atomic and nuclear phenomena
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
photoelectric effect
The photoelectric effect is a phenomenon in physics that occurs when light, usually in the form of photons, interacts with matter, leading to the emission of electrons from a material. This effect played a crucial role in the development of quantum theory.
Here are the key points about the photoelectric effect:
Emission of Electrons: When light shines on a metal surface, electrons can be emitted from the material. This emission of electrons is known as the photoelectric effect.
Quantized Energy: The photoelectric effect provided evidence for the quantization of energy. Classical wave theory predicted that increasing the intensity (brightness) of light should eject electrons with any energy. However, experiments showed that electrons were only emitted if the light had a minimum frequency, regardless of its intensity.
Threshold Frequency: The minimum frequency of light required to cause the photoelectric effect is known as the threshold frequency. Below this frequency, regardless of how intense the light is, no electrons are emitted.
Einstein's Explanation: Albert Einstein explained the photoelectric effect in 1905 by proposing that light consists of discrete packets of energy called photons. The energy of a photon is directly proportional to its frequency (E = hν), where E is energy, h is Planck's constant, and ν is frequency.
Work Function: The energy required to remove an electron from the surface of a material is called the work function. If the energy of a photon is greater than the work function, the electron is emitted.
The photoelectric effect has practical applications in devices such as photovoltaic cells (solar cells) where light is used to generate electric current. It also played a crucial role in the development of quantum mechanics, challenging classical wave theory and contributing to the understanding of the particle-like nature of light
threshold frequency (fT)
minimum frequency of light that causes ejection of electrons
energy of light
E = hf
h = 6.626 × 10-34
kinetic energy of ejected electrons
Kmax = hf - W
W = work function
work function
W = hfT
atomic absorption and emission
if a photon does not carry enough energy, then the electron cannot jump to a higher energy level. When an electron falls from a higher-energy level to a lower-energy level, a photon of light is emitted with an energy equal to the energy difference between the two orbits
nuclear binding energy
mass defect
concept in nuclear physics that refers to the difference between the mass of an atomic nucleus and the sum of the masses of its individual protons and neutrons. It is associated with the energy released during the formation of a nucleus.
Here are the key points about mass defect:
Binding Energy: The mass defect arises due to the binding energy that holds the protons and neutrons together in the nucleus. When nucleons (protons and neutrons) come together to form a nucleus, some of their mass is converted into energy according to Einstein's famous equation, E=mc².
Nuclear Binding Energy: The nuclear binding energy is the energy required to disassemble a nucleus into its individual protons and neutrons. This energy is equivalent to the mass defect.
Stability of Nuclei: Nuclei with higher binding energies (and thus larger mass defects) are more stable. This is because the energy required to break them apart is higher, making them less prone to spontaneous disintegration.
Mass Defect Calculation: The mass defect (Δm) is calculated as the difference between the mass of the nucleus (m_nucleus) and the sum of the masses of its individual protons (m_proton) and neutrons (m_neutron): Δm = m_proton (number of protons) + m_neutron (number of neutrons) - m_nucleus.
Units: Mass defect is typically expressed in atomic mass units (u) or MeV/c² (mega-electronvolts per speed of light squared).
Relation to Nuclear Reactions: In nuclear reactions, the mass defect is associated with the release or absorption of energy. During nuclear fusion, where lighter nuclei combine to form a heavier nucleus, mass is converted to energy, and the mass defect is manifested as the released energy.
Einstein’s equation
E = mc²
strong nuclear force
strong enough to compensate for the repulsive electromagnetic force between the protons
binding energy
allows the nucleons to bind together in the nucleus
weak nuclear force
contributes to the stability of the nucleus, but is about one-millionth as strong as the strong nuclear force
isotopic notation
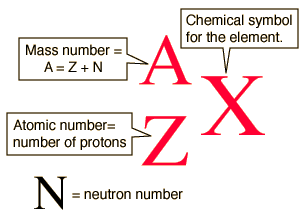
fusion
occurs when small nuclei combine to form a larger nucleus
fission
process by which a large nucleus splits into smaller nuclei
radioactive decay
naturally occurring spontaneous decay of certain nuclei accompanied by the emission of specific particles
isotopic decay arithmetic and nucleon conservation
XZA —> YZ’ A’ + emitted particle
when balancing nuclear reactions:
sum of atomic numbers must be the same on both sides of the equation
sum of mass numbers must be the same on both sides of the equation
alpha decay
emission of an α particle
verry massive compared to a beta particle and carries double the charge
interact with matter very easily; do not penetrate shielding very extensively
For Equation:
atomic number of daughter nucleus is two less than that of the parent nucleus
mass number of daughter nucleus is four less than that of the parent nucleus
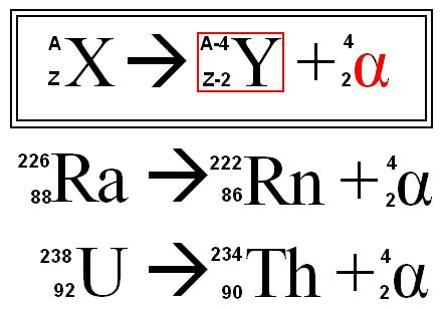
beta decay
emission of a β particle, which is an electron and is given the symbol of e- or β-
more penetrating than an alpha particle
in some cases of induced decay (positron emission), a positron is released, which has the mass of an electron but carries a positive charge
For Equation:
a neutron is converted into a proton and a β- particle (Z = -1, A = 0) is emitted. Hence, the atomic number of the daughter nucleus will be one higher than that of the parent nucleus
mass number will not change
During β+ decay, a proton is converted into a neutron and a β+ particle (Z = + 1, A = 0) is emitted. Hence the atomic number of the daughter cell will be one lower than that of the parent nucleus
mass number will not change

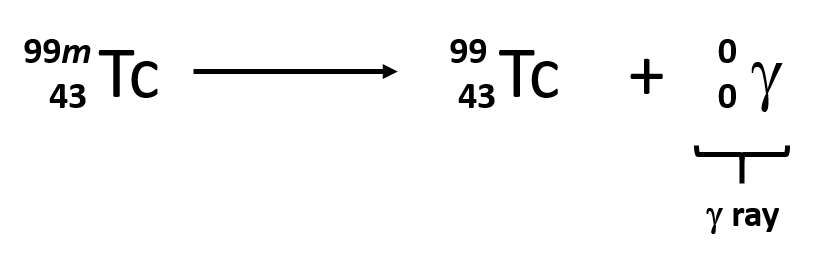
gamma decay
emission of γ rays, which are high-energy (high frequency) photons
γ rays carry no charge and simply lower the energy of the parent nucleus without changing the mass number or the atomic number
electron capture
certain unstable radionuclides are capable of capturing an inner electron that combines with a proton to form a neutron, while releasing a neutrino
For equation:
atomic number is one less than that of the parent nucleus
mass number remains the same
Equation:
XZA + e- —> YZ-1 A
Half life (T1/2)
time it takes for half of the sample to decay
exponential decay
Δn/Δt = -λn
n = # of radioactive nuclei that have not yet decayed in a sample
λ = decay constant
n = n0e^(-λt)
n0 = # of undecayed nuclei at t = 0
relationship between decay constant and half life
λ = ln(2) / (T1/2