Chem Lab
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
You will be testing the reaction for completion with starch iodide paper. If excess hypochlorite is present, the paper will turn what color?
Option A
blue-black
Option B
purple-pink
Option C
red-orange
Option D
it won't change color
Option A
blue-black
During the work-up of the oxidation reaction you will perform a micro-extraction in a centrifuge tube. The organic solvent you will be using to perform the extraction is dichloromethane (DCM). Will the DCM be on the top layer or the bottom layer during the extraction? Why?
Option A
The organic layer will be on top of the water layer because halogenated solvents like dichloromethane are less dense than water.
Option B
The organic layer will be on top of the water layer because halogenated solvents like dichloromethane are more dense than water.
Option C
The water layer will be on top of the organic layer because halogenated solvents like dichloromethane are more dense than water.
Option D
The water layer will be on top of the organic layer because halogenated solvents like dichloromethane are less dense than water.
Option C
The water layer will be on top of the organic layer because halogenated solvents like dichloromethane are more dense than water.ii
During the work-up of the oxidation reaction you will perform a micro-extraction in a centrifuge tube. The organic solvent you will be using to perform the extraction is dichloromethane (DCM). Will your camphor product be more soluble in the aqueous layer or the organic layer?
Option A
Organic
Option B
Aqueous
Option A
Organic
When you are instructed to add a drying agent (like sodium sulfate) to a solution at the end of an organic work-up, how can you tell when you have added enough?
Option A
You add the drying agent until it all clumps in the bottom of the flask.
Option B
You measure how many grams of solution you have and add an equal number of grams of drying agent.
Option C
You add just enough drying agent until some of it does not clump upon addition (i.e. you see "dry" drying agent in your flask).
Option D
You measure the volume of solution and add half that volume of drying agent.
Option C
You add just enough drying agent until some of it does not clump upon addition (i.e. you see "dry" drying agent in your flask).
The procedure says DCM should be used on the bench disposed of in halogenated waste. However, very recently DCM is under a lot of scrutinty for its toxicity, so we have to be careful when handling it and disposing of waste. So this semester, we will be using DCM exclusively IN THE FUME HOOD and disposing of it in a SPECIAL DCM WASTE BOTTLE.
Just so we are clear: Where are we allowed to use DCM this week?
Option A
In the fume hood
Option B
On the bench
Option C
In the hallway
Option A
In the fume hood
Following this experiment, what will I do with my camphor product before leaving lab?
Option A
Throw it away in the trash
Option B
Throw it away in solid waste
Option C
Label the flask, seal it and leave it on the bench
Option D
Label the flask, seal it, and store it in my tote until next week
Option D
Label the flask, seal it, and store it in my tote until next week
What is the stereochemical relationship between the molecule (1S)-(+)-borneol (also called isoborneol) and (1R)-(+)-borneol?
Option A
enantiomers
Option B
diastereomers
Option C
same molecule
Option D
not related
Option B
diastereomers
We used an enantiomerically-pure starting material for this reaction. Suppose we used racemic borneol instead. Propose an analytical technique that could be used to evaluate the stereochemistry of the new camphor product(s).
A technique that could be used to evaluate the stereochemistry of the new camphor product would be using polarimetry. Possibly running and rotating the new product to measure to evaluate.
What was the role of acetic acid? Select all that apply.
Option A
solvent
Option B
proton donor
Option C
oxidizing agent
Option D
reducing agent
Option A
solvent
Option B
proton donor
What was the purpose of adding sodium bisulfite (NaHSO3) solution during the reaction workup?
Option A
To neutralize excess acetic acid
Option B
To neutralize excess HOCl
Option C
To precipitate out the product
Option D
To reduce camphor
Option B
To neutralize excess HOCl
If your solid camphor product has a melting point slightly lower than you expect, what can you conclude about the product? Select all that could apply.
Option A
The product cannot possibly be camphor
Option B
The product could be camphor contaminated with solvent (it is wet)
Option C
The product is actually borneol
Option D
The product could be camphor contaminated with starting material
Option B
The product could be camphor contaminated with solvent (it is wet)
Option D
The product could be camphor contaminated with starting material
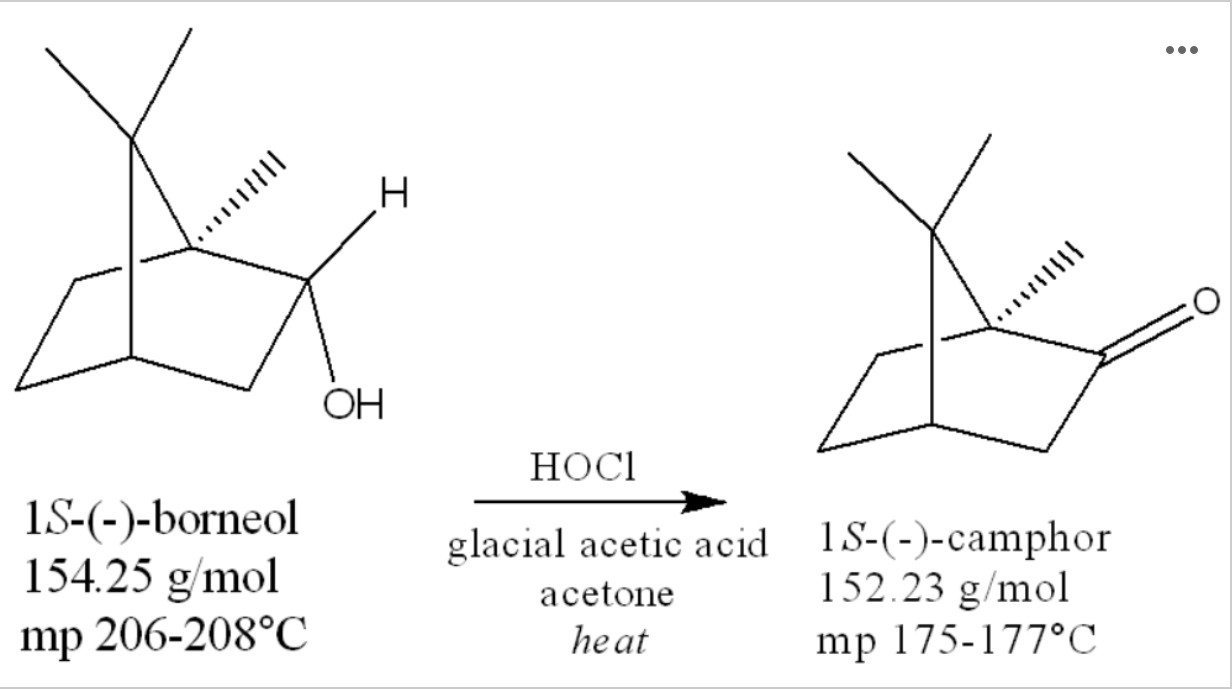
The oxidation of borneol to camphor is shown below:
Suppose you are monitoring the reaction progress by IR spectroscopy. What peaks would you expect to see appear/disappear as the reaction proceeds? Select all that apply.
Option A
Disappearance of strong, broad stretch (dog tongue) at 3200-3400 cm-1
Option B
Appearance of strong, broad stretch (dog tongue) at 3200-3400 cm-1
Option C
Disappearance of strong, very broad stretch (cow udder) at 3200-3600
Option D
Appearance of strong, very broad stretch (cow udder) at 3200-3600 cm-1
Option E
Disappearance of very strong, sharp stretch around 1700 cm-1
Option F
Appearance of very strong, sharp stretch around 1700 cm-1
Option A
Disappearance of strong, broad stretch (dog tongue) at 3200-3400 cm-1
Option F
Appearance of very strong, sharp stretch around 1700 cm-1
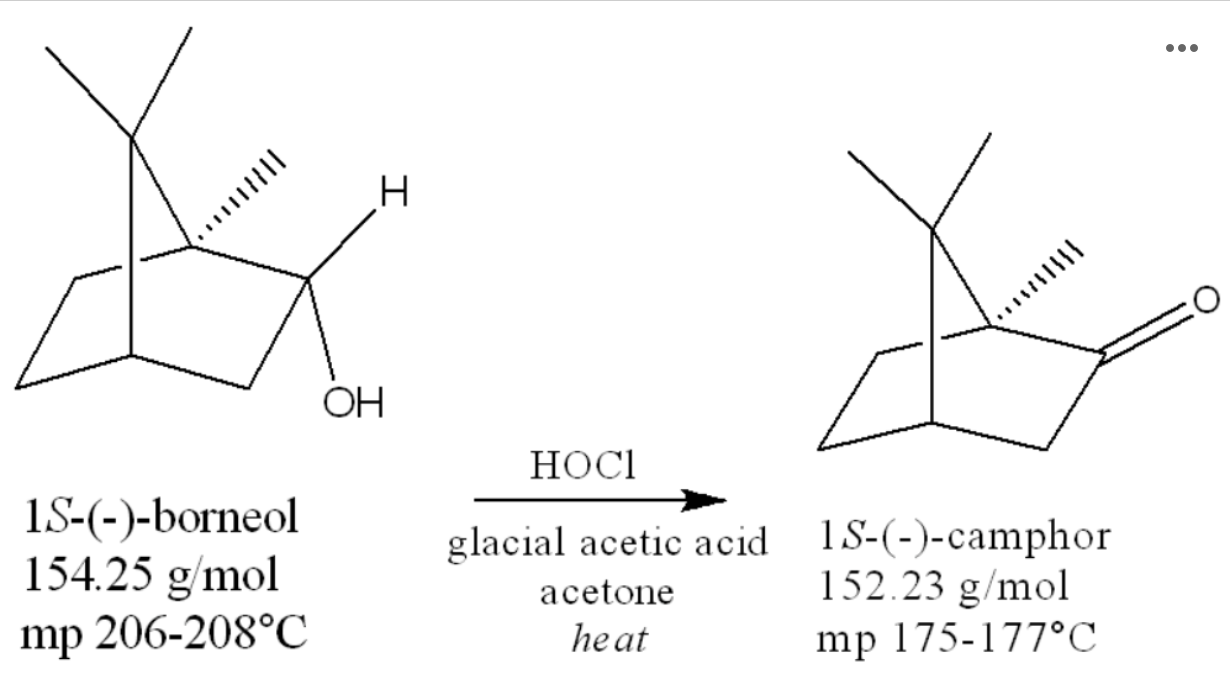
The overall reaction for the oxidation of borneol is shown below:
The atom being oxidized is. . The atom being reduced is .
-OH, chlorine
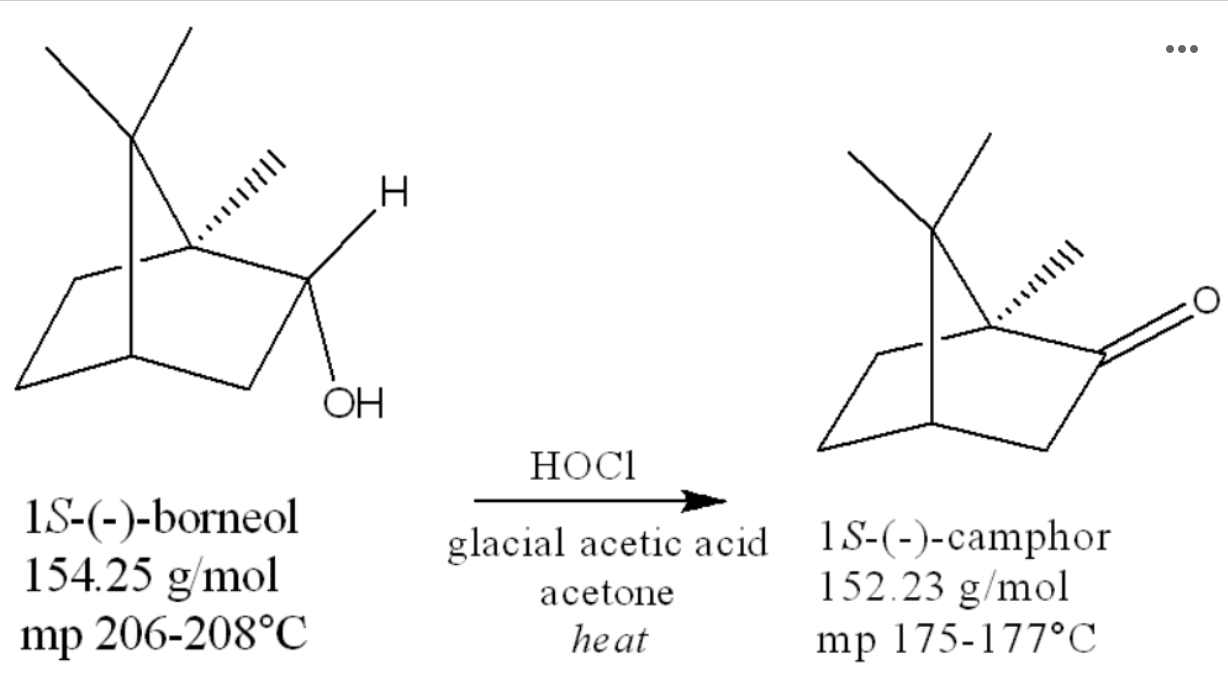
The oxidation of borneol to camphor is shown below:
If you started with 0.35 g borneol, what is your theoretical yield of camphor? Report your answer to three decimal places.
0.345
True or false: In addition to conducting the Experiment 2: Reduction of Camphor, I will also be setting up my Diels-Alder reaction for Experiment #3, and I should have procedures written for both.
True
False
True
oxidation
increasing number of C-O bonds
reduction
increasing number of C-H bonds
oxidizing agent
oxidizes something else; gets reduced during reaction
reducing agent
reduces something else; gets oxidized during reaction
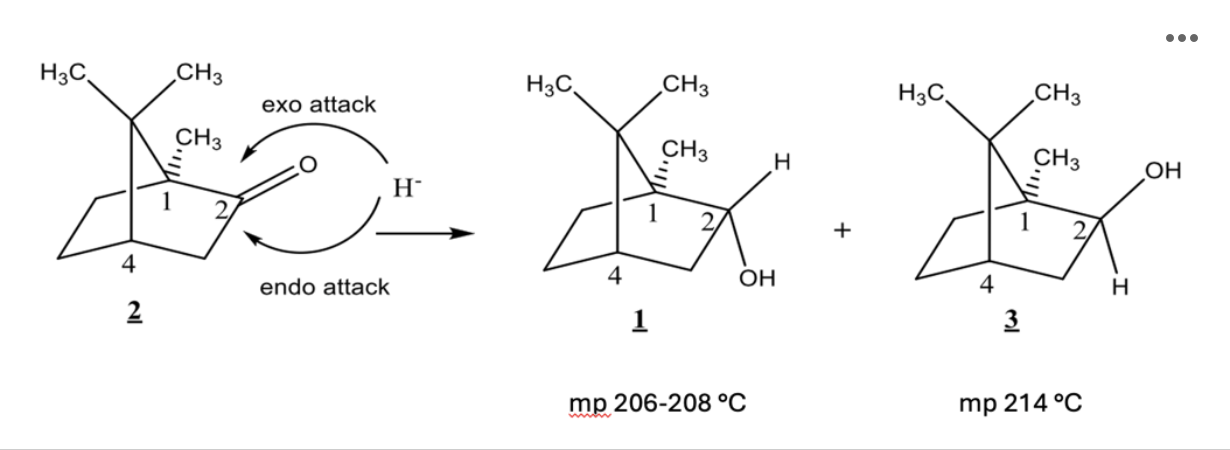
The reaction for this week is shown below:
What is the stereochemical relationship between compounds 1 and 3?
Option A
same molecule
Option B
isomers
Option C
enantiomers
Option D
diastereomers
Option E
meso
Option D
diastereomers
NaBH4 is a reducing agent. What does it add to the camphor molecule to generate borneol/isoborneol?
Option A
proton (H+, hydrogen cation)
Option B
hydrogen atom ("H dot")
Option C
hydride (H-, hydrogen anion)
Option D
neutron
hydride (H-, hydrogen anion)
NaBH4 is a reducing agent. Theoretically, how many moles of camphor can be reduced by ONE MOLE of NaBH4?
4
After the reaction mixture cools slightly, you are instructed to carefully add ice water. What is the purpose of this?
After the reaction mixture cools slightly, we are instructed to add ice water to destroy any remains of reactive that might continue the reduction and stop it overall. The cooling helps bring down the heat and solidify.
After the reaction mixture cools slightly, we are instructed to add ice water to destroy any remains of reactive that might continue the reduction and stop it overall. The cooling helps bring down the heat and solidify.As the reduction of camphor to borneol proceeds, the product precipitates as a white solid. How will the crude solid product be collected?
Option A
gravity filtration
Option B
vacuum filtration
Option C
distillation
Option D
extraction
Option B
vacuum filtration
After isolating the crude product, you will dissolve it in ether and treat it with sodium sulfate. Why?
After isolating the crude product, I will dissolve in ether and treat it with sodium sulfate because the solution remains with water. treating it with sodium sulfate after dissolving will remove the remain of water that wasn't removed from the first step, helping confirm a more pure product.
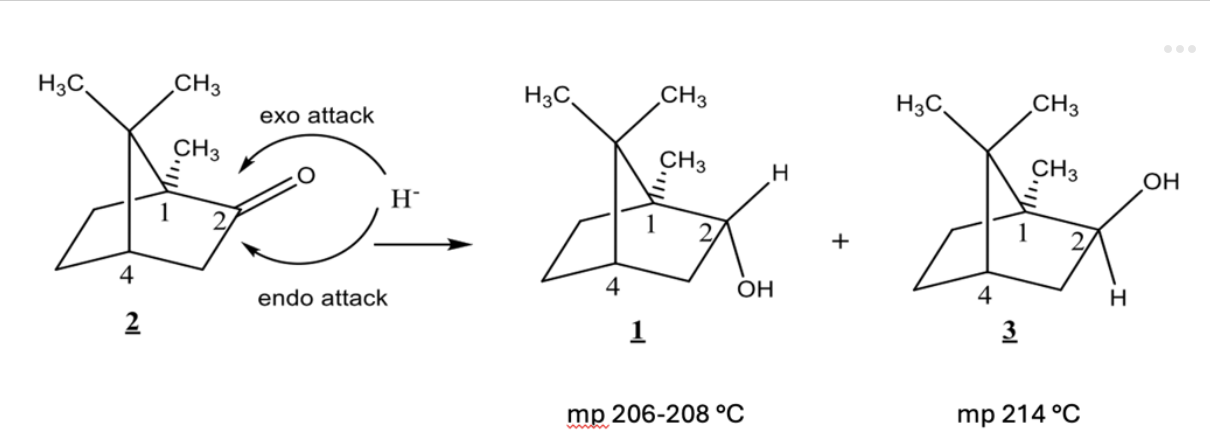
True or false: Compounds 1 and 3 (shown below) have identical 1H NMR spectra.
False
The IUPAC names of many of these compounds contain (+) and (-) notations, which indicate the direction each compound rotates plan-polarized light. Specifically, (+) compounds rotate light in the direction, and (-) compounds rotate light in the direction.
dextrorotatory, levorotatory
True or false: (+) and (-) notation directly tells you if a chiral center is R or S.
False
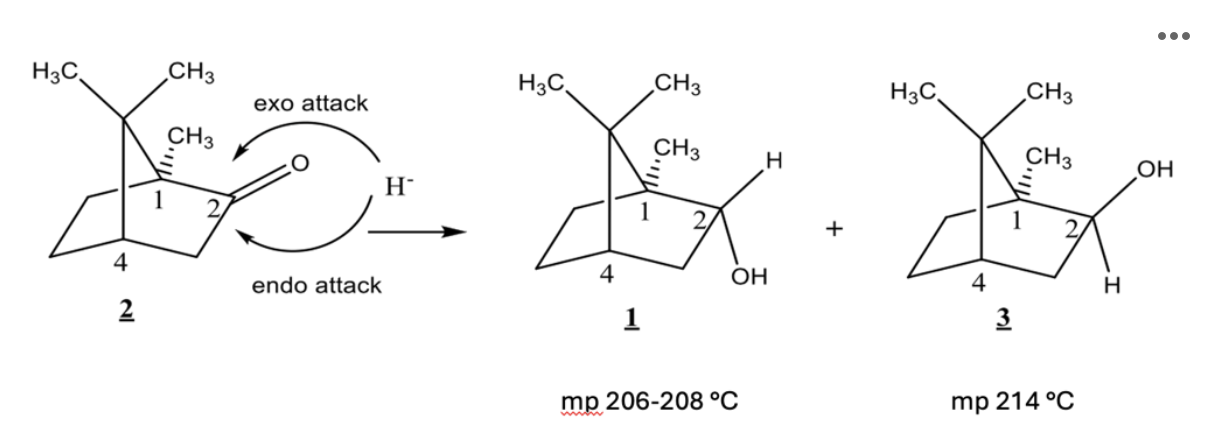
The reaction you completed is shown below:
Isoborneol (Compound 3) has which of the following:
Option A
exo hydrogen
Option B
endo hydrogen
Option C
exo OH
Option D
endo OH
Option E
1H NMR signal at 4.0 ppm
Option F
1H NMR signal at 3.6 ppm
Option B
endo hydrogen
Option C
exo OH
Option F
1H NMR signal at 3.6 ppm
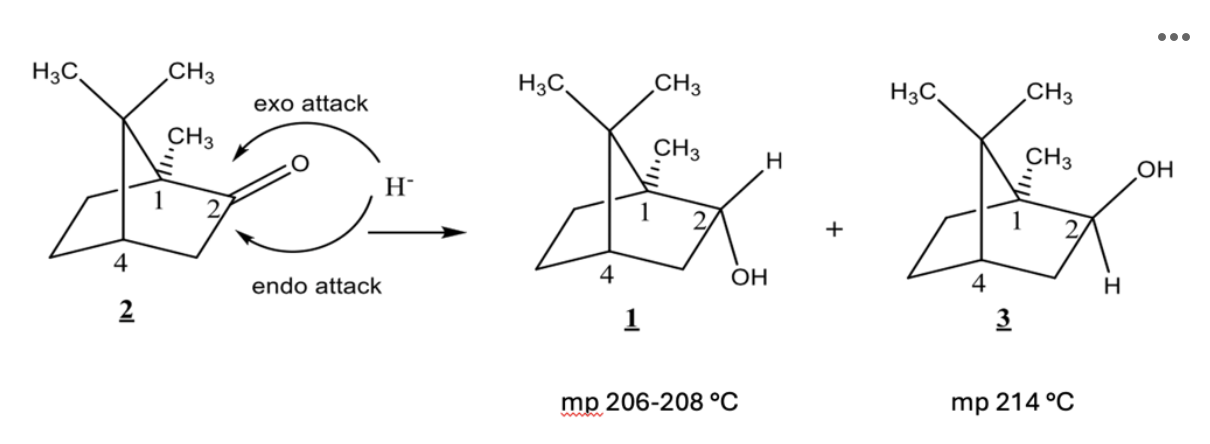
The reaction you completed is shown below:
Based on the mechanism of this reaction, which would you expect to be the major product?
Option A
Compound 1 (borneol)
Option B
Compound 3 (isoborneol)
Option B
Compound 3 (isoborneol)
Would IR spectroscopy have been a useful technique to characterize the reaction product mixture? Why or why not?
IR spectroscopy would not be a useful technique to characterize the reaction product mixture because both of the products are alcohols and are very similar in functional groups and IR absorption. It would be more difficult to tell apart the product.
You set this experiment up ~1 week in advance to allow the product crystals to form slowly. Suppose you come into lab this week, and you have no crystals in your flask. What should you do? Select all possible options.
Option A
freak out!
Option B
steal some product from your benchmates when they aren't looking
Option C
write "the experiment failed I have no data" in my lab report
Option D
stick your flask in an ice bath for a bit
Option E
gently scratch the inside of your flask using a stir rod or spatula
Option F
use the rotovap to evaporate roughly half of the THF volume in your flask, then re-cool your mixture over ice
Option D
stick your flask in an ice bath for a bit
Option E gently scratch the inside of your flask using a stir rod or spatula
Option F
use the rotovap to evaporate roughly half of the THF volume in your flask, then re-cool your mixture over ice
reagents: furan
diene
reagent: maleic anhydride
dienophile
reagent: tetrahydrofuran (THF)
reaction solvent
reagent: deuterated chloroform
NMR solvent
After isolating your crystals via vacuum filtration, you will rinse them with COLD THF. Why does this solvent need to be cold?
The solvent needs to be cold in order to separate any possible impurities that may have remained despite the vacuum filtration.
True or false: the Diels-Alder reaction occurs via a concerted mechanism.
true
You'll notice that for the last three experiments, we've used the terms "exo" and "endo" to describe the substituents on six-membered rings instead of the traditional "axial" and "equatorial" terminology you're used to. Why do you think that is?
I think that is because the ring system used is rigid and isn't able to be flipped and instead are closed in shape.
The Diels-Alder reaction forms new sigma bonds and new pi bond.
2, 1
When we interpret 1H NMR spectra, we evaluate which of the following? Select all that apply.
Option A
number of signals
Option B
integration of each signal
Option C
chemical shift of each signal (ppm)
Option D
splitting pattern of each signal
all the above
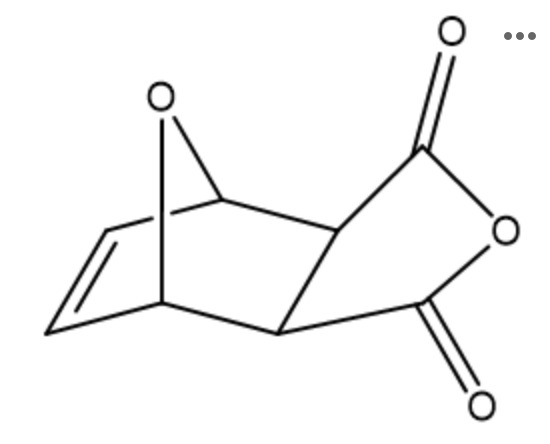
How many signals would you expect to see in the 1H NMR spectrum of our product?
3
Where do you dispose of excess THF? (hint: look up the structure!)
Option A
halogenated waste
Option B
non-halogenated waste
Option C
sink
Option D
that special DCM waste that's still sitting in the hood for some reason
Option B
non-halogenated waste
You learned in lecture that almost all Diels-Alder reactions will exclusively form the endo product. However, Diels-Alder reactions using furan favor the exo product. Why?
Option A
furan retains its aromaticity in the product, forcing the ring to flatten
Option B
steric hindrance in the transition state favors the exo orientation
Option C
the exo product is stabilized by hydrogen bonding, but the endo product is not
Option D
the reaction occurs too quickly for the more stable endo product to form, so the kinetic exo product is formed instead
Option B
steric hindrance in the transition state favors the exo orientation
The bicyclic product of this reaction possesses which of the following? Select all that apply.
Option A
a six-membered cyclohexane ring
Option B
a six-membered cyclohexene ring
Option C
a bridging oxygen atom across the ring
Option D
an endo substituent
Option E
an exo substituent
B, C, E
As you analyze your 1H NMR spectrum, you notice a weird set of peaks that don't belong to your product. The peaks are both multiplets that resonate at frequencies of 3.76 ppm and 1.85 ppm. Can you propose a reasonable explanation as to what compound these peaks belong to, and how they got in your NMR sample? (There might be a resource on Blackboard that can help you...)
A reasonable explanation as to what compound those peaks belong to are of THF. They got into the NMR sample because the crystal;s may not have been fully dried, seeming the product was still wet with THF.
Furan is an excellent and highly reactive diene. Why?
Option A
it contains an oxygen atom
Option B
it is locked in the s-cis conformation
Option C
it is locked in the s-trans conformation
Option D
it is close in structure to the reaction solvent (THF)
B
Maleic anhydride is a fairly good dienophile. Why?
Option A
it is locked in the s-cis conformation
Option B
it is locked in the s-trans conformation
Option C
it contains strong electron-withdrawing groups
Option D
it has a high boiling point so we can aggressively heat it
CTrue or false: deuterated chloroform (CDCl3) produces a signal in my 1H and 13C NMR spectra when used as an NMR solvent.
True or false: deuterated chloroform (CDCl3) produces a signal in my 1H and 13C NMR spectra when used as an NMR solvent.
True
What features of our 13C NMR spectrum will we analyze? (Hint: it's not the same as 1H NMR!)
A, C
True or false: The aromatic ring of acetanilide is activated toward electrophilic aromatic substitution.
True
Reagent: Br2
electrophile
Reagent: pi electrons of aromatic ring
nucleophile
reagent: acetic acid
solvent
Reagent: HBr
by product
Do some outside research: What is the approximate melting point of the hypothetical ortho-brominated product (o-bromoacetanilide) in degrees C?
100The scheme for the bromination of acetanilide is shown below.
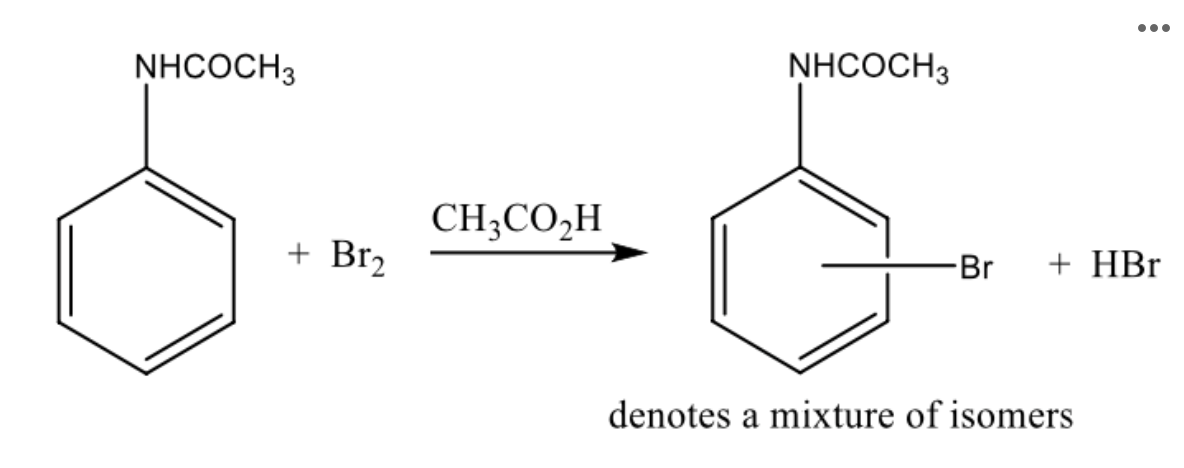
The scheme for the bromination of acetanilide is shown below. Based on the directing-group effect of the amide group, which isomer(s) could theoretically be formed in this reaction?
ortho, para
You will be using bromine solution in this experiment. Which of the following measures will we take to ensure our safety while handling this nasty chemical? Select all that apply.
Option A
wear gloves
Option B
dispense bromine in hood only
Option C
cap or stopper my flask containing bromine before returning to the bench
Option D
neutralize excess bromine in our reaction using sodium thiosulfate solution before moving onto product collection/purification
all the above