4.8 Chemical analysis
1/30
Earn XP
Description and Tags
Completed flashcard set
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Define pure substance in chemistry and in everyday language
Chemistry: A pure substance is a single element or compound not mixed with any other substance
Everyday language: A pure substance can mean a substance that has had nothing added to it, so it is unadulterated and in its natural state
What is true about pure elements and compounds?
Pure elements and compounds melt and boil at specific temperatures
State a way that you can distinguish a pure substance from a mixture
Pure elements and compounds melt at specific temperatures.
Melting and boiling point data can be used to distinguish pure substances from mixtures
Define formulation
A formulation is a mixture that has been designed as a useful product
Many products are complex mixtures in which each chemical is present in a measured quantity and has a particular purpose
Describe how formulations are made
Formulations are made by mixing the components in carefully measured quantities to ensure that the product has the required properties
Give examples of formulations
Fuels
Cleaning agents
Paints
Medicines
Alloys
Fertilisers
Foods
What can chromatography be used for?
To separate substances out of mixtures based on their solubilities
It can then be used to identify the substances
Describe the two phases of chromatography
Mobile phase: where the molecules can move
The solvent is the mobile phase because it moves
Stationary phase: where the molecules cannot move
The paper is the stationary phase because it does not move
What is the Rf value and give its formula
Rf value is the ratio of the distance moved by a compound (centre of spot from origin) to the distance moved by the solvent
Rf = distance moved by substance / distance moved by solvent
Suggest how chromatographic methods can be used for distinguishing pure substances from impure substances
Different compounds have different Rf values in different solvents
The compounds in a mixture may separate into different spots depending on the solvent but a pure compound will produce a single spot in all solvents
What determines which substances move further up the chromatography paper
Molecules with higher solubility in the solvent and which are less attracted to the paper will spend more time in the mobile phase and will therefore move further
Explain what separation of a substance in chromatography depends on
The mobile phase moves through the stationary phase and anything dissolved in the mobile phase moves with it
How quickly a substance moves depends on its distribution between the two phases - whether it spends more time in the mobile phase or the stationary phase
Substances that spend more time in the mobile phase than the stationary phase will move further
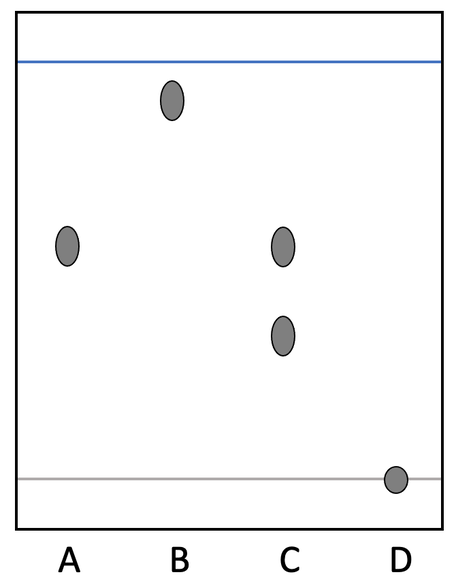
Label the chromatogram
Solvent front: the furthest part reached by the solvent
Origin: must be in pencil as pencil is insoluble so will not dissolve in solvent and be carried up the paper
State the test for hydrogen
Give the result
Burning splint held at the open end of a test tube of the gas
Hydrogen burns rapidly with a squeaky pop sound
State the test for oxygen
Give the result
Glowing splint is inserted into a test tube of the gas
The splint relights in oxygen
State the test for carbon dioxide
Give the result
Carbon dioxide is shaken with/ bubbled through an aqueous solution of calcium hydroxide (limewater)
The limewater goes cloudy
State the test for chlorine
Give the result
Damp litmus paper is put into chlorine gas
The litmus paper is bleached and turns white
What is limewater?
An aqueous solution of calcium hydroxide
(Think: hydroxide, alkaline, limewater used in cleaning)
What type of tests can be used to identify metal ions?
Flame tests
Lithium, sodium, potassium, calcium and copper compounds produce distinctive colours in flame tests
Sodium hydroxide solution
A precipitate can be formed which have characteristic colours
State the results of the flame tests
Memorise:
Crimson lipstick
Crimson, Li (lithium)
Lithium compounds result in a crimson flame
The curious green frog
Cu (copper), green
Copper compounds result in a green flame
A yellow sun
Yellow, Sodium Na+
Sodium compounds result in a yellow flame
California is hot! (red/orange)
Calcium, red
Calcium compounds result in an orange-red flame
A pretty lilac in a flower Pot
Lilac, potassium
Potassium compounds result in a lilac flame
If a sample for a flame test contains a mixture of ions, what happens?
Some flame colours can be masked
Describe the sodium hydroxide tests for metal ions
Forms white metal hydroxide precipitates:
Solutions of magnesium ions
Solutions of calcium ions
Solution of aluminium ions
Only aluminium hydroxide precipitate dissolves in excess sodium hydroxide solution
* Remember: magnesium + calcium white food supplements, aluminium foil used for food, odd one out so dissolves
Forms coloured precipitates
Solutions of copper(II) form blue
Solutions of iron (II) form green
Solutions of iron (III) form brown
* Remeber: copper = young boy, green brown is earthy (II, III)
Name the negative ions you need to be able to identify
Carbonate CO32- ions
Halide ions
Sulfates SO42-
Describe how to identify carbonates
Carbonates contain CO32- ions
Add a sample in a test tube and add a few drops of dilute acid - carbon dioxide will be produced. Connect the tube to a tube of limewater. If carbonate ions are present, the carbon dioxide will released will turn limewater cloudy when it bubbles through it
Describe how to identify halide ions
Halide ions in a solution produce precipitates with silver nitrate solution in the presence of dilute nitric acid
Halide ions with silver nitrate solution
Presence of dilute nitric acid
A silver halide forms
Silver chloride: white
Silver bromide: cream
Silver iodide: yellow
Memorisation: swedish, hallo, silver jewellery, n, chlorine suncream pool, bro.., idied yellow gon
Describe how to identify sulfates
Sulfate ions in solution produce a white precipitate with barium chloride solution in the presence of dilute hydrochloric acid
White precipitate barium sulfate forms
Sulfate ions and barium chloride solution
In the presence of dilute hydrochloric acid
If sulphate ions are present a white precipitate of barium sulfate will form.
What is instrumental method analysis?
Give advantages of this over chemical tests
Using specific scientific instruments instead of conducting tests
Advantages:
Very sensitive; they can detect even very small amounts of substances
Very fast
Very accurate
For flame emission spectroscopy:
If a sample contains multiple different metal ions, the line spectrum produced will show the lines for all of them
Give an example of an instrumental method used to identify substances
Flame emission spectroscopy
Used to analyse metal ions in solutions
Describe the method of flame emission spectroscopy
A sample is placed in a flame
As the ions heat up, their electrons become excited (absorb energy and move up to a higher energy level)
When they drop back to their original energy level, they transfer energy as light
The light passes through a spectroscope which can detect different wavelengths of light to produce a line spectrum
The combination of wavelengths emitted by an ion depends on its charge and its electron arrangement
Since no two ions have the same charge and the same electron arrangement, different ions emit different wavelengths of light
Therefore, each ion produces a different pattern of wavelengths and has a different line spectrum
The intensity of the spectrum indicates the concentration of ion in the solution
Therefore, line spectrums can be used to identify ions in solution calculate their concentrations
What do spectroscopes do?
Spectroscopes can detect different wavelengths of light to produce a line spectrum
Every different metal ions produces a unique pattern of wavelengths (as no two metal ions have the same electron arrangement and charge) so they all produce different line spectrums
The green ink contains more than two compounds. Suggest two reasons why only two spots are seen on the chromatogram
Some of the compounds are colourless (in solution)
Some compounds have the same Rf values