Lab Exam 2
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
A titration is best defined as an analytical technique for determining the
concentration of a specific solute in a solution
What color is the indicator used in today's exercise in acidic solutions?
Colorless
The equivalence point of a titration is when the indicator changes color and alerts you to stop.
False
How will you determine that you are approaching the end point of a titration?
The solution will change color and then return to the original color within 30 seconds with stirring
What TWO kinds of chemical reactions takes place between KHP and NaOH?
neutralization
double replacement
Which of the following is NOT a common active ingredient in commercial antacid tablets?
acetaminophen
Neutralization of stomach acid with an antacid produces a salt.
True
TWO functions of Benedict's solution make it useful for clinical diagnoses.
To react with sugars that can be oxidized
To produce a color change that can be detected visually or colorimetrically
The term renal glucose threshold means
Blood glucose level above which glucose appears in the urine
A disease that can be diagnosed by an elevated level of a sugar other than glucose in the blood or urine is
galactosemia
Fatty acids are termed unsaturated if they are less likely to contribute to cardiovascular disease and obesity.
False
Saponification involves
Hydrolysis of triglycerides using strong base
Formation of a carboxylate salt
Formation of a substance that allows emulsification
Soaps can lose their cleaning power in hard water due to precipitation of sodium salts of carboxylate ions.
False
two main functions of bile salts in the body.
Activation of enzymes that hydrolyze triglycerides
Emulsification of fats/oils in intestinal fluids
Benedict Reaction
Option C
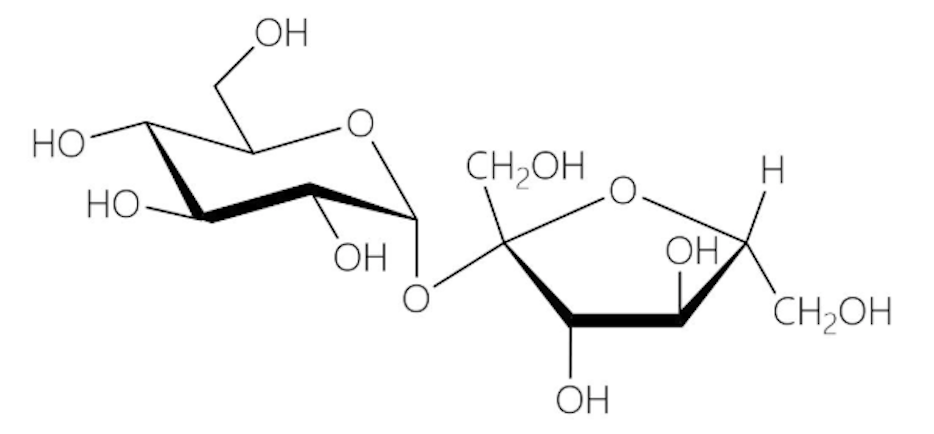
Sucrose
Comprise of alpha-D-glucose and beta-D-fructose molecule
Alpha 1 → beta 2 glycosidic linkage
Not a reducing sugar because the anomeric carbons of both sugars are forming an acetal
Sweetener, sugarcane, preservative
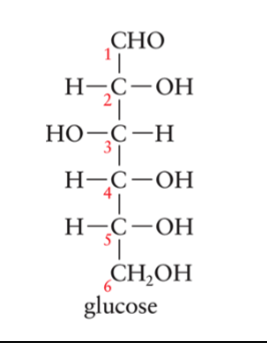
D-Glucose
Known as blood sugar since it’s transported by the blood to body tissues for energy requirements
Other sugars are absorbed by the liver and converted to glucose
Commonly used as sweetener, and baby foods
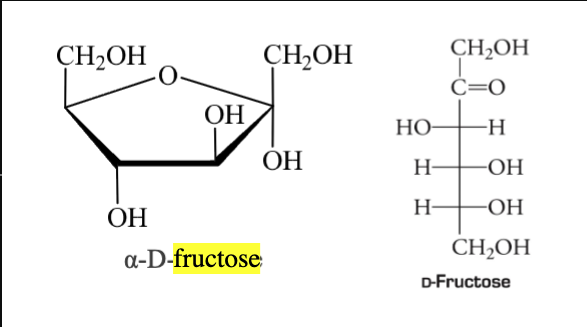
D-Fructose
Most important ketohexose
Known as levulose/fruit sugar due to being the sweetest monosaccharides and use as sweetener
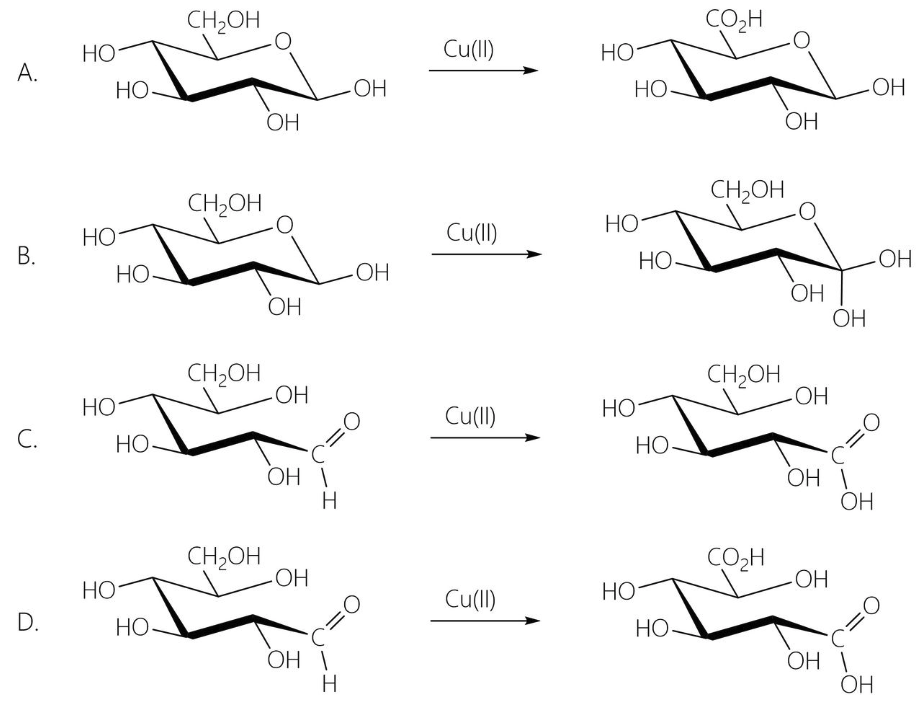
Reducing Sugars
Simple sugars that can be oxidized if they have:
Aldehyde
Ketone that can be isomerized to aldehyde in solution
Hemiacetal formed by reversible intramolecular condensation of an aldehyde and an alcohol
Benedict’s Reagent
Contain copper II sulfate (CuSO4)
Blue color due to presence of hydrated copper II ions (Cu+2)
Reducing sugar oxidized, and the Cu+2 reduced to Cu+ forming bright orange-red copper I oxide (Cu2O)
Semiqualitatively test since color is related to the glucose concentration
Test for reducing sugars
A non-reducing sugar can give a positive Benedict’s test if it is first hydrolyzed into reducing sugars
Xanthoproteic Test
Amino acid will only test positive if the aromatic ring bears amino or hydroxyl groups
Albumin
The protein fraction that shows the largest, darkest band
Arrhenius Acid & Base
Only apply in aqueous solution, H+ & OH- are involved
Acid: provide H+ (proton) when dissolved in water (aqueous solution)
Base: provide OH- when dissolved in water (aqueous solution)
Bronsted-Lowry Acid & Base
Proton transferring, and apply to non-aqueous solution
Acid: hydrogen-containing substance that donate H+ to another substance
Base: any substance that can accept H+ from another substance
Lewis Acid & Base
Electron transferring, the most inclusive theory
Acid: substance accepts a pair of electron
Base: substance provides a pair of electron
Ex: binding between a metal ion and ligand (molecule/ion that donates electron pairs to central method ion to form coordinate bond)
Hydrolysis
Chemical reaction where a bond is broken by water
May produce acidic or basic solution depending on the reaction
Ex: NH4 + H2O → NH3 + H3O
Amphiprotic/Amphoteric
A substance that can act as both acid or base depending on its reacting partner. Essential for life and biochemical reactions
Ex: water, HCO3-, H2PO4-
Buffer
Consists of weak acid and weak conjugate base
Buffer capacity: amount of acid (H+) or base (OH-) that can be absorbed without causing significant change in pH
Respiratory and Kidney Physiological
Respiratory Acidosis
pH ↓
PCO₂ ↑
HCO₃⁻ ↑ (renal compensation, if chronic)
Respiratory Alkalosis
pH ↑
PCO₂ ↓
HCO₃⁻ ↓ (renal compensation, if chronic)
Metabolic Acidosis
pH ↓
HCO₃⁻ ↓
PCO₂ ↓ (respiratory compensation: hyperventilation)
Metabolic Alkalosis
pH ↑
HCO₃⁻ ↑
PCO₂ ↑ (respiratory compensation: hypoventilation)
Titrant/Standard
Is the base in the experiment, using buret to add drop-wise until endpoint is reached
Known concentration and volume
Equivalence Point
Theoretical point of completion
Ma * Va = Mb * Vb
Can also be obtained using pH meter during titration, inflection point (steepest slope on the curve)
mmol = M * V (unit is ml)
Endpoint
Measured based on the presence of indication
The difference between theoretical and actual is negligible, and refers to as indicator error
Back-Titration
Adding excess titrant to analyte solution (initial titrant)
React leftover titrant with second standard to calculate leftover titrant
Find used titrant by subtracting initial with leftover, then calculate the analyte
Useful when first titration is slow, or when analyte is non-soluble solid as in the case of antacid titration
Roles of HCl in Stomach
Combine with precursor pepsinogen to activate pepsin enzyme
Denature (unfold) proteins in order to efficiently promote degradation by enzymes
Provide pH for pepsin to break down proteins molecules efficiently
Phenolthalein
Color indication used in titration
Colorless when <8.2, and pink if higher
pH transition overlaps steep pH change near equivalence point
Glucose Reference Range
80-110 mg/dL
Diabetes Mellitus
Metabolic disorder that lead to hyperglycemia
Type I diabetes: destruction of beta cell of pancreas, little to no insulin production
Type II diabetes: insulin resistance in the body even if there are adequate amounts
Symptoms include excessive thirst, frequent urination, hunger, slow healing
Galactosemia
Genetic disease in which enzyme for galactose is deficient
Can result in mental retardation and permanent physical impairment
Diagnosed by positive Clinitest and negative glucose test strip
Glucosaria
Kidney cannot completely reabsorbed sugar, and so there are glucose in the urine
Hormonal disorders such as Cushing’s syndrome, hyperthyroidism
Fanconi’s syndrome: abnormal renal tubular reabsorption due to genetic defect or damage to kidneys by drugs or heavy metal
Renal glycosuria: renal threshold for glucose to be extremely low
Clinitest Tablet
Contains copper sulfate (CuSO4), sodium carbonate (Na2CO3), sodium citrate, and sodium hydroxide
Test for reducing sugars
Negative result: blue
Positive result: green, orange
Reagent Test Strips
Contains glucose oxidase that specifically oxidizes glucose to gluconic acid and hydrogen peroxide
Contains peroxidase to catalyze hydrogen peroxide to change the color
Positive result: color change
Negative: no color change
Lipids
Low polarity, insoluble in water
Soluble in nonpolar solvents such as membranes
Triglycerides are the most abundant. Recommend 25-35% of TG intake
Chromatography
Measure the middle point of the spot
Rf factor: solventspot
Biuret Test
Test positive when there are ≥ 2 peptides bonds
The lone pair of nitrogen in peptide bonds form covalent bond with copper (II) ion to change color to purple
Lead Acetate Test
Test for presence of sulfur-containing amino acids
Positive: form insoluble black precipitate
Xanthoproteic Test
Test for proteins that contain aromatic amino acids
Positive in the formation of