3.5- Chemical Kinetics
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is the rate equation?
rate= K [A]^n[B]^m
What is the rate equation not dependent on?
The stochiometric equation (ie the actual equation of the reaction)
If rate=k [C2H4]^2 [H3PO4], what order of reaction are ethene and phosphoric acid?
This reaction is 2nd order in respect to C2H4 and first order in respect to H3PO4
What is the order of the overall reaction?
It is third order overall (add powers)
Practice determining the rate equation maths
How do you determine the rate equation?
Look at initial conc of A and B in experiment 1, 2 and 3. See what changes and what stays the same and compare to changes in initial rate.
ie if conc of A stays the same in 1 and 2 but B doubles and the rate increases by 4 times, B is 2nd order as 2 squared= 4
If C is there but when its conc changes and the others stay the same and the rate stays the same, what is the order in respect to C?
Zeroth order
What change in the reaction conditions would cause the value of the rate constant to change?
Temperature and catalyst
If the conc of 'A' was increased, what would happen to the rate contant?
Stay the same
Deduce the units for the rate constant in rate= k [NO]^2[H2]
rate: moldm-3s-1
[NO]^2: mol2dm-6
[H2]: moldm-3
Therefore,
mol: 1= x + 2 + 1
x= -2
dm: -3= y - 6 - 3
y= 6
s= -1
mol-2dm6s-1
Define 'rate determining step'
The slowest step in a multistep reaction that determines the overall rate
How do you write a balanced equation for the overall reaction when the reaction is multi-step?
List all reactants for all steps and list all products for all steps, when they match then cancel to find what change occurs
Write a balanced equation for this reaction:
Step 1: (slowest) (CH3)2CCH2 + H+ -> (CH3)3C+
Step 2: (fastest) (CH3)3C+ + H2O -> (CH3)3COH + H+
(CH3)2CCH2 + H2O -> (CH3)3COH
Describe the role of H+ in this mechanism
Catalyst as used in step 1 and regenerated in step 2
Suggest a rate equation for the reaction
Step 1: (slowest) (CH3)2CCH2 + H+ -> (CH3)3C+
Step 2: (fastest) (CH3)3C+ + H2O -> (CH3)3COH + H+
Rate= k [(CH3)2CCH2] [H+]
For the mechanism:
Step 1: HBr + O2 -> HBrO2
Step 2: HBrO2 + HBr -> 2HBrO
Step 3: HBrO + HBr -> Br2 + H2O
Step 4: HBrO + HBr -> Br2 + H2O
If the rate equation is rate= k[HBr][O2], which step is the rate determining step?
Step 1 as the reactants match the rate equation
For:
Cl2 -> 2Cl (slow)
H2 + Cl -> HCl + H (fast)
H + Cl -> HCl (fast)
How would the rate of the reaction be affected if the initial concentration of Cl2 is doubled?
It would double (nb the rate constant would stay the same)
How would the rate of reaction be affected if the initial concentration of H2 is doubled?
It would not be affected
Learn how to determine the multiple steps from a balanced equation !!!
If balanced equation is
NO2 + CO -> NO + CO2
and the rate equation is
Rate= k[NO2]^2
what are the two steps?
Slowest step: 2NO2 -> NO + NO3
(nb: NO as one of final products, NO3 not one so must be reactant in second step)
Fastest step: NO3 + CO -> NO2 + CO2
(Only one NO2 molecule in balanced equation as they cancel)
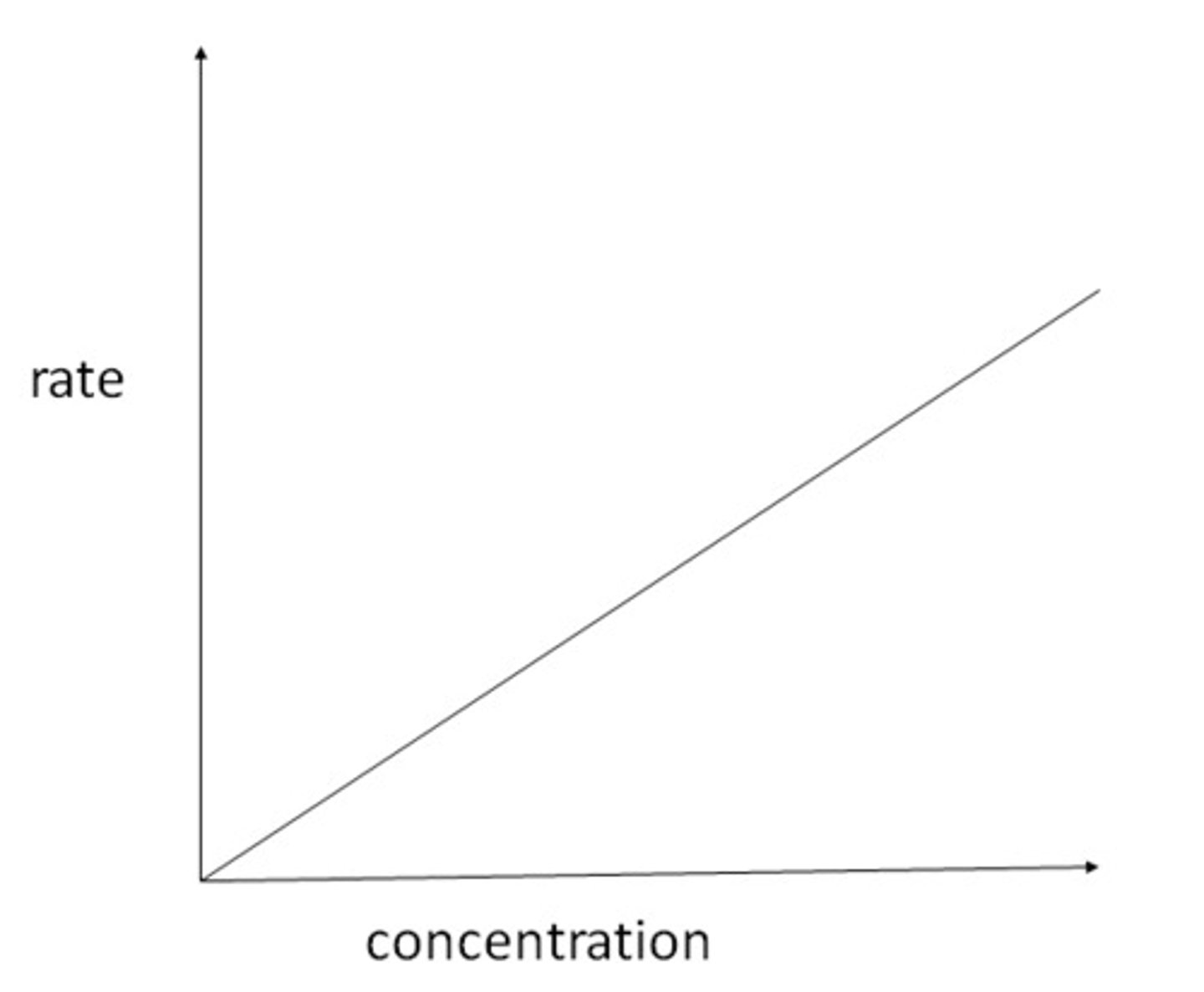
First order rate/concentration graph
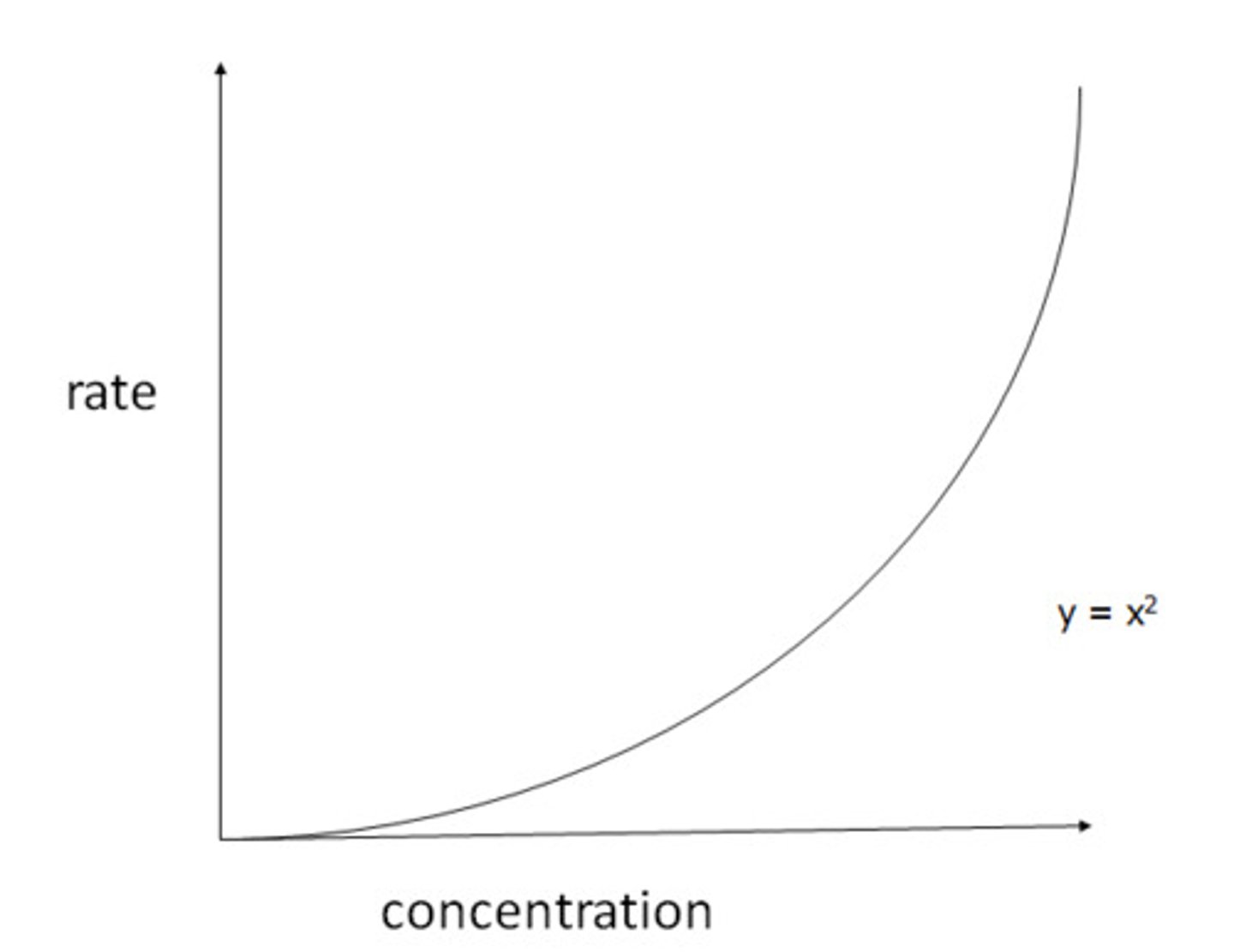
Second order rate/concentration graph
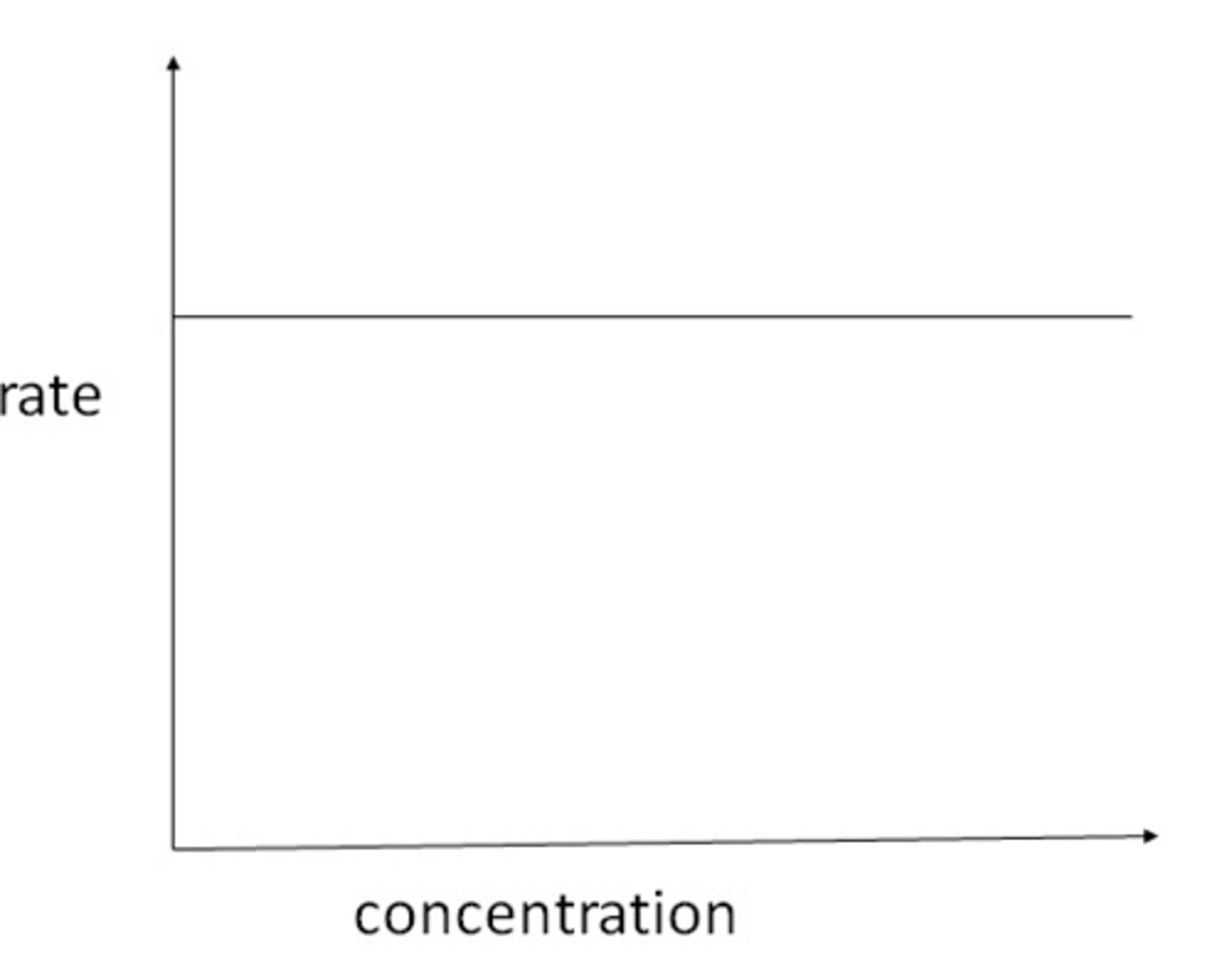
Zeroth order rate/concentration graph
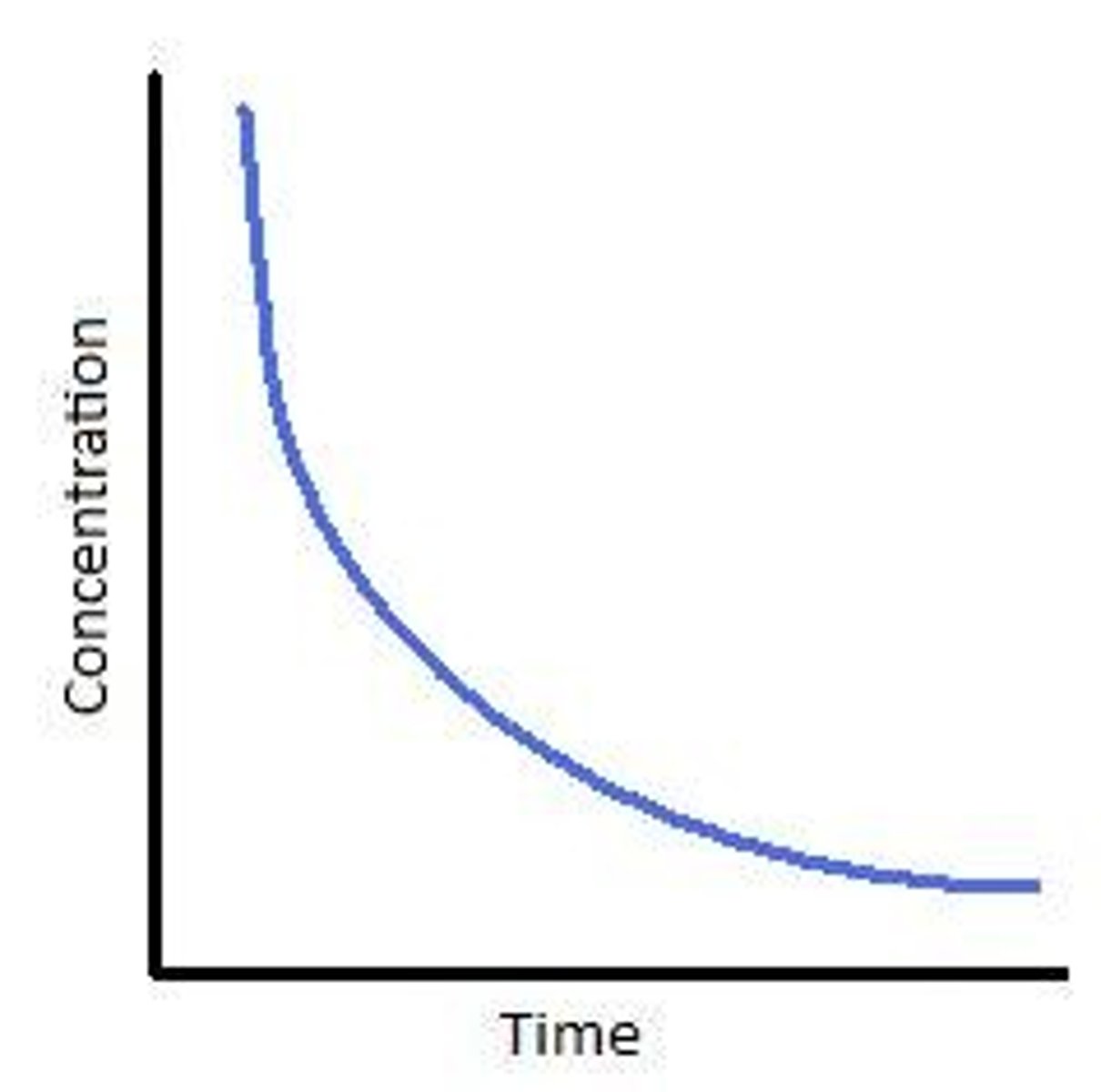
First order concentration/time graph
Zeroth order concentration/time graph
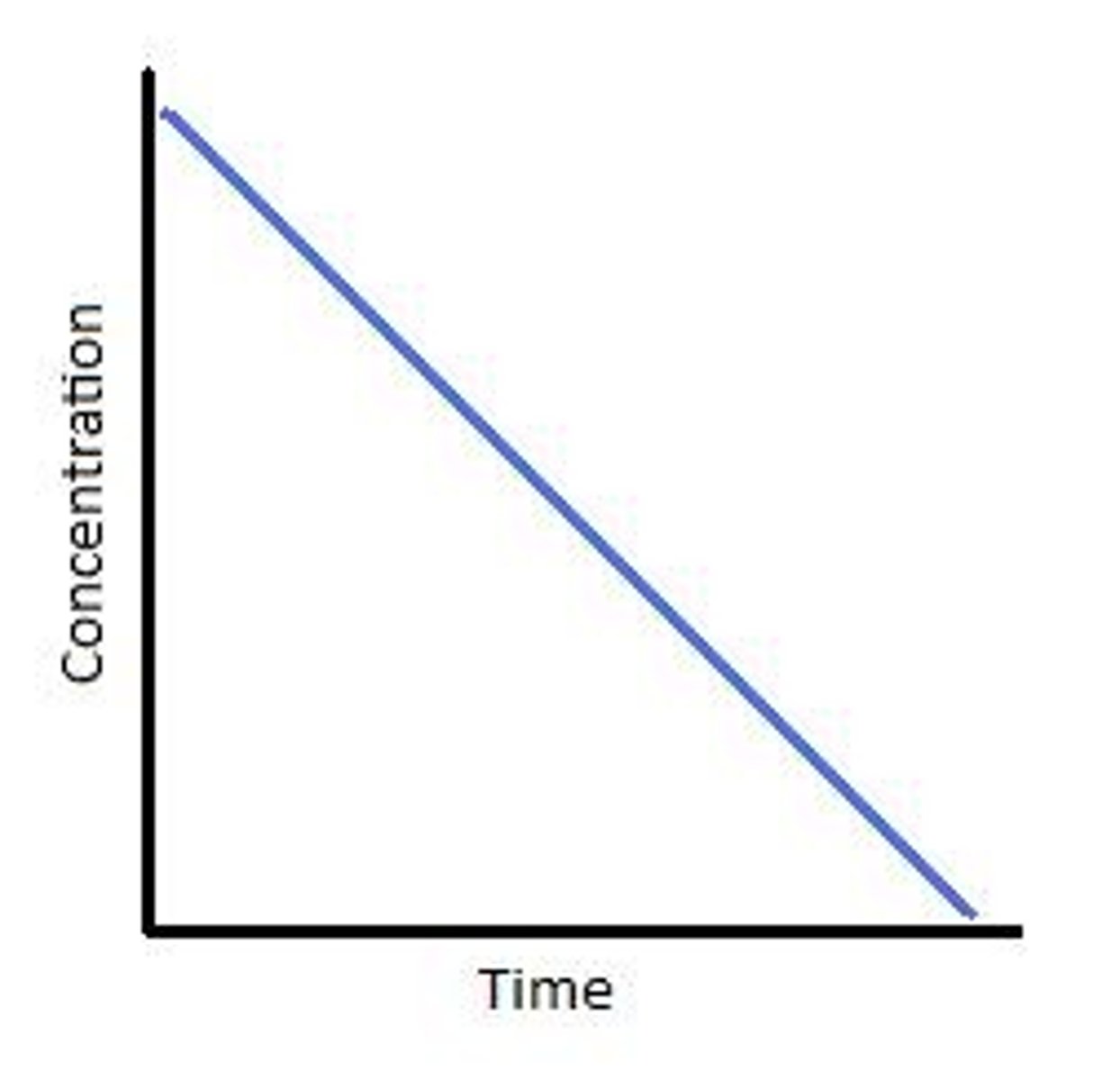
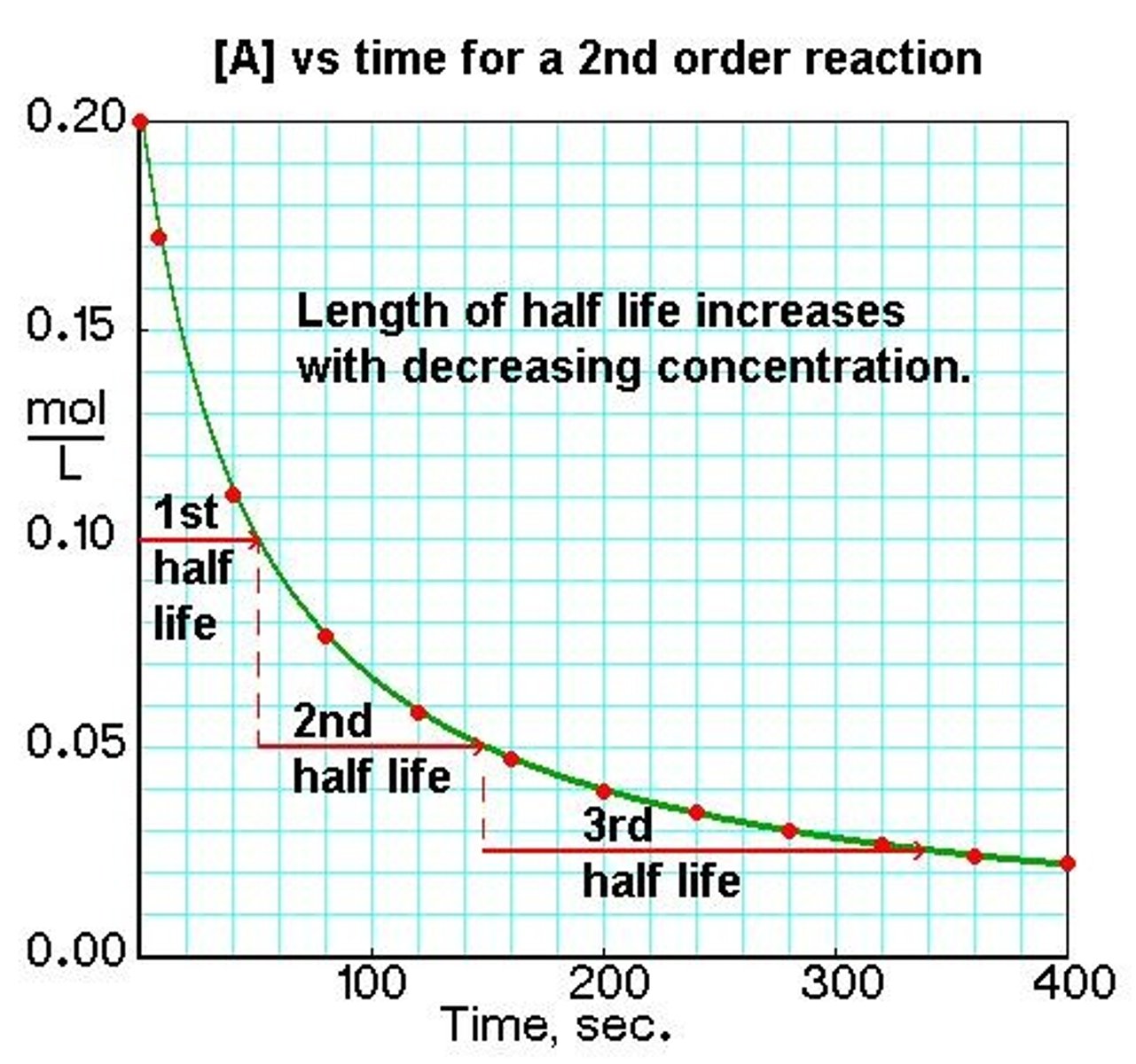
Second order concentration/time graph
What is the Arrhenius equation?
k = Ae^(-Ea/RT)
What does each part of the equation stand for and what are the units?
k: rate constant
A: pre-exponential factor
e: Natural log constant
Ea: activation energy (Jmol-1)
R: gas constant (JK-1mol-1) (8.31)
T: temperature (K)
How can it be written in linear form?
lnK = lnA - Ea/RT
Learn how to use linear form of equation using y= mx + c to find A and Ea
If the equation of the line was y= -4146.3x + 8.0651
what is Ea and A?
lnK= y
-4146.3= -Ea/R
Therefore, Ea= 4146.3 x 8.31
1/T= x
8.0651= ln A
A= e^8.0651
Rate= k[C][D]
In an experiment at 25 degrees C, the initial rate of reaction is 3.1 x 10^-3 moldm-3s-1 when the initial conc of C is 0.48 moldm-3 and the initial conc of D is 0.23 moldm-3.
Calculate a value for k.
3.1 x 10^-3= k x 0.48 x 0.23
k= 0.02807 mol-1dm3s-1
If k= 0.028, lnA= 16.9, R= 8.31 and T= 25 degrees C, what is the Ea of the reaction
lnK= -Ea/RT + lnA
ln0.028 = -Ea/ 8.31 x (25+273) + 16.9
Ea= 50705 Jmol-1
= 50.7 kJmol-1
Define order of reaction
The power to which the concentration of a reactant is raised in the rate equation
If H+ was a catalyst, what could be the effect on the rate of a reaction by decreasing the pH from 1 to 0?
Conc of H+ is 10x greater at 0 than at 1 so the rate will increase x10
If the rate equation is rate= k [N2O5], how would you answer whether an equation with 1 N2O5 or 2 N2O5 is the correct one?
Rate determining step must have one N2O5 molecule as the reactant so the reactants match the rate equation
What are the units of a second order reaction?
mol2dm-6
What is sampling?
Taking samples from the reaction mixture at regular time intervals
What is quenching?
The sudden stopping of a chemical reaction to allow for analysis to occur without the reaction proceeding further
How is quenching usually done?
By adding the sample to ice water
Why?
This cools and dilutes the reactants
What are the advantages?
Can be used for a large range of reactions
Disadvantages?
Labour and time intensive as each sample must be analysed individually
When is sampling only appropriate?
When a reaction mixture is homogeneous
Why not when heterogeneous?
Sample taken may not be representative of the overall mixture
Why can sampling be done when a reaction uses a heterogeneous catalyst?
Removing a sample takes it away from the catalyst, so the reaction rate is immediately reduced