ID-IDs between fatty acid chains
PD-PDs between C-O/C=O on glycerol