chem of life chaper 8
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
whats a mass number?
number of both neutrons AND protons
whats an atomic number
the number of protons in the nucleus of an atom
what does a nuclear reaction do?
changes the arrangement or number of particles in a nucleus
name 3 ways nuclear reactions are different from chemical reactions
- they dont need to be balenced bc elements can change identity
- they involve a lot more energy
- not affected by temperature or catalysts
what is a nuclear symbol?
identifies the element and shows the mass and atomic number of an atom

whats an isotope?
Atoms of the same element that have the same amount of protons but different numbers of neutrons
what are stable nuclei?
nuclei of naturally occuring isotopes that remain unchanged indefinitely
What are radioisotopes?
nuceli that are unstable and eventually break into 2 or more pieces
what are beta particles?
electrons produced from a nuclear reaction
What is an alpha particle?
Two neutrons and two protons, the same as a helium nucleus
(when an alpha particle is omitted from an element, it loses 2 neutrons and 2 protons, changing the element)
What is a positron?
a particle with the mass of an electron but a positive charge
what 2 things must happen for a nuclear equation to be balanced?
- the sum of the mass numbers of the products and the reactants must be equal
- the sum of atomic numbers of products and reactants must be equal
what is alpha decay?
when an element loses an alpha particle (2 protons, 2 neutrons. mass # changes by -4)
what is beta decay?
when a neutron turns into a proton and a electron (beta particle) mass # doesnt change
(ex: 19 protons and 21 neutrons-> 20 protons and 20 neutrons)
what are the 2 forms of energy that can appear from a nuclear reaction?
kinetic energy
electromagnetic radiation
what is kinetic energy in a nuclear reaction?
when tiny particles shoot out of the nucleus very fast (ex: alpha and beta particles)
whats a photon?
the smallest possible particle of light that travels at the speed of light
what is electromagnetic radiation?
energy that travels through space as waves (photons) at the speed of light
what is gamma radiation?
electromagnetic radiation of very high energy
what happens when a reaction produces only gamma radiation?
the gamma emission produces a more stable nucleus of the same element
what is ionizing radiation?
radiation that has enough energy to remove an electron and turn a molecule into a positively charged ion
what is a radical?
very unstable ion with an odd number of electrons (missing one)
(most radicals attck neighboring molecules creating more radicals)
what are two ionizing radiation detectors?
geiger counter and scintillation counter
what is the equivalent radiation dose?
the amount of tissue damage that is produced by ionizing radiation
what does the equivalent does depend on?
the amount of energy we absorb
the type of radiation
what is the radiation weighing factor
the relative effect of each type of radiation compared to x-rays
What is activity?
the measurement of how many atoms break down in a second. unit of measurement- curie
how are alpha emitters hazardous?
if theyre ingested or inhaled theyll produce extensive damage to nearby tissues
how can beta radiation be blocked?
by materials such as cloth, plastic, and wood.
what happens when a beta particle hits an atom?
they can produce x-rays
what can block gamma and x-ray radiation?
very dense materials such as a concrete wall or a sheet of lead
what is exponential decay?
when something decreases by the same percentage over equal time periods. (ex- cut in half over and over until its gone)
what is half life?
the time require for half the sample to breakdown
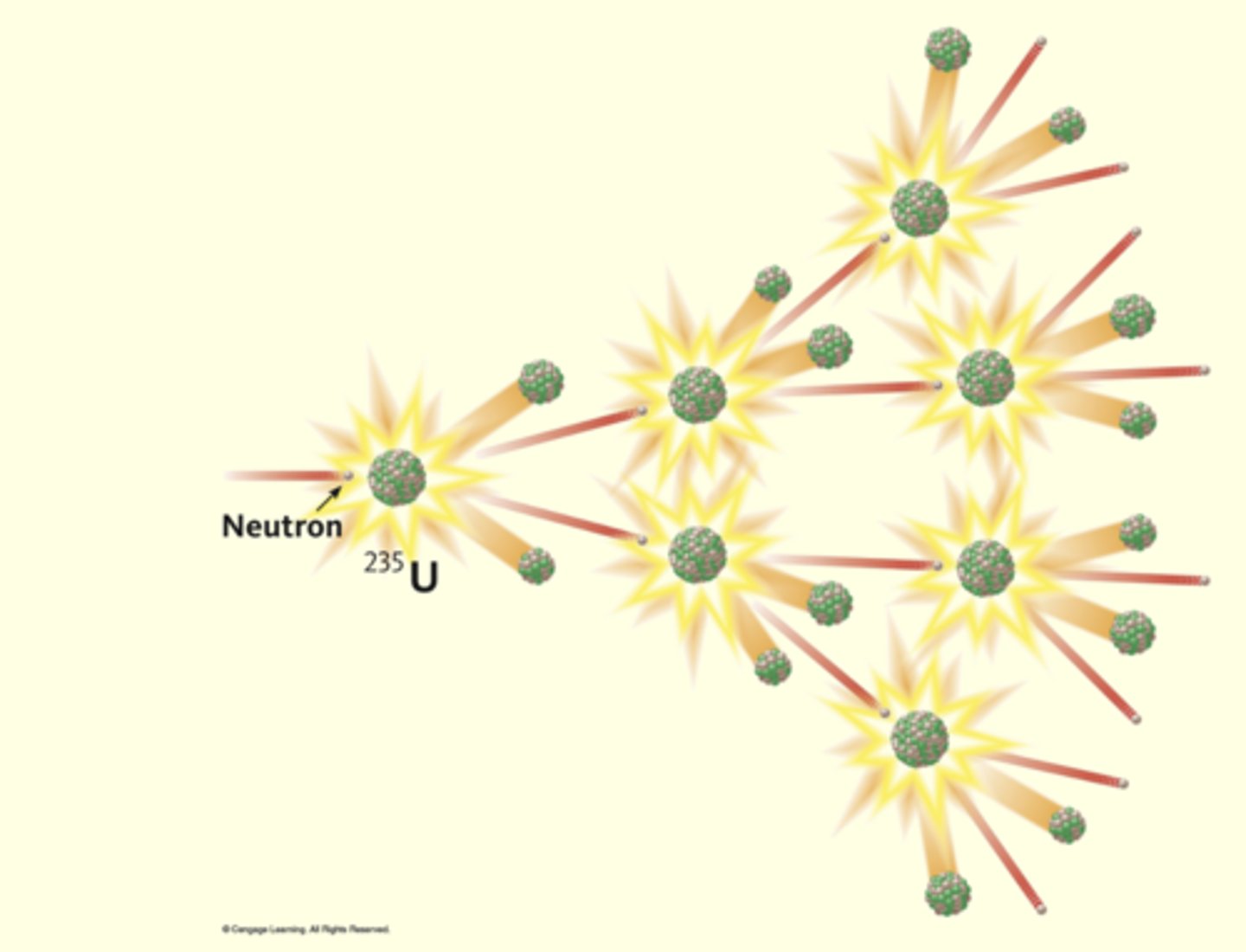
what happens during nuclear fission?
when a neutron strikes a large nucleus it splits into 2 similarly sized pieces. all fission reactions produce more neutrons which create more fisson reactions
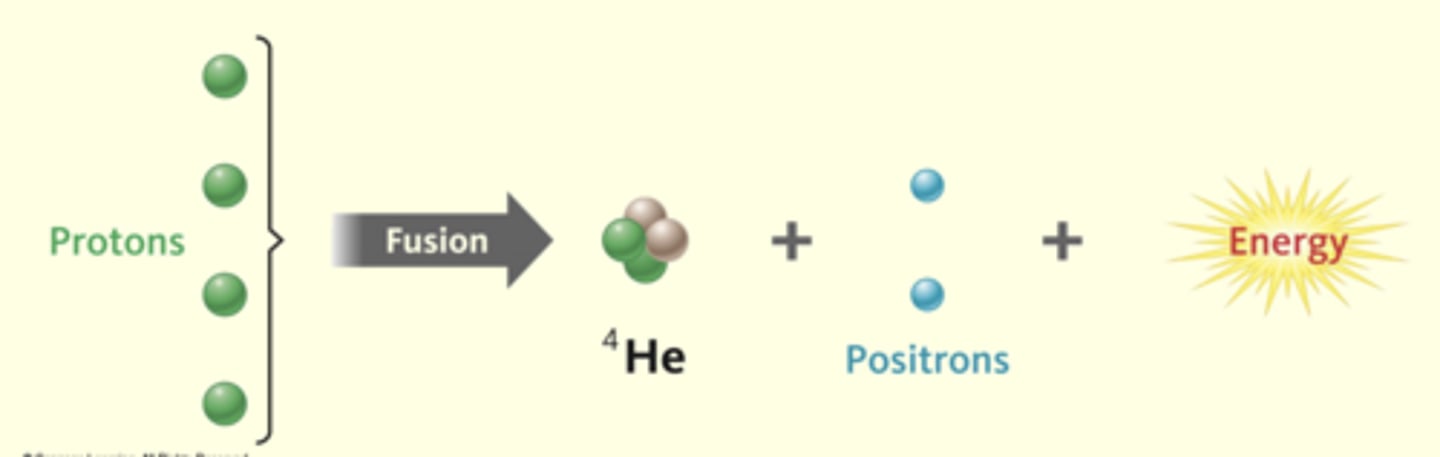
what is nuclear fusion?
when two or more small nuclei collide and stick together to make a larger nucleus.
(requires a lot of energy and high temperatures)