3.3.9.2 acylation
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
carboxylic acids be turned into… to be used in…
into other functional groups
to be used in organic synthesis
give two groups that can be formed from carboxylic acids
acyl chlorides
acid anhydrides
→ both called acid derivatives
what are acyl chlorides?
where the -OH group of the acid is replaced by a chlorine atom

how are acyl chlorides named?
they are named after the corresponding carboxylic acids → the -oate is replaced by -oyl
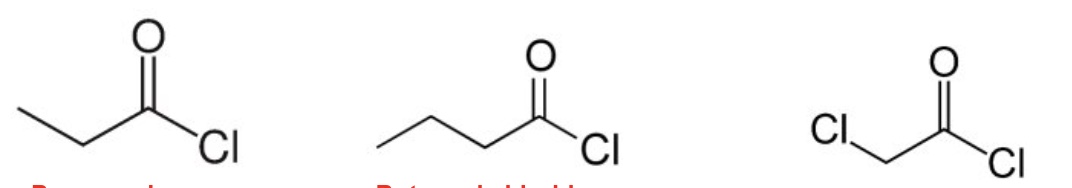
name the following acyl chlorides

what does this carboxylic acid derivative (acyl chlorides) possess?
a good leaving group (Cl-)
what does this good leaving group (Cl-) activate?
it activates the adjacent carbonyl group by electron withdrawal
what is a good leaving group?
its a stable species which is removed during a chemical reaction
what is the by-product of when acyl chlorides react?
HCl
what is the hazard of HCl?
it is corrosive
how do acyl chlorides + acid anhydrides react with nucleophiles?
in a reduction reaction
what mechanism do acyl chlorides + acid anhydrides undergo?
nucleophilic addition elimination
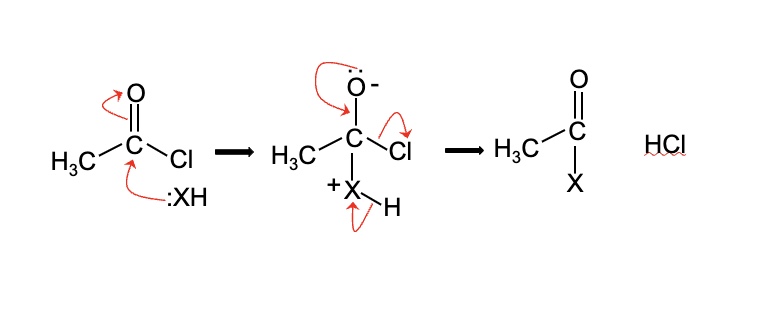
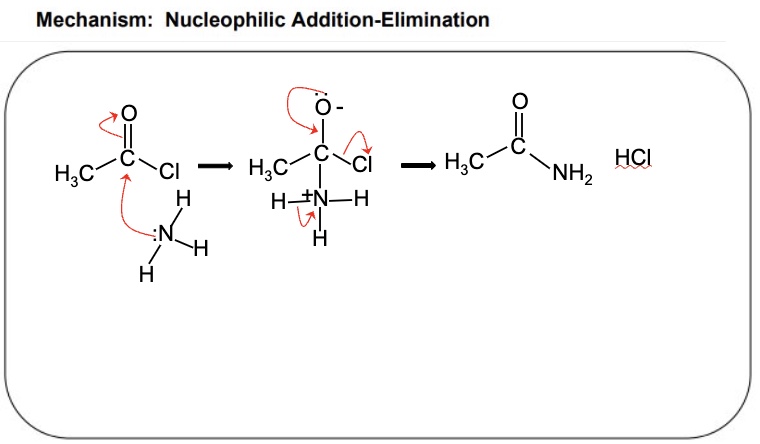
describe the nucleophilic addition elimination mechanism that acyl chlorides undergo
in the first step, the nucleophilic adds to the ᵟ⁺ carbon of the carbonyl causing electrons to move from the carbonyl bond onto oxygen forming an oxyanion
in the second step, the carbonyl bond is remade causing the elimination of the leaving group on the acyl chloride (Cl-) or acid anhydride
finally the nucleophile loses a proton (H) + the by-product is made
why does the carbon atom have a greatest chance of being readily attacked by nucleophiles?
because the chlorine + oxygen are more electronegative than the carbon atom
give the general equation + mechanism using the example of ethanoyl chloride
CH₃COCl + :XH → CH₃COX + HCl
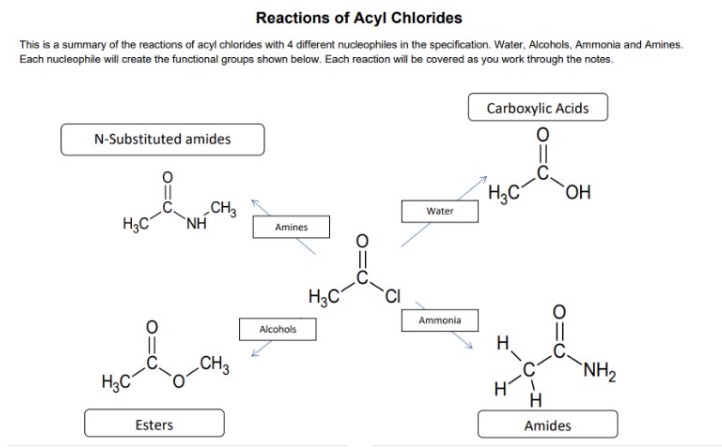
what are the 4 common nucleophiles that react with acyl chlorides?
H₂O
NH₃
ROH
RNH₂
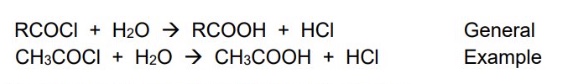
one way of forming carboxylic acids is by the oxidation of primary alcohols or aldehydes by acidified potassium dichromate, but give another way of forming them with acyl chlorides
acyl chlorides react with water to form carboxylic acids
give the general symbol equation for the formation of carboxylic acids using acyl chlorides + give an example one using ethanoyl chloride
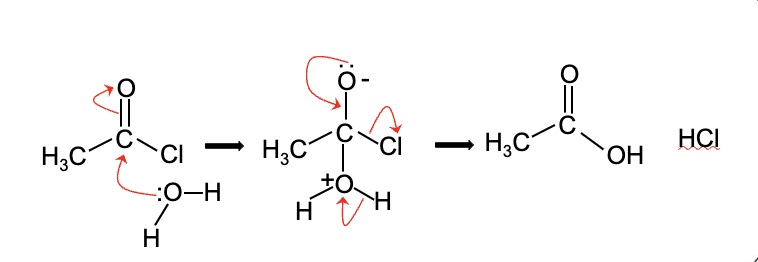
draw + name the mechanism for the reaction between ethanoyl chloride and water
mechanism: nucleophilic addition elimination
one way of forming esters is by esterification, outline another way of forming esters using acyl chlorides
acyl chlorides react with alcohols to form esters → they undergo nucleophilic addition elimination
give the general equation for the formation fo esters from acyl chlorides
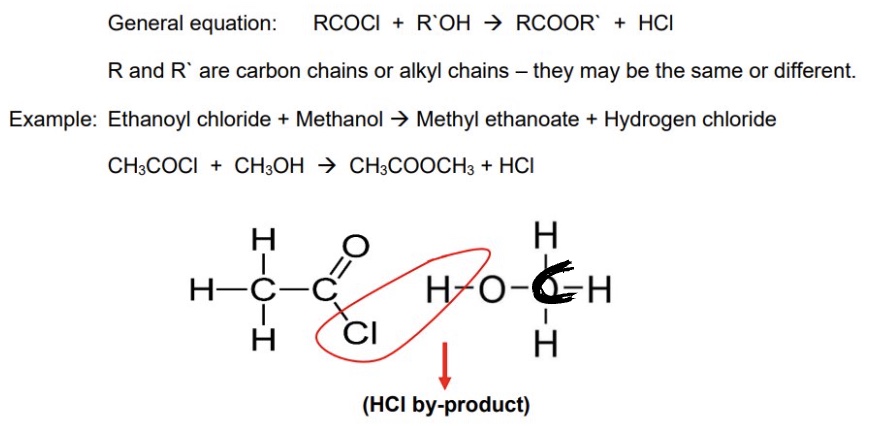
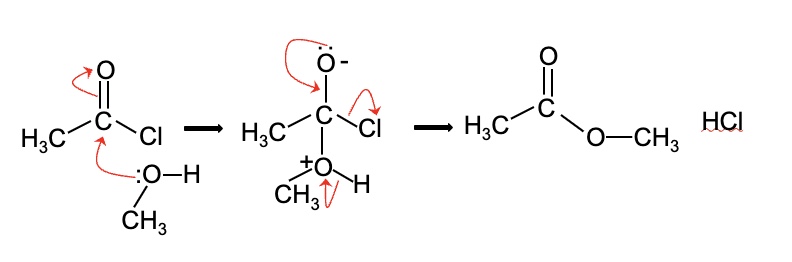
draw the mechanism for the reaction between ethanoyl chloride + methanol
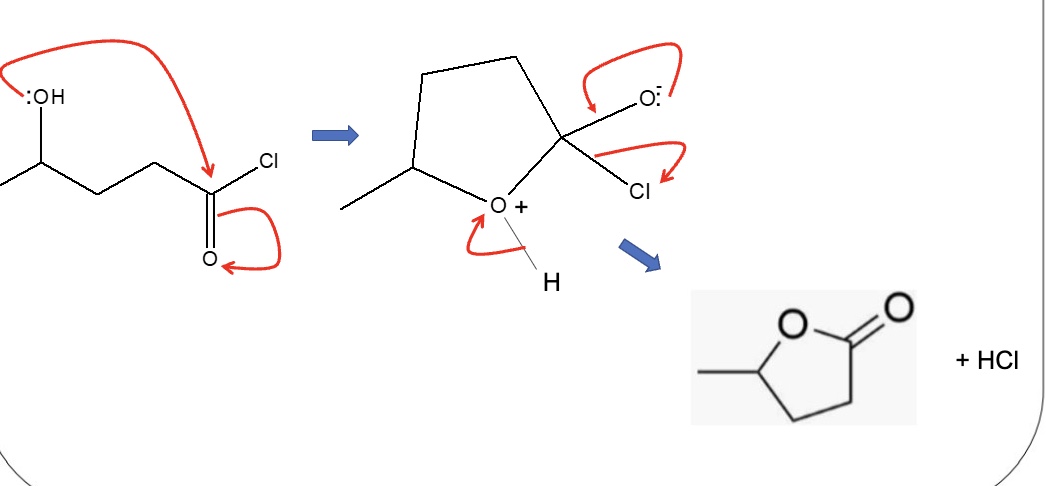
give 3 reasons why acyl chlorides may be preferred to carboxylic acids in the formation of esters
no acid catalyst is required (esterification uses H₂SO₄)
purer product
yield of ester is higher as the reaction is not reversible (whereas esterification is)

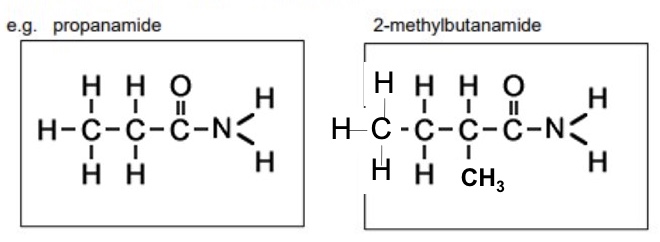
give the general displayed formula for amides
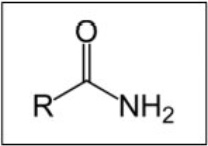
how are amides named?
using the suffix -anamide
how are amides formed?
when using ammonia (NH₃) as the nucleophile
eg draw propanamide and 2-methylbutanamide
acyl chlorides react with ammonia to form what?
amides
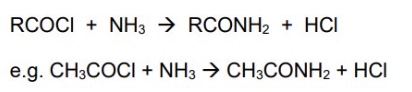
give the general symbol equation for the reaction between acyl chlorides + ammonia
give an example using ethanoyl chloride
name + draw the mechanism between acyl chlorides + ammonia using the example of ethanoyl chloride

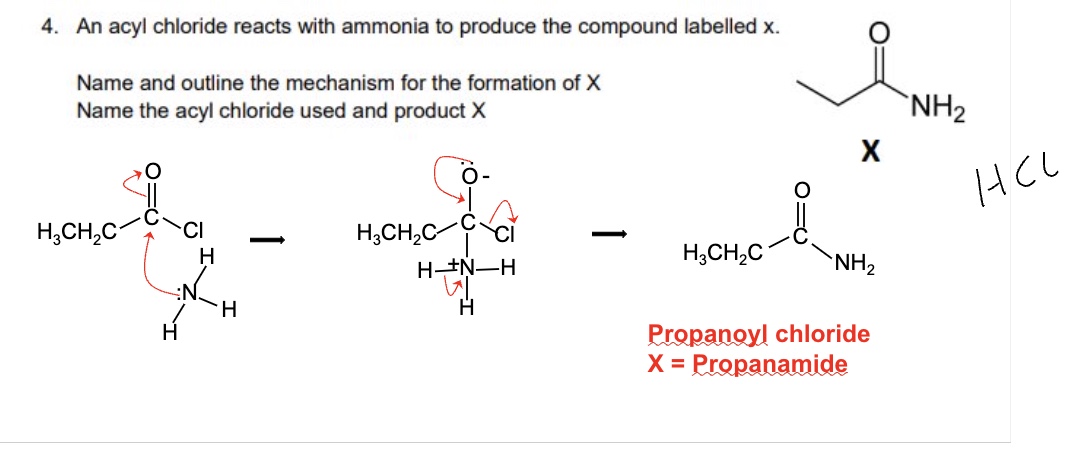

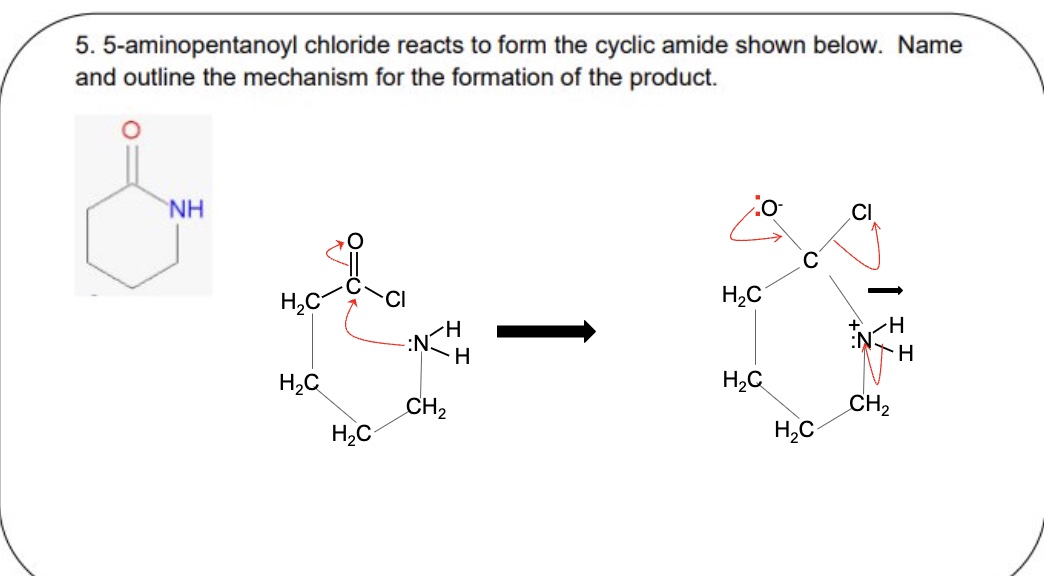
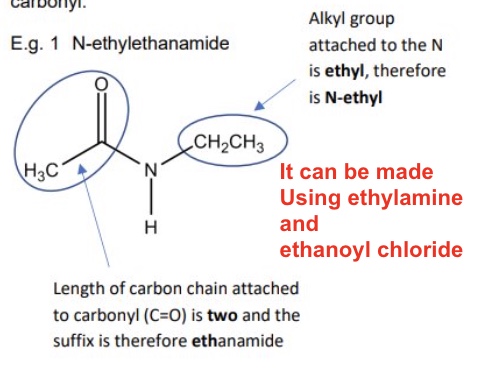
outline how N-substitued amines are named
using an N-alkyl prefix to indicate the alkyl group attached to the N only
and the suffix -anamide to indicate the length of carbon chain attached to the carbonyl
example in image
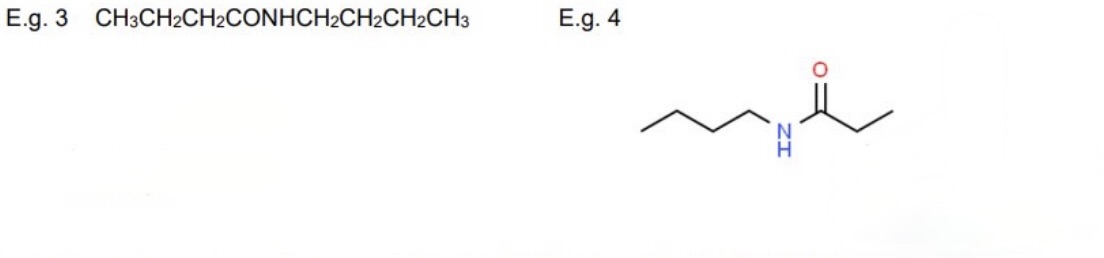
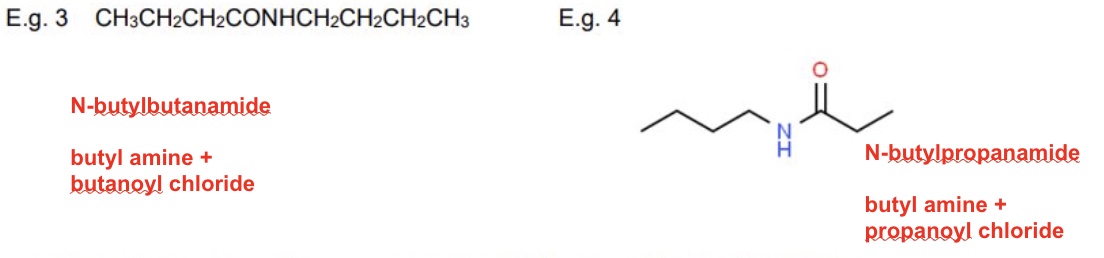
for these examples, name them + identify the reagents used used to form them
what are the two ways of naming amines?
either as alkyl amine or aminoalkane
eg ethyl amine or aminoethane
eg propyl amine or aminopropane
acyl chlorides react with amines to form what?
N-Substituted amides
give the general symbol equation for the reaction between acyl chlorides + amines
give an example using ethanoyl chloride
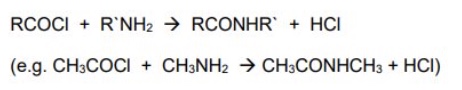
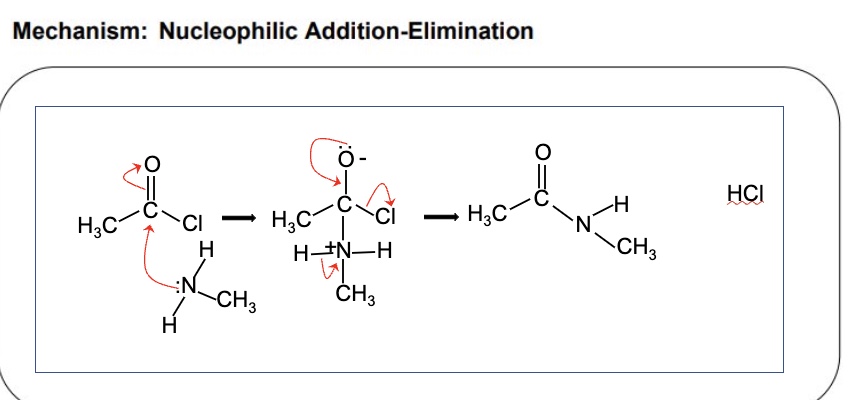
name + draw the mechanism for the reaction between acyl chlorides + amines using the example of ethanoyl chloride
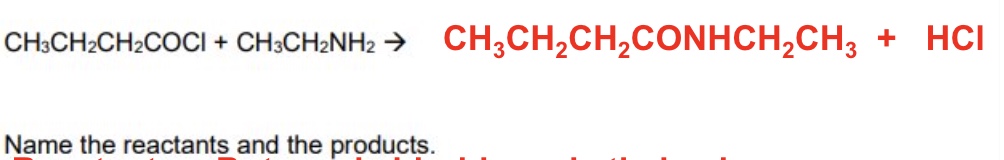


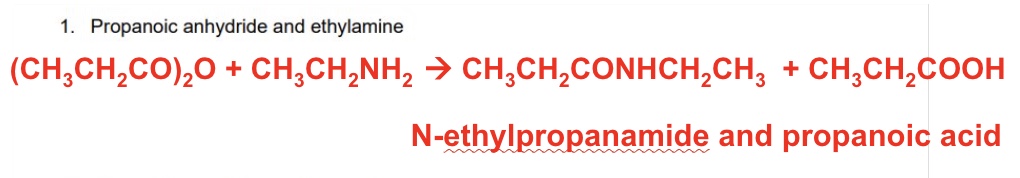
reactants: propanol chloride + diethylamine
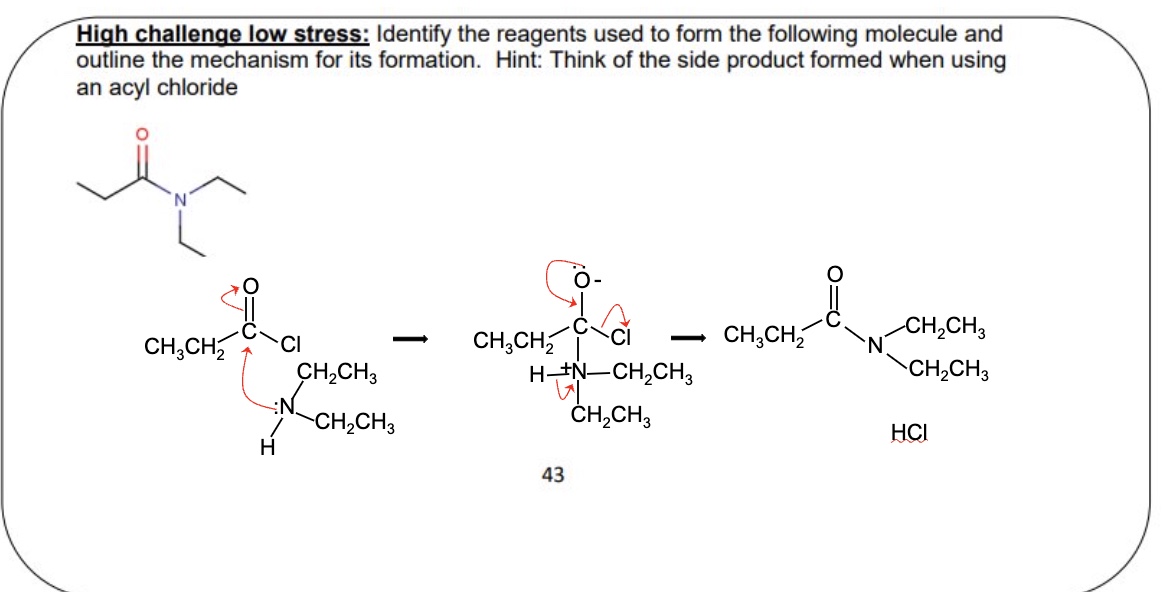
give the other carboxylic acid derivative
acid anhydrides
give four reasons why acid anhydrides are preferred to acyl chlorides in the formation of esters
acid anhydrides are cheaper
acid anhydrides react less exothermically meaning the reaction is less violent/dangerous
acid anhydrides are less vulnerable to hydrolysis
no corrosive HCl is formed
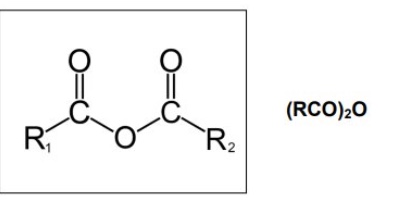
what are acid anhydrides?
another carboxylic acid derivative where -OH group of carboxylic acid is replaced by —OCOR
give the general displayed + structural formula for acid anhydrides
(this structural formula is only used for symmetrical acid anhydrides)
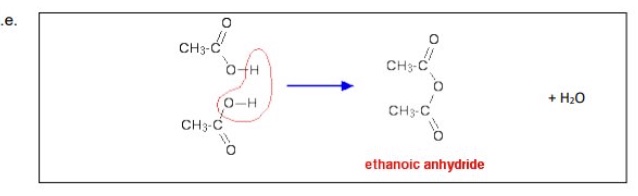
when are acid anhydrides formed?
when two molecules of carboxylic acids join together with elimination of water
outline how acid anhydrides are named
it is named after the carboxylic acid that was used in its synthesis
the anhydride suffix -anhydride replaces the -acid suffix at the end of the carboxylic acid name
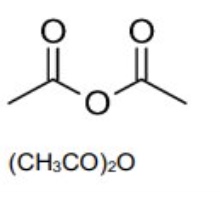
eg name this acid anhydride
ethanoic anhydride

name these molecules + give the structural formula for 1
propanoic anhydride, (CH₃CH₂CO)₂O
ethanoic propanoic anhydride

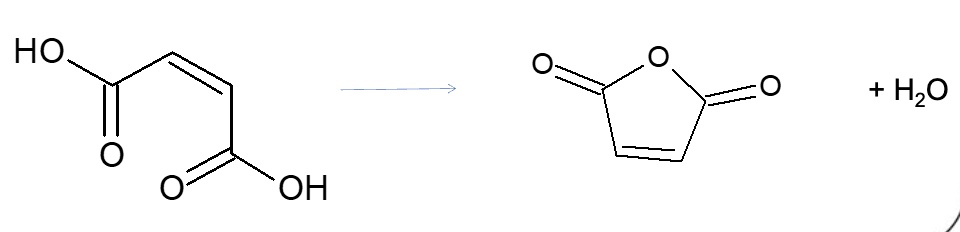
what is the mechanism for reactions of acid anhydrides?
nucleophilic addition elimination
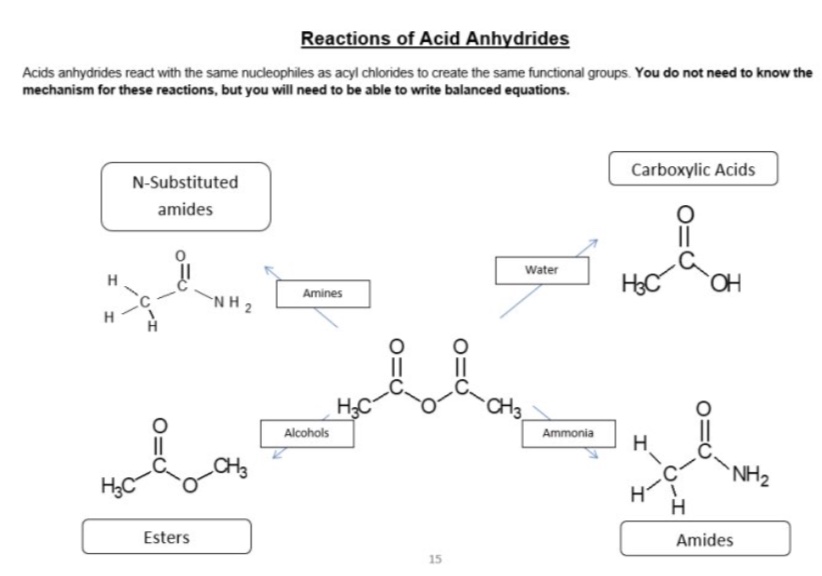
what do acid anhydrides possess?
a good leaving group
define what a good leaving group is + give the one of acid anhydrides
a good leaving group is a stable species, which is removed during a chemical reaction
RCOO-
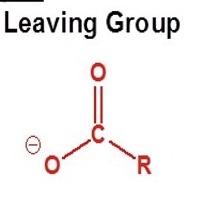
what does the good leaving group (RCOO-) do?
it activates the adjacent carbonyl group by electron withdrawal
outline the by-product of when acid anhydrides react (by nucleophilic addition elimination)
acid anhydrides react to form the corresponding carboxylic acid as a by-product
outline the similarity + difference between the reactions of acyl chlorides + acid anhydrides
they react in the same way to produce the same carboxylic acid
but will produce a different side product

we have already seen 2 ways of forming carboxylic acids: oxidation of primary alcohols or aldehydes + acyl chlorides react with water
outline the third way of forming carboxylic acids
Cos can also be prepared using acid anhydrides
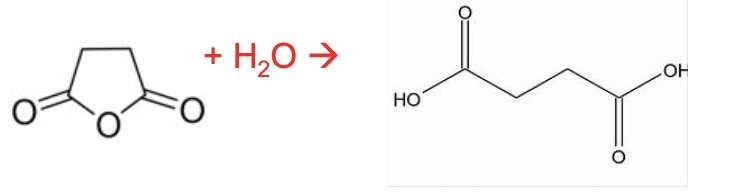
acid anhydrides react with water to form carboxylic acids
if it is a symmetrical acid anhydride, then 2 of the same CA is formed
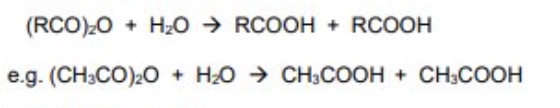
give the equation using skeletal formula for the reaction between ethanoic anhydride + water
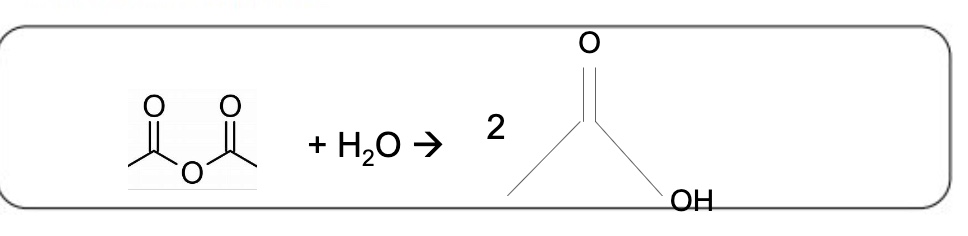
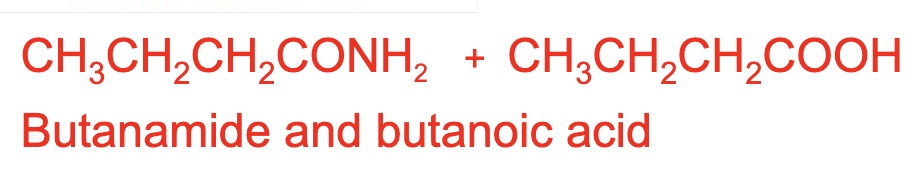
write an equation for the reaction between propanoic anhydride + water
should cancel out to 2 CH₃CH₂COOH

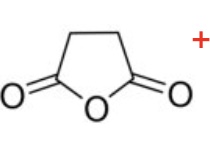
write an equation for the reaction of the acid anhydride shown in the image with water
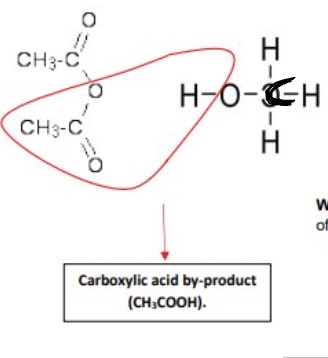
acid anhydrides react with alcohols to form what? give the mechanism for this
esters
nucleophilic addition elimination
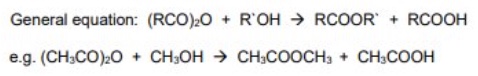
give the general symbol equation for the reaction between acid anhydrides + alcohol
give an example using ethanoic anhydride + methanol




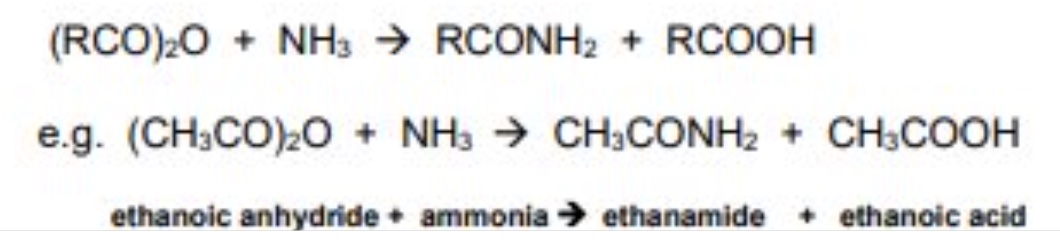
outline the reaction between acid anhydrides + ammonia
acid anhydrides react with ammonia to form amides (+ carboxylic acid as a by-product)
mechanism is nucleophilic addition elimination
give the general symbol equation for the reaction between acid anhydrides + ammonia
give an example using ethanoic anhydride

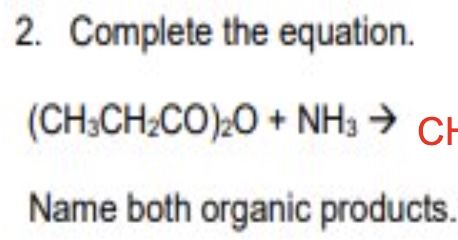

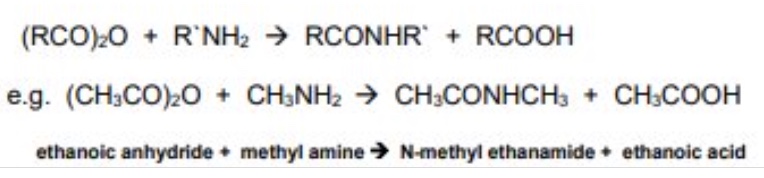
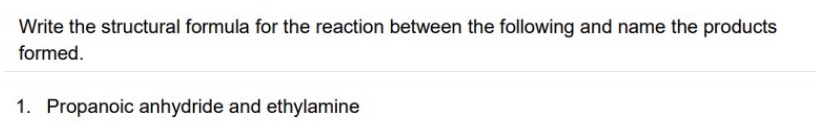
outline the reaction between acid anhydrides + amines
acid anhydrides react with amines to form N-substituted amides
mechanism is nucleophilic addition elimination
give the general symbol equation for the reaction between acid anhydrides + amines
give an example using ethanoic anhydride