Atomic Orbitals & Electron Configuration: Quantum Numbers, Rules, and Notation
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
What is an atomic orbital?
A region of space around the nucleus where an electron is likely to be found.
How do atomic orbitals differ from the Bohr model?
Orbitals represent the probability of finding an electron rather than fixed circular paths.
What is the maximum number of electrons in the s sublevel?
2 electrons.
What is the maximum number of electrons in the p sublevel?
6 electrons.
What is the maximum number of electrons in the d sublevel?
10 electrons.
What is the maximum number of electrons in the f sublevel?
14 electrons.
What does the principal quantum number (n) indicate?
The energy level or shell of an electron.
What are the possible values for the principal quantum number (n)?
1, 2, 3, 4, ...
What does the angular momentum quantum number (l) indicate?
The sublevel or orbital shape.
What are the values for the angular momentum quantum number (l) for p orbitals?
l = 1.
What does the magnetic quantum number (m) indicate?
The specific orbital orientation in space.
What are the possible values for the magnetic quantum number (m) for p orbitals?
-1, 0, +1.
What does the spin quantum number (mₛ) indicate?
The direction of electron spin.
What are the possible values for the spin quantum number (mₛ)?
+½ or −½.
What is the Aufbau principle?
Electrons fill orbitals starting with the lowest energy orbitals first.
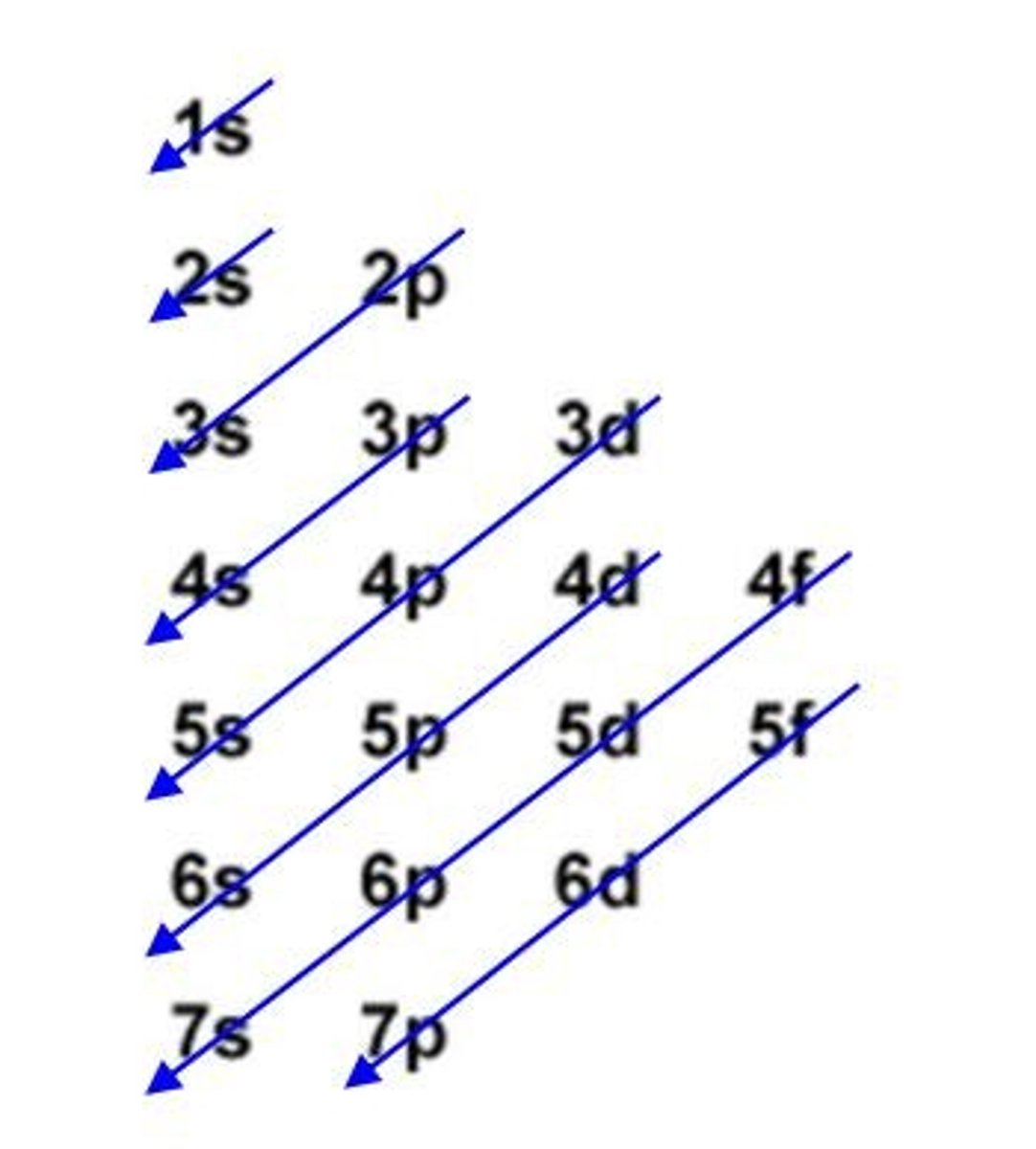
What is the Pauli Exclusion Principle?
No two electrons in an atom can have the same set of four quantum numbers.
What is Hund's Rule?
Electrons fill equal-energy orbitals singly before pairing to minimize electron-electron repulsion.
How is electron configuration notation written?
By listing the energy level, sublevel, and number of electrons (e.g., 1s² 2s² 2p⁶).
What is noble gas notation?
A shorthand for electron configuration that uses the noble gas from the previous period in brackets.
What is an orbital diagram?
A visual representation using boxes for orbitals and arrows for electrons.
How many valence electrons does chlorine (Cl) have?
7 valence electrons.
What is the electron configuration for phosphorus (P)?
1s² 2s² 2p⁶ 3s² 3p³.
What is the maximum number of electrons that can fit in the d sublevel?
10 electrons.
What is the electron configuration for calcium (Ca)?
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² or [Ar] 4s².
What sublevel does an electron with quantum numbers n = 4, l = 2 belong to?
The d sublevel.
How many unpaired electrons does nitrogen have?
3 unpaired electrons.