Carbohydrates and Lipids (not finished)
1/40
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
what is the monomer of carbohydrates?
monosaccharides
what is the monomer of lipids?
fatty acids, glycerol, phosphate groups
what is the monomer of proteins?
amino acids
what is the monomer of nucleic acids?
nucleotides
how are macromolecules formed?
condensation reactions → molecules combine together forming covalent bonds and resulting in polymers, water removed
how are polysaccharides formed?
when two hydroxyl (OH) groups on different monosaccharides interact to form a (covalent) glycosidic bond
how are polypeptides formed?
by condensation reactions where two amino acid monomers interact to form a strong covalent peptide bond
how are nucleic acids formed?
separate nucleotides are joined by condensation reactions to form a phosphodiester bond
occur between the phosphate group of one nucleotide and the pentose sugar of the next
what is the name of the reaction that breaks macromolecules into their monomers?
hydrolysis (breaking with water)
what is the general formula of monosaccharides?
CnH2nOn
what are the properties of monosaccharides?
colourless crystalline molecules
soluble in water
what is the formula of glucose?
C6H12O6
what is the difference between alpha glucose and beta glucose?
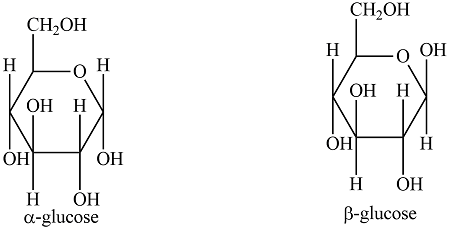
what polysaccharides are formed from alpha glucose?
starch and glycogen
what polysaccharide is formed from beta glucose?
cellulose
what are the properties of glucose?
stable structure due to strong covalent bonds
soluble in water as it is polar
soluble in water (easily transported)
source of chemical energy when bonds are broken
properties of carbohydrates
compact → large quantities stored in a small space
insoluble
useful structurally as strong and durable, insoluble and slightly elastic, chemically inert → eg cellulose
what is the structure of amylose?
unbranched helix-shaped chain with 1,4 glycosidic bonds between a glucose molecules
helix shape = compact = resistant to digestion
what is the structure of amylopectin?
1,4 glycosidic bonds between a glucose molecules as well as 1,6 glycosidic bonds = branched
branches result in many terminal glucose molecules that are easily hydrolysed for use
what is the structure of glycogen?
a glucose joined by 1,4 and 1,6 glycosidic bonds
more branched than amylopectin = more free ends where glucose can be hydrolysed
means glucose can be broken down quickly
what is the structure of cellulose?
can be straight and unbranched
polymer of b glucose monomers
every alternate molecule of b glucose must flip
alternating pattern allows hydrogen bonding adding strength
what are microfibrils?
linking of several molecules of cellulose through hydrogen bonds
how are glycoproteins formed?
covalent bonds between carbohydrates and polypeptides
what is the function of glycoproteins?
receptors in cell recognition and identification
receptors for cell signalling molecules (like hormones and neurotransmitters)
endocytosis
cell adhesion and stabilisation
how do glycoproteins link to ABO blood types?
glycoproteins act as antigens that identify cells as self or non self
cells recognised as non self trigger immune responses
which glycoprotein antigens correspond to which blood type?
type A → type A glycoprotein antigens
type B → type B
type AB → both types
type O → neither
what are some examples of lipids in living organisms?
fats, oils, waxes, steroids
what do saturated fatty acids contain?
only single carbon-carbon bonds
what do unsaturated fatty acids contain?
one or more carbon-carbon double bond
how are triglycerides formed?
esterification
ester bond formed when hydroxyl group of a glycerol molecule bonds with the carboxyl group of a fatty acid
ester bond is a condensation reaction
how are phospholipids formed?
glycerol and fatty acids
unlike triglycerides they only have two fatty acids bonded to a glycerol molecule
the third has been replaced with a phosphate ion
what are the properties of phospholipids?
phosphate is polar, soluble in water (hydrophilic)
fatty acid tails are nonpolar, insoluble in water (hydrophobic)
amphipathic → hydrophilic and hydrophobic regions
what is released from the hydrolysis of triglycerides?
glycerol and fatty acids
what are lipids (in terms of energy)?
energy dense due to high number of C-H bonds
what is produced when lipids are respired?
a lot of water compared to carbohydrates → metabolic water and can be used as a dietary water source in the absence of drinking water
camel’s hump filled with lipids that provide metabolic water
bird’s egg is filled with lipids to provide energy and metabolic water
where are lipids stored in animals?
adipose tissue
subcutaneous fats below the skin
visceral fats around major organs
what are the properties of saturated fatty acids?
only single carbon-carbon bonds
‘saturated’ with hydrogen
straight molecules so lipids can pack tightly together
increases their melting point causes them to be solid at room temp
what are the properties of unsaturated fatty acids?
not all the carbon-carbon bonds are single bonds
‘unsaturated’ with hydrogen
double bonds cause kinks in the structure so they cannot pack as tightly together
often liquid at room temp
what is a monounsaturated fatty acid?
a fatty acid with one C=C double bond
what are polyunsaturated fatty acids?
a fatty acid with many C=C double bonds