Chemistry - Flash Card Set 2
1/63
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
what is electromagnetic radiation (EM)?
a form of energy (also known as light) and is described in waves
what is wavelength (λ)?
the distance between the peaks of each wave
what is a photon?
a particle of light
what is the formula between wavelength (λ), frequency (v), and speed of light (c)?
c = λv ; v = c/λ ; λ = c/v
how are wavelength and frequency related?
inversely related ; shorter wavelength, higher frequency and longer wavelength, lower frequency
what is the electromagnetic spectrum?
the variety of light and which kind we can see based on their wavelengths
what colors are associated with wavelength?
reds = long wavelength/low frequency ; blues = short wavelength/higher frequency
what is the formula to calculate the energy of a photon?
E = hv ; E is energy (in J), h is Planck’s Constant (6.626 × 10⁻³⁴Js), v is the frequency (in s⁻¹)
the farther the energy level is from the nucleus…
the higher the energy of the electrons
what is “ground state?”
when electrons fill up the lower energy levels first and as new ones are added, go into higher levels
what is “excited state?”
when electrons get the energy to jump from its current energy level to a higher one
what is absorption?
electron gains potential energy and moves farther from the nucleus
low potential energy → high potential energy
what is emmission?
electron loses potential energy and moves closer to the nucleus
high potential energy → low potential energy
one way an electron can give off energy to return to ground state is…
through the form of light (emission)
what are orbitals?
shapes that describe the regions where an electron is likely to be found; a complete set of orbitals in a given energy level is called a subshell
list the 4 common orbital types and their shapes
s orbital - spherical
p orbital - dumbell
d orbital - clover and donut with lobe above and below
f orbital - complex tetrahedral
how many orbitals are there in each energy level and how many electrons can each orbital hold?
one s orbital - 2 electrons
three p orbitals - 6 electrons
five d orbitals - 10 electrons
seven f orbitals - 14 electrons
how can you categorize electron configuration on a periodic table?
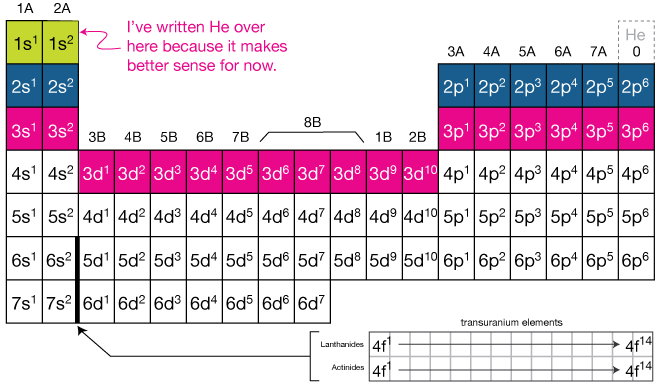
what is electron quantization?
the orbitals in an atom can only have a specific set of values; an electron requires a particular amount of energy to either give or release in order to move it to a different energy level
what is electron configuration?
when each element has its own unique configuration that describes which subshells have electrons and how many
example: Boron = 1s²2s²2p¹
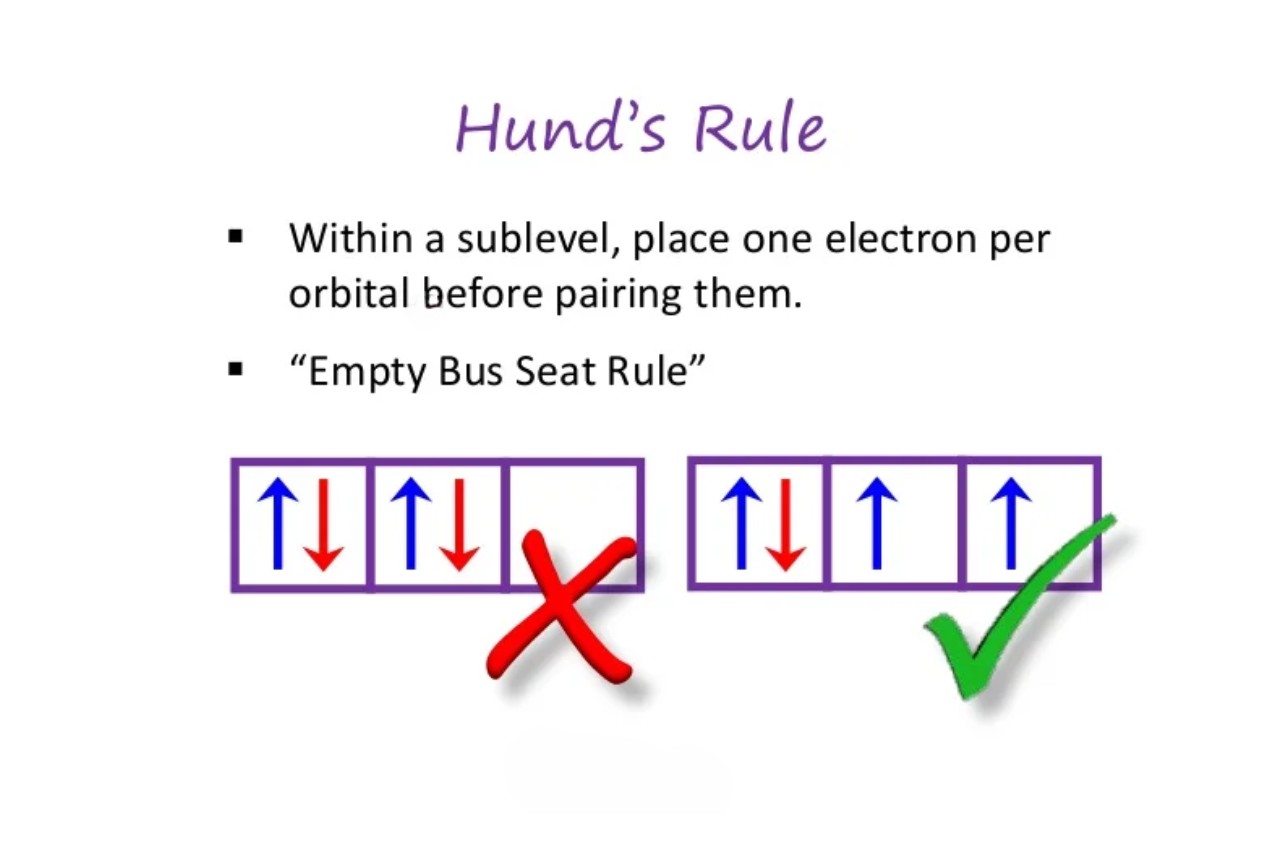
what are orbital diagrams or the “spinning” property of electrons?
a graphical form of electron configuration, representing the mass and charge of electrons as arrows and which directions they spin (up or down); Hund’s Rule: ground state atoms will fill unoccupied orbitals first
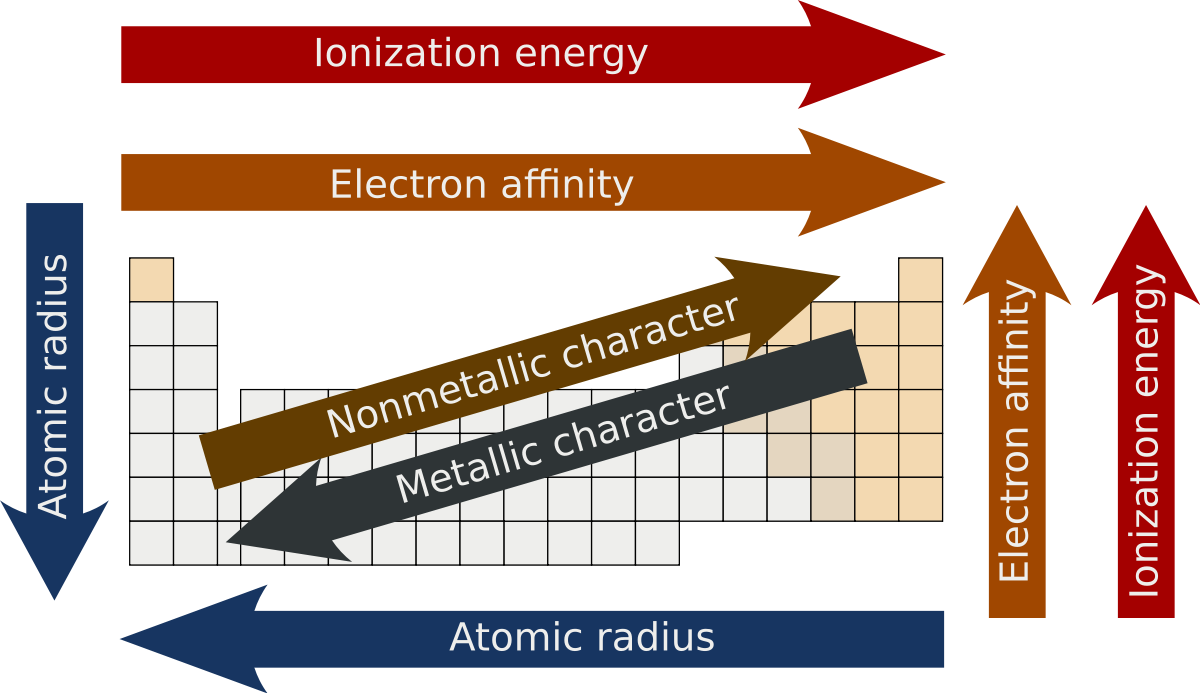
what are the types of periodic trends?
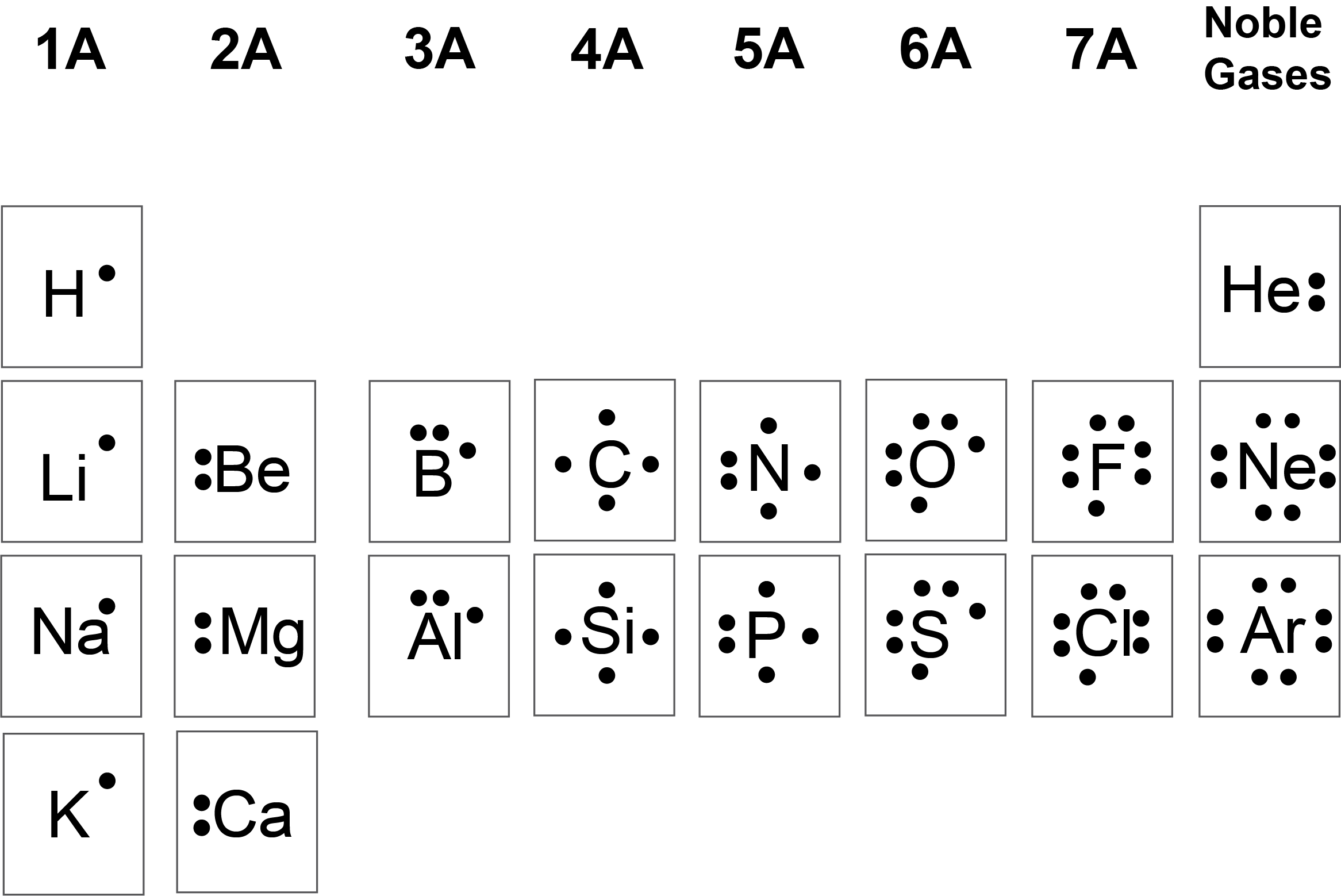
what are valence electrons and how do you find them?
the electrons occupying the outermost energy levels of an atom are called the valence electrons and are responsible for chemical bonding; for elements in group 1A-8A, the number of valence electrons in an atom is simply the group number
simply put, how do you draw Lewis dot strucures?
the chemical symbol in the center, surrounded by as many dots as that atom has valence electrons
what is the Octet Rule?
atoms react in such a way as to give them 8 electrons in their outermost energy level (valence shell); noble gases usually do not react because their shell is already filled with 8
what is the Duet Rule?
hydrogen atoms attempt to pick up another electron to give the same configuration as Helium
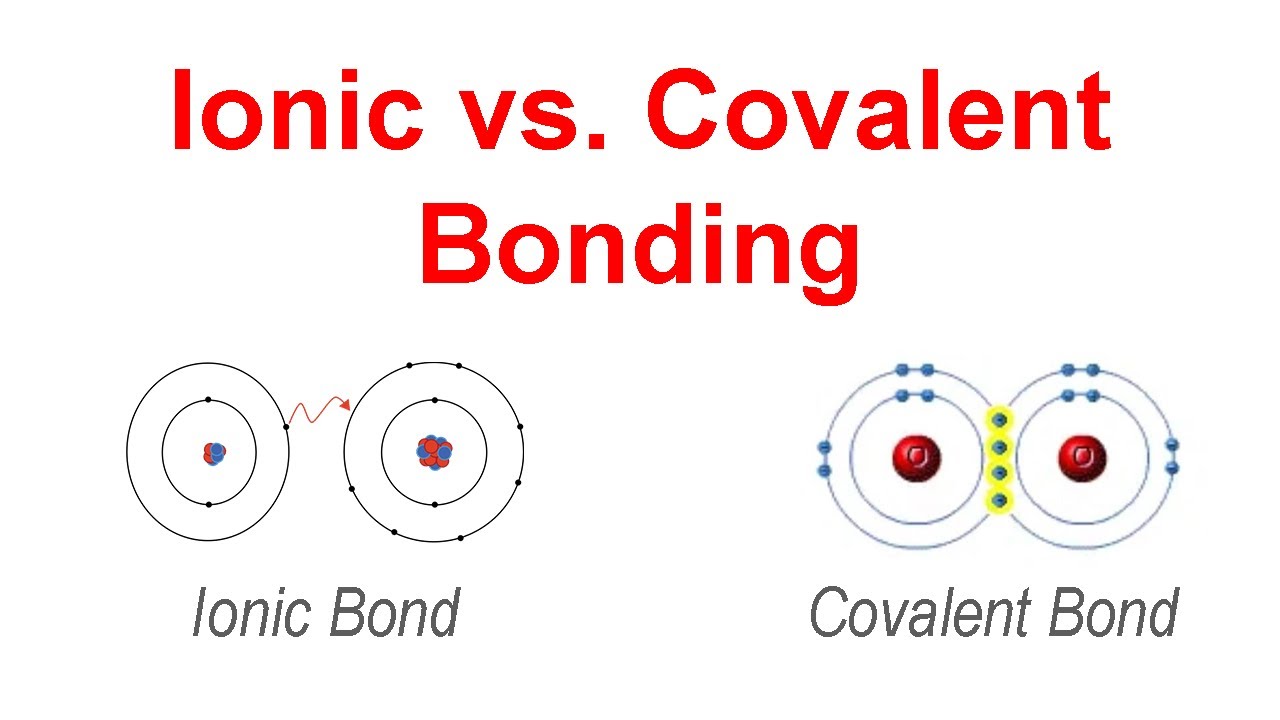
what are the types of bonding?
ionic: mutual attraction of oppositely charged ions through the gain or loss of electrons of an atom (metals & nonmetals)
covalent: a pair of electrons shared by two atoms
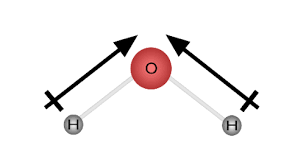
what is the VSPER theory?
Valence Shell Electron Pair Repulsion: electron pairs move as far away from each other as possible; electrons all have negative charges so they repel one another, implying a specific angle
how do you find the polarity of a bond?
if two atoms which are covalently bonded have substantially different electronegativity values (> 0.4 units) then the bond is polar covalent
what are polar and non-polar covalent bonds?
polar: unequal sharing of electrons
non-polar: equal sharing of electrons
what is a dipole moment?
when electrons are drawn to more electronegative atoms which develop a “partial negative charge” and a “partial positive charge” on opposite sides; only occurs in polar molecules
atomic elements are…
all metals and noble gases
non-metal elements are…
molecular
seven common diatomic molecules
H₂, N₂, O₂, F₂, Cl₂, Br₂, I₂
molecular (covalent) compounds are made of…
non-metals
ionic compounds are made of…
metal + non-metal and polyatomic ions
elements are
atomic and molecular
compounds are
molecular and ionic
an ionic compound should have what kind of charge?
no total charge (they neutralize)
in ionic compounds, the cation is always listed
first
what are polyatomic ions?
ions which are derived from two or more atoms; molecules that have collectively gained or lost electrons to become an ion
ions are held together by…
covalent bonds
polyatomic ions can contain both
metals an non-metals
which is the only commom polyatomic cation?
NH₄+
the classifications of compounds include…
binary non-metals (molecular compounds) and ionic compounds
binary non-metals are made of…
non-metal + non-metal
ionic compounds are made of…
cation + anion
how do you name binary nonmetal compounds?
prefix + name of 1st element and prefix + base name of 2nd element + -ide
what are the first 10 prefixes and their numerical values?
mono-
di-
tri-
tetra-
penta-
hexa-
hepta-
octa-
nona-
deca-
what should H₂O, NH₃, and PH₃ always be called?
water, ammonia, and phosphine
when do you not use prefixes in naming binary nonmetal compounds?
when hydrogen (H) appears first
what does (g) and (aq) mean?
“gas” and “aqueous" (can dissolve in water)
aqueous (aq) compounds are named as…
acids with -hydro as a prefix and -ic as a suffix
when naming compounds, prefixes are only used for…
binary nonmetals
how do you name individual cations?
the name of the metal + “ion”
how do you name individual anions?
change the ending of the element’s name to “-ide”
how do you name ionic compounds?
the name of the cation + the name of the anion
what are hydrates?
an ionic compound which has a fixed amount of water molecules within its structure
for every one unit of a particular molecule, there is a subsequent amount of water molecules associated with it
what are oxyanions and how do you name them?
an anion containing different numbers of oxygen atoms but have the same charge
“-ite” before “-ate”
less oxygen than “-ite” = “hypo”
more oxygen than “-ate” = “per-”
how do you name oxyanions containing hydrogen?
the H is always listed first and the normal charge of the ion is changed by +1 or even +2
prefix “bi-” or the word “hydrogen” before the name of the anion
what is an oxyacid and how do you name it?
compounds which contain exactly as many H+ ions as are necessary to cancel out the negative charge of an oxyanion
“-ous” before “-ic”
“-ite” before “-ate”
what is the mole (mol) and how much is just one of it?
the mole (mol) is a unit of quantity in chem that is used to count large numbers of submicroscopic pieces of matter (such as atoms)
1 mol = 6.022 × 10²³
how do you find # of atoms? how do you find # of mol?
(mol) x (Avocadro’s number) = atoms
(atoms) / (Avocadro’s number) = mol
what is molar mass?
the mass of 1 mole of atoms in grams
atomic mass (amu) = molar mass (grams)