GEN CHEM FLASHCARDS FOR EXAM 1
1/126
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
127 Terms
metal + nonmetal
metal nonmetal-ide
transition metal + nonmetal
transition metal(charge)nonmetal-ide
metal + polyatomic ion
Metal (charge) polyatomic ion
nonmetal +nonmetal
prefix- nonmetal nonmetal-suffix
acids
ate - ic acid
ite - ous acid
oxoacids
hydro - anion - ic acid
alkanes
prefix-ane
methane
CH4
ethane
C2H6
propane
C3H8
butane
C4H10
heptane
C7H16
hexane
C6H14
pentane
C5H12
octane
C8H18
hydrocarbons formula
C(number of carbons) H(2n+2)
energy of photon equation
wavelength equation
λ = hc / E
rydberg equation
1/λ = R(1/nf² - 1/ni²)
unit for wavelength (unless stated otherwise)
meters
energy equation
E = hv or E = hc/λ
kinetic energy
KE = ½mv² = hv - Φ
KE = Ephoton - work function
KE = (hc / λ ) - work function
energy change equation
En = -R(1/n²)
R value for Bohr’s equation
2.18E-18
ionization energy
(1/f² - 1/λR) = 1/n²
de Broglie equation
λ = h/p = h/mv
heisenberg’s uncertainty principle
it is impossible to know both momentum and position of a particle with absolute certainty
mass must always be measured in:
kg
velocity must always be measured in:
m/s
uncertainty equation
ΔxΔp ≥ h/4π
Δx - (change in uncertainty in position)
Δp - (change in uncertainty in momentum)
node
area where amplitude is at a minimum
schrodinger’s equation is used for:
radial probability density
n
principle quantum number
l
-angular momentum number (n-1)
-also number of angular nodes
magentic quantum number
ml
allowed values for ml
-l, -l+1, 0, l-1, l
s orbital corresponding value
0
p orbital corresponding value
1
d orbital corresponding value
2
f orbitalcorresponding value
3
radial nodes
n - l -1
s

px
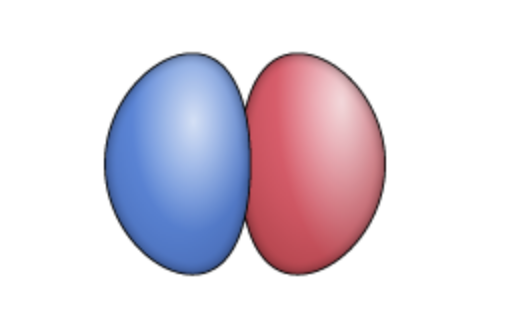
py
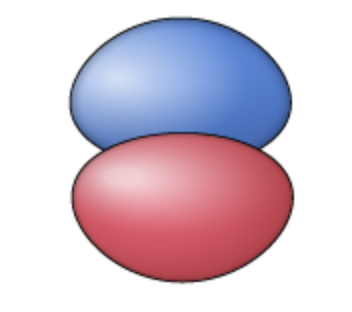
pz
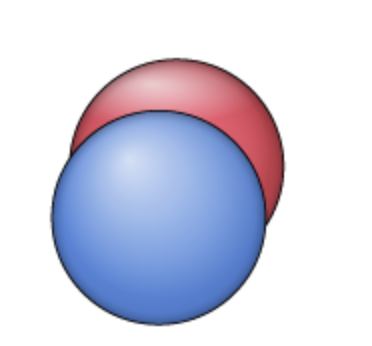
dxy
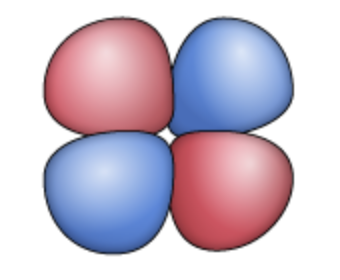
dxz
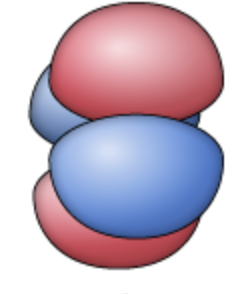
dyz
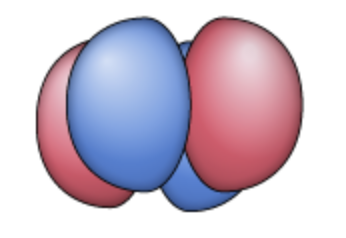
dx²-y²
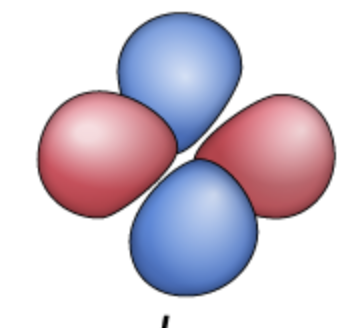
dz²
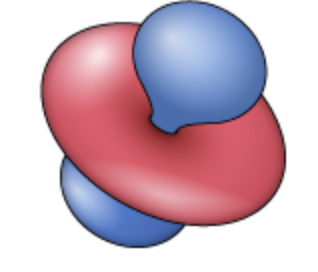
spin quantum number
ms
energy of hydrogen electron
En = -R(z²/n²)
electron configuration sequence
1s² 2s² 2p^6 3s² 3p^6 4s² 3d^10 5s²…
degeneracy
equivalency of energy
effective nuclear charge equation
atomic number - core electrons
core electrons
all electrons that are NOT in highest shell
valence electrons
all electrons in HIGHEST shell
how should an orbital diagram be filled
with all up arrows, and then down-facing pairs
what happens in an excited atom
the previous shell jumps to the highest level without filling its shell
Do iso-electronic atoms have the same effective nuclear energy?
No, they are all different, depending on the atom
What do iso-electronic atoms have in common
same number of valence electron
mono-
1
di-
2
tri-
3
tetra-
4
penta-
5
hexa-
6
hepta-
7
octa-
8
nona-
9
deca-
10
naming hydrated compounds
ionic compound prefix-hydrate
leading zeros
NEVER significant
trailing zeros
significant if there is a DECIMAL
trapped zeros
ALWAYS significant
accuracy
how close measurements are to true value
precision
refers to how close measurements are to each other
atoms to moles
divide by avogadro’s number
moles to atoms
multiply by avogadro’s number
density equation
mass / volume
moles to grams
multiply moles by molar mass
grams to moles
divide mass by molar mass
percent abundance equation
(x)(isotope A) + (1-x)(isotope B)
then multiply by 100 to get percentage
number of neutrons
atomic mass - atomic number
alkenes
contain one or more carbon-double bonds
alkynes
contain one or more triple bonds
mnemonic for remembering wavelength spectrum
Raging Martians Invaded Venus Using X-Ray Guns
max number of electrons
2n²
tera
10^12
giga
10^9
mega
10^6
kilo
10³
deci
10^-1
centi
10^-2
milli
10^-3
micro
10^-6
nano
10^-9
pico
10^-12
femto
10^-15
alkali metals
group 1