ch.8 flash cards (reactions)
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
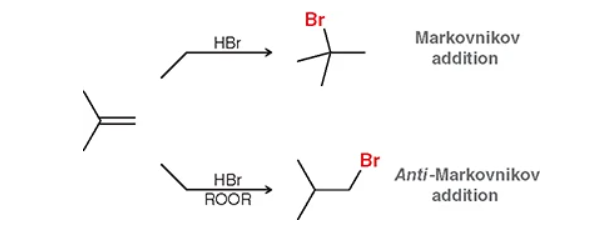
what type of reaction uses H-X and/or peroxides? how would you solve?
hydrohalogenation — you would follow the general trend of identifying the substrate (X = Br, Cl, I, etc.) and determine sterics (more crowed or anti = less crowed carbon.) Quick trick: are you single? —> if there a peroxide with the HBr youre taken, get away from the hoes (carbons). No peroxide (no hoes?) = get some (carbons)
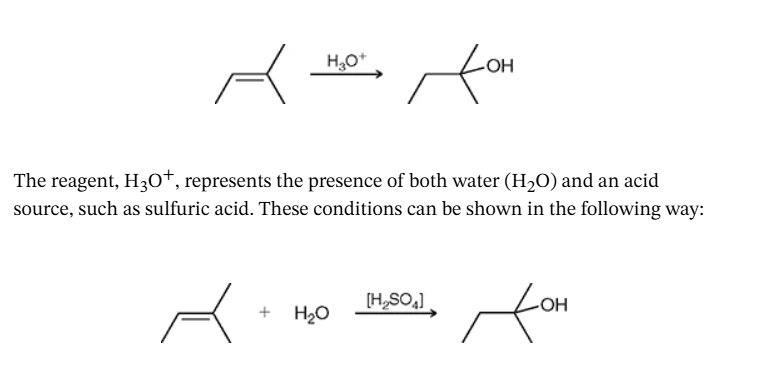
what would you identify this reaction as? key factors: acid and water
acid catalyzed hydration —> tips: look for an acid that isn’t connected to a halide AND the addition of water or brackets on acid. This will always result in the addition of H and OH and they wanna be where the party is at. (mark)
- remember P.A.D: create a carbocation → water will attack → remove an H from the water to create an OH group
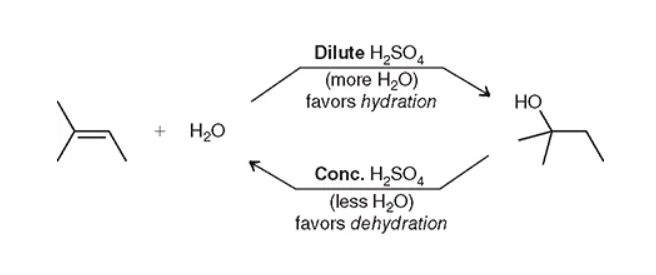
what is the difference between concentrated vs diluted acid?
diluted has water and will result in hydration, concentrated will result is the dehydration reaction
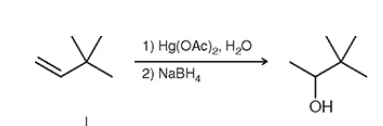
what the type of reaction is this? hints: look at Hg and NaBH4
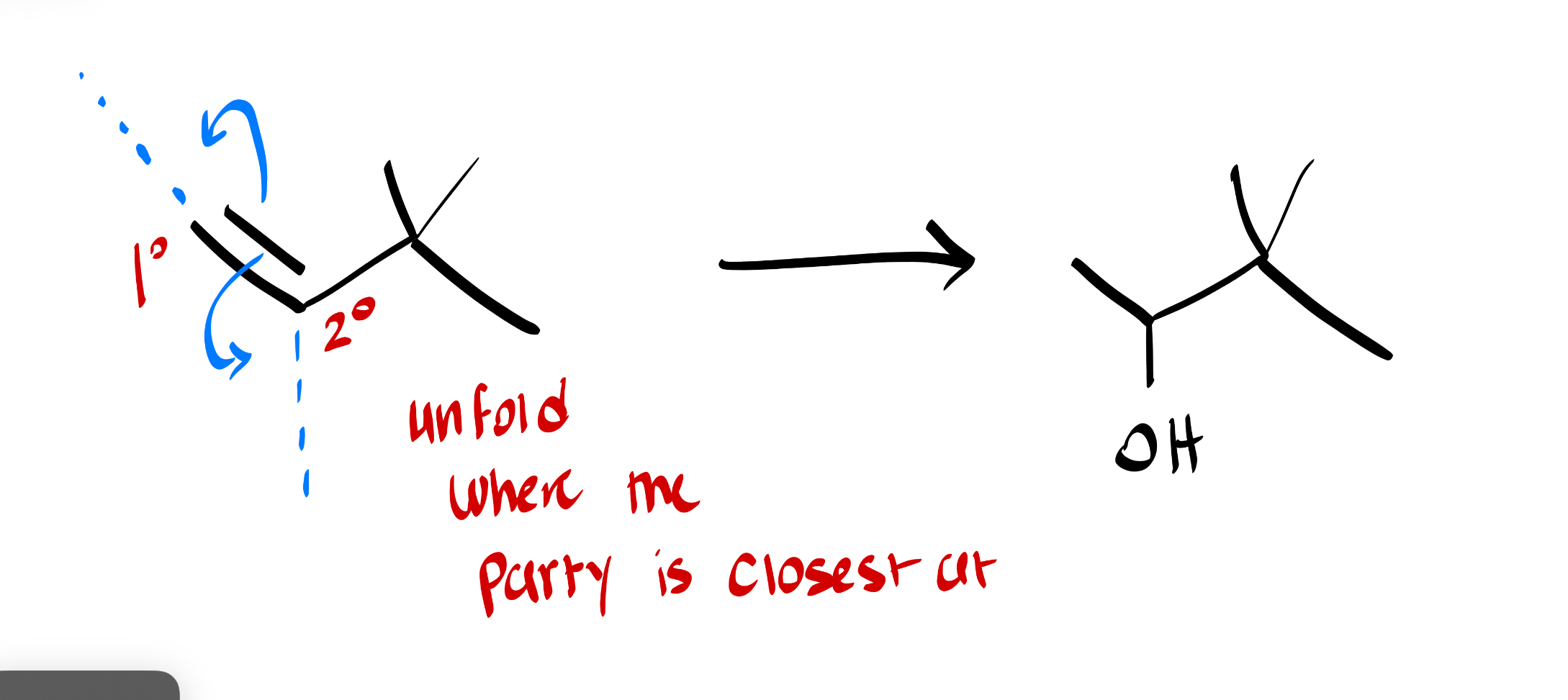
this is a oxymercuration-demercuration reaction. (mark) very fast way to solve: place the OH group where the party is closest at. no shifts will happen he is happy where he’s at. (think of unfolding left or right only.)
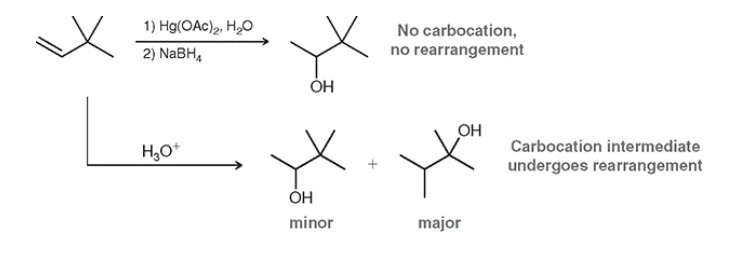
what is the difference between mercury-demercury and an acid catalyzed reaction
rearrangements —> most acid-catalyzed will be a 1,2 hydride shift or 1,2 methyl shift. fast way to memorize: move to where is party is most full (tert)
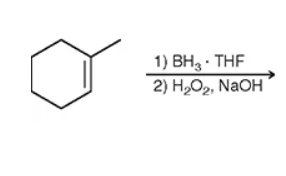
huh? what?
hydroboration-oxidation: will be anti and add an H and OH (anti = anti social = less carbon —> OH will most likely be the end carbon & H will be added adjacently).
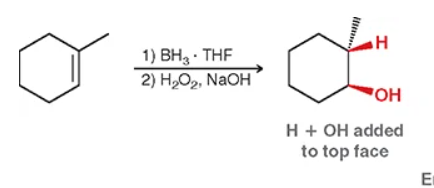
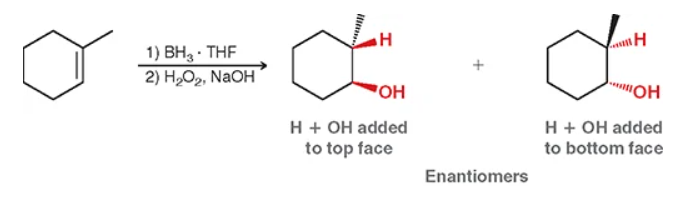
for hydroboration-oxidation what does syn mean?
same side with wedges or dashes (ex: OH and H will be on the same timing) quick tip: if you spot TWO chiral centers it WILL have syn. (use En to abbreviate).
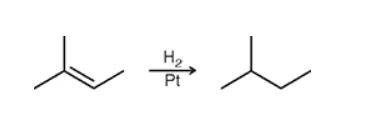
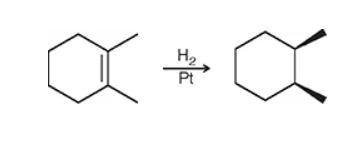
hint: it causes an alkene → alkane
catalytic hydrogenation: remove the double bond and call it a day
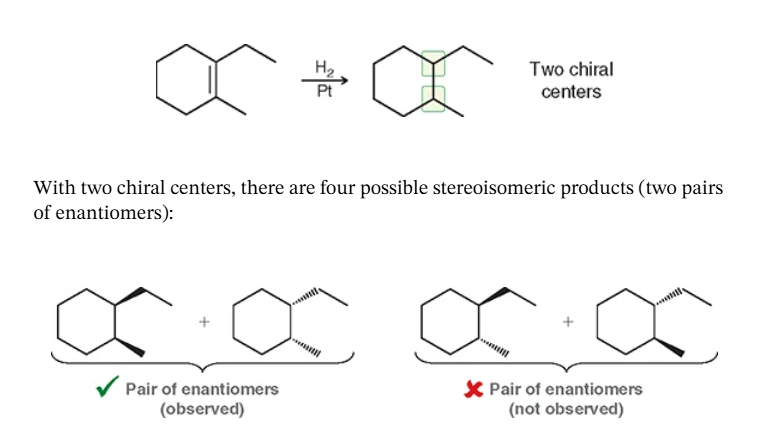
for catalytic hydrogenation what causes En vs Meso?
if a chiral center is created & symmetry —> if there is symmetry there will be no En’s if there is asymmetry there will En’s
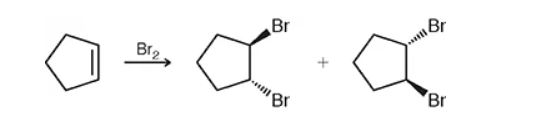
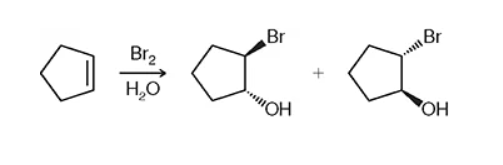
halogenation vs halohydrin formation
halogenation will be just a halide x2 (ex: Br2) while halohydrin will include water and sub in the halide and OH. wedges and dashes will be opposite of each other and one of each.
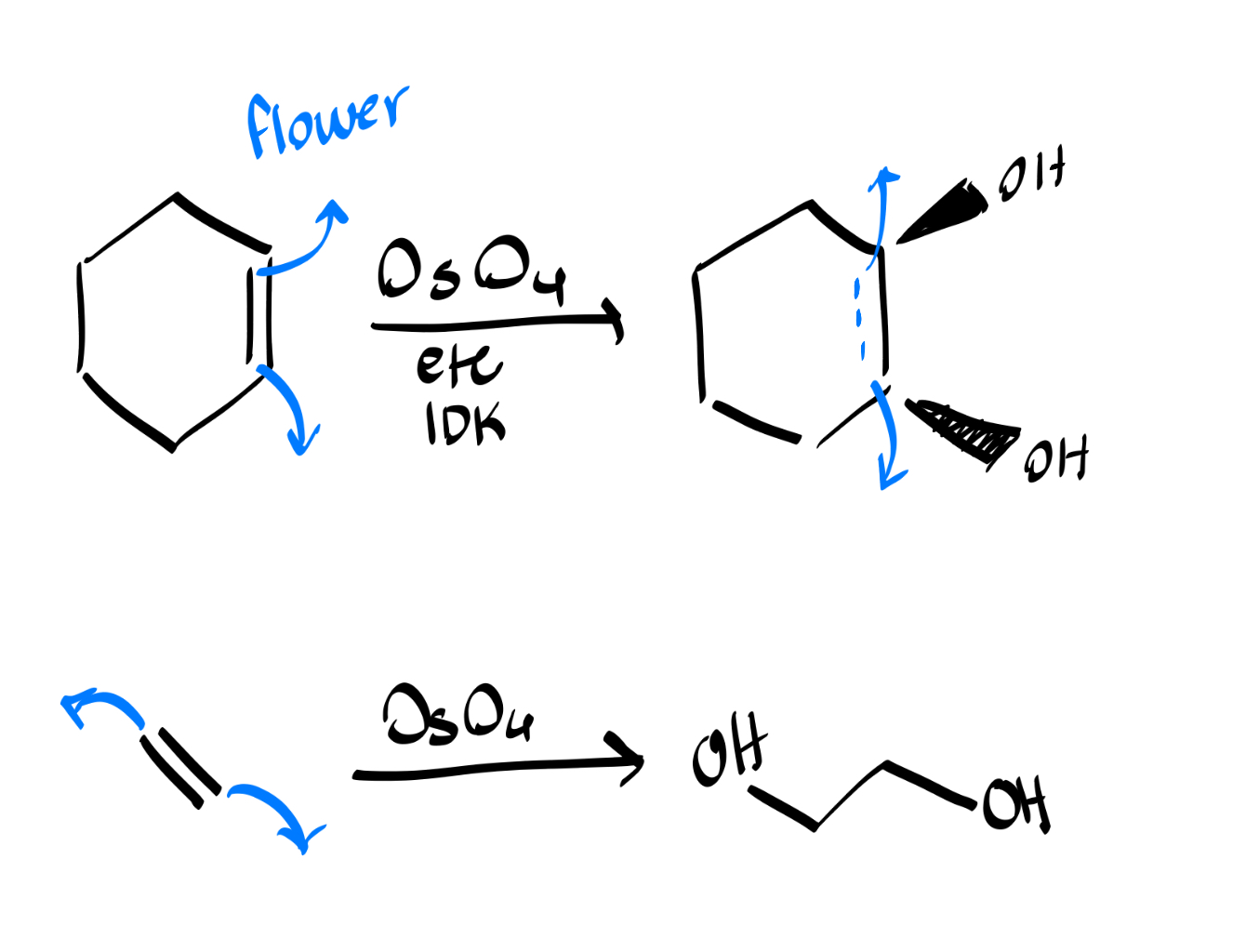
syn dehydroxylation?
will always use OsO4: the reaction is like a flower blooming outwards with OH wedges
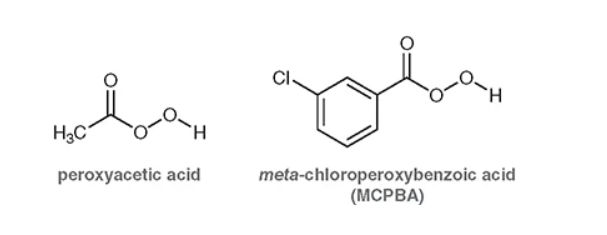
these are known as peroxy acids —> what do they do?
they will create an epoxide (inwards flower with wedges from the double bond to an O) and with the addition of an acid after create an outwards flower with wedge and dash OH.
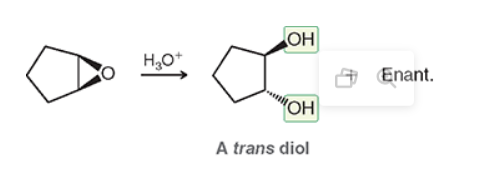
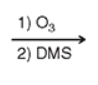
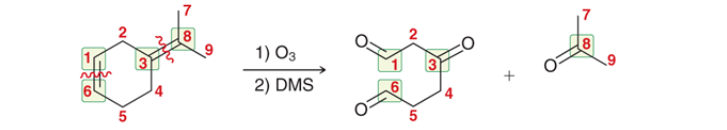
what to do with this?
Ozonolysis: snip the double bond in half —> create functional groups (almost always a ketone and/or aldehyde). for cyclos you will unravel them and create ketones on the end(s) of a carbon chain.