Chemistry MCAT
1/393
Earn XP
Description and Tags
tenets of transition
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
394 Terms
what is the atomic number of an element equal to?
the number of proton’s in the element’s nucleus
neutron
Uncharged because it contains a positive and negative charge, which can be loberated during b+ and b- decay as positron and electron, respectively.
mass number
number of protons + number of neutrons
isotope
an atom that shares an atomic number (number of protons, basically same element) with another atom but has a different mass number due to having more/less neutrons.
charge of an electron
1.6E-19 C
valence electrons
determine the way an atom reacts and its chemical properties
what do rows allow us to determine?
The energy level at which the element's valence electrons reside.
The number of each period is that period's principal quantum number
where are metals found on the periodic table?
left and middle
general characteristics of metals
High conductance of heat and electricity
High melting point
Lustrous
Malleable (can deform without breaking) and ductile (can be drawn into wire shape)
general characteristics of non-metals
Poor conductance of heat and electricity
Not lustrous
Gaseous (volatile)
Brittle
metalloids
between metals and nonmetals on periodic table, no general characteristics
going from left to right on periodic table
increases effective nuclear charge
effective nuclear charge
The actual attraction that outer electrons feel from the nucleus when you account for shielding. Inner electrons shield the outer electrons from the charge of the nucleus. This means that, even though the electrons are all being pulled upon by the same nucleus, that pull varies for each electron.
periodic trend for atomic radii
Atomic radius increases as you go left and down.
How does one determine whether an atom's ion will be bigger or smaller?
Gain electron --> Zeff spread over more electrons --> Individual electrons pulled less strongly --> Increase in orbit radius
Lose electron --> Decrease in orbit radius
What special case must we remember when considering Ionization energy?
Ionization energy is disproportionately high when attempting to cationize an atom with an octet because octets are very stable.
what is the atomic mass?
= mass number - mass lost in nuclear binding
However, the mass lost to binding energy is so small that the atomic mass and the mass number are basically the same.
What is the number following the element name when describing an isotope?
mass
what is atomic weight
Atomic weight is the weighted average of all different stable isotopes one might find of an element.
What does it mean for energy in this universe to be quantized?
That the energy in this universe is quantized means that the energy only comes in discrete packets, of which you can have only whole number values.
What is the atomic number of an element equal to?
The number of protons in the element's nucleus
What is an atomic emission spectrum?
Remember, the distances between energy levels are different in every atom. Therefore, the photons released when an electron falls from an excited state to ground state are different in every atom. We can use the photons an atom releases to determine its identity.
absorption spectrum
An absorption spectrum is like the atomic emission spectrum, only instead of cataloguing the photons that are emitted when excited electrons fall, absorption spectra catalogue the photons that are absorbed when ground state electrons are promoted to excited states.
Remember, the difference in energy going from energy level 1 to 2 is the same amount of energy as going from energy level 2 to 1, so the photons emitted by the emission spectrum are the same photons absorbed in the absorption spectrum.
Planck’s Constant
6.626*10-34 J*s
Heisenberg Uncertainty Principle
The Heisenbergy Uncertainty Principle states that it is impossible to determine the momentum and position of an electron simultaneously.
Measuring momentum impacts the certainty of the position while measuring the position affects the momentum of the electron.
Pauli Exclusion Principle
The Pauli exclusion principle says that no two electrons in a given atom can posess the same set of four quantum numbers. Specifically, this means that electrons in the same orbital must have opposite spins in order to be unique.
What is the purpose of the four quantum numbers?
The four quantum numbers exist so we can quickly and easily describe every electron's position in the atom. For example, if I assign the quantum numbers 2, 0,0,1/2, I know exactly which electron I am talking about: the electron in the second energy level, in the s orbital, in the up spin configuration.
first quantum number
The principal quantum number, n
+ Property: orbital size & energy level/shell
+ Allowed values: positive integers (1, 2, 3, etc.)
How does one calculate the energy required to promote an electron from one energy level to another?
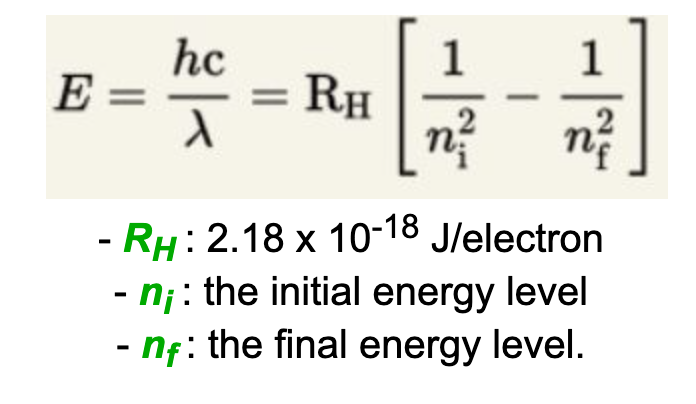
What does the magnetic quantum number tell you?
Orbital residence of an electron within a subshell e.g. a particular electron located in Px, Py, or Pz
what is the fourth quantum number
The spin quantum number, ms
+ Property: Spin orientation of e- in an orbital
+ Possible number: -/+ 1/2
What is the speed of light in air equal to?
3.00*108 m/s
effective nuclear charge
We need to know about effective nuclear charge because inner electrons shield the outer electrons from the charge of the nucleus. This means that, even though the electrons are all being pulled upon by the same nucleus, that pull varies for each electron, The actual attraction that outer electrons feel from the nucleus when you account for shielding. Increases as one goes right and up periodic table.
electron affinity
energy released by gaseous atom on gain of an electron.
electronegativity
force with which an atom will pull covalently shared electrons towards itself. Due to Zeff.
What is the formula for effective nuclear charge?
Zeff = Z - S
Z = # protons
S = # shielding (non-valence) electrons
What is the equation to calculate the centripital force and centripital acceleration on an object?
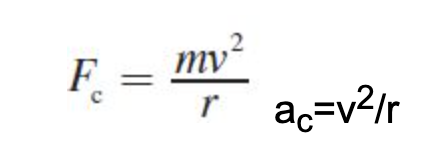
molar mass
Molar mass is how much mass you will have of something given a mole of it. It is usually expressed in g/mol
What is normality (N)?
What is normality (N)?
Remember when you learned that sometimes 1 mole of a molecule can be made up of more than one mole of a certain atom? For example, 1 mole of H2O is made of 2 moles of H and 1 mole of O?
Well, normality is a way to account for this difference in the molecule and the atoms into which it could potentially split up. Normality is like Molarity, in that it is measured in moles per liter. But, Normality is not the moles of the substance itself per liters. Instead, Normality is the moles of what we are interested in per liters.
In the below example, we are interested in Hydrogen ions. Notice how 3 different compounds at the same molarity, 1M can have very different Normalities due to their differential contribution of hydrogen ions to the solution.

What is the law of constant composition?
aka law of definite properties, states that the elemental composition of a pure substance never varies
empirical formula
An emiprical formula is the most-reduced whole number ratio of the atoms in the compound.
6 major reactions seen on test day
1. Combination
2. Decomposition
3. Combustion
4. Single Displacement
5. Double Displacement
6. Neutralization
What is the archetypal form of the combination reaction?
A + B → C
when two or more reactants come together to make 1 new product
combustion reaction
A combustion reaction is when a fuel is oxidized to create CO2 and H2O. The fuel will almost always be a hydrocarbon of some sort. The oxidizing agent will almost always be Oxygen.
CH + O2 → H2O and CO2
Neutralization
Neutralization reactions are a specific type of double-displacement reaction in which an acid reacts with a base to produce water and a salt.

What are the 2 special d-orbital modifications we must make to the electron filling order?
Only occurs in d and f (NOT P)
The 2-special cases of electron filling exist for the Chromium group of elements and the Copper group of elements.
For the chromium group, instead of taking on a normal 4s23d4 configuration, they will take a 4s13d5 configuration. This is because filling the 3d orbital up with 5 electrons (half) provides the atom with extra stability. So much extra stability, that the atom will pull an electron from the s orbital to fill the d orbital.
The same thing happens for the copper elements, only the d orbital electrons go from 9 to 10 instead of 4 to 5. For example, Copper's electron configuration is actually 4s13d10 instead of 4s23d9
What is an energy shell?
Energy shells are different distances at which electrons can orbit a nucleus with stability. The more energy an electron has, the further out they go.

What is the n+l rule?
The relative energies of subshells can be compared by comparing their values of n+l. Used to distribute electrons in order of low energy subshells to high energy subshells in the Aufbau principle.
1. The subshell with higher n+l value is higher in energy.
2. If two subshells share the same n+l value, the subshell with the higher n value is higher in energy
E.g. 2p vs 3s. n+l = 3 in both cases. 3s is higher in energy because (n = 3) > (n = 2)
What are the names of n, l, ml, and ms
Mnemonic: PAMS
n = Principal quantum number
Describes shell (energy level, orbit distance around nucleus) of an electron
[0, infinity)
l = Azimuthal quantum number
Describes subshell type (spdfg)
[0, n-1]
ml = Magnetic quantum number
Describes number of orbitals within a subshell (e.g. X, Y, Z orbitals in p subshell)
[-l, +l]
ms = Spin quantum number
Describes angular momentum of the electron
[-1/2 or + 1/2]
Shell, subshell, orbital, and spin
ionic character
= extent of ionic bond engagement by a compound. Increases with increasing total transfer of valence electrons. A function of difference between two atoms' electronegativities.
Difference must be at least 1.7
How does one name an organic molecule?
1. Identify the longest carbon chain containing the highest-order functional group. The number of carbons and the type of functional group will determine the parent name of the molecule.

2. Number the chain starting from the highest priority functional group. If the functional groups have the same priority, numbering the chain should make the numbers of any branches in the chain lowest.

3. Name the Substituents and place the names before the parent name. If there is more than one substituent of the same type, indicate it with the prefixes di, tri, etc. If there is more than one substituent of different types, place them alphabetically.
4. Assign numbers to each substituent.
5. Complete the name.

how are alkanes named as substituents and parent chains?
1. Alkane substituents are given the suffix -yl. For example, Meth-yl, Eth-yl, Prop-yl.
2. Alkane parent chains are given the default parent name, which is -ane. For example, Methane, Ethane, Propane.
How are alcohols named as substituents and parent chains?
1. Alcohol substituents are given the name (hydroxy-)
2. Alcohol parent chains replace -ane with -ol.
How are aldehydes named as substituents and parent chains?
1. Carbonyl substituents, whether they are aldehydes or ketones, are given the name (oxo-)
2. Aldehyde parent chains replace the -e of the parent name with -al
How are ketones named as substituents and parent chains?
1. Carbonyl substituents, whether they are aldehydes or ketones, are given the name (oxo-)
2. Ketone parent chains replace the -e of the parent name with -one.
What are the various forms of isomers?
1. Constitutional Isomers
2. Stereoisomers
a. Conformational Isomers
b. Configurational isomers
I. geometric
II. optical
i. enantiomers
ii. diastereomers

What are the 4 types of newman projections (ranked in decreasing stability/ higher energy)?
1. Staggered
a. Anti - Two largest groups are 180 degrees apart
b. Gauche - Two largest groups are 60 degrees apart
2. Eclipsed
a. Eclipsed - Two largest groups are 120 degrees apart
b. Totally Eclipsed - Two largest groups are 0 degrees apart

What are the ideal bond angles for a carbon with four bonds (sp3)?
What are the ideal bond angles for a carbon with four bonds (sp3)?
109.5 degrees
what does cyclopentane take to make ring strain as low as possible?
envelope
what does cyclobutane take to make ring strain as low as possible?
puckered
what are flagpole interactions?
Flagpole interactions refer to a specific type of steric strain that is exhibited by axial molecules on a ring. This is why it is more stable to place large groups in equatorial positions. You avoid this strain.
What is an optical isomer?
An optical isomer is an isomer that changes the way the molecule rotates plane-polarized light. Enantiomers and Diasteromers are both considered optical isomers.
chirality
Chirality is a word for handedness. What handedness means is that two objects are mirror images of one another, but due to the nature of their structure it is impossible to super-impose one on to the other. This is most easily seen in our hands. Hands are mirror images, but we can't super impose them.
Describe the physical and chemical properties of enantiomers?
The physical and chemical properties of enantiomers are basically equivalent, except in the case of two important exceptions.
1. Optical activity
2. Reactions in chiral (biological and natural) environments. For example, a lot of enantiomers of normal molecules like glucose and proteins can be toxic to humans.
optical activity
Optical activity refers to the rotation of plane-polarized light by chiral molecules. Enantiomers will rotate plane polarized light the same magnitude, but in opposite directions.
If you know the direction of rotation for one config, its enatiomer will be the exact oppisite. But between diastereomers it they could both rotate the same way or be opposite like enantiomers, theres no way to tell how the other diasteromer will rotate light from one given.
A molecule that rotates plane polarized light to the right aka clockwise, is dextrorotary (d-) and is labeled (+).
A molecule that rotates plane polarized light to the left aka counterclockwise is levorotary (l-) and is labeled (-).
MUST BE DETERMINED EXPERIMENTALLY, DOESNT RELATE TO R and S/ D and L (sugars)
specific rotation
Specific rotation is the amount a compound rotates light per concentration of the molecule and length of the tube through which the light passes. Constant regardless of concentration at any given temp

Mnemonic: ACL --> Aobs over CL
What three factors affect how much plane polarized light is rotated?
1. The identity of the molecule doing the rotating (gives specific rotation)
2. The concentration of the molecule in the solution
3. The length of the tube through which the light is passing (more length means it hits more molecules and gets rotated more)
Amides, Esters, and anhydrides are produced from by ____ reactions?
Condensation: these molecules are produced by bringing together two molecules and losing a small one, in the case of these reactions, the small molecule lost is water.
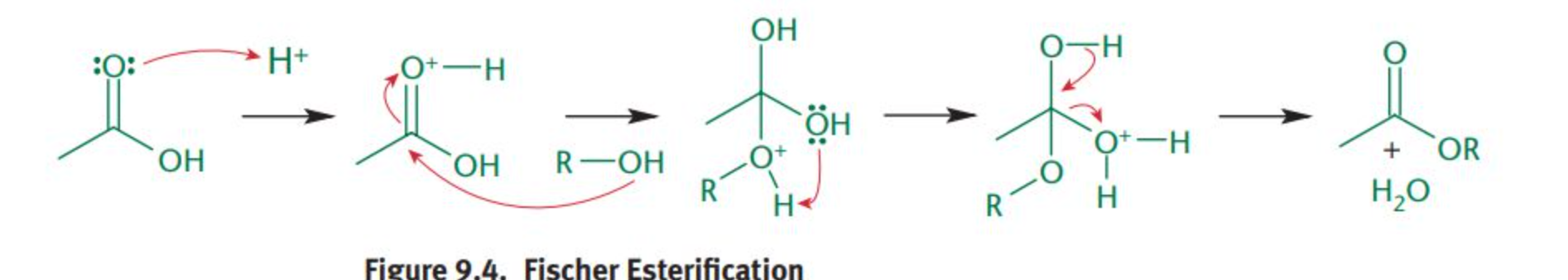
fischer esterification
a reaction in which carboxylic acids are activated by acid and reacted with alcohols to produce esters. This mechanism follows the normal nucleophilic acyl substitution mechanism.
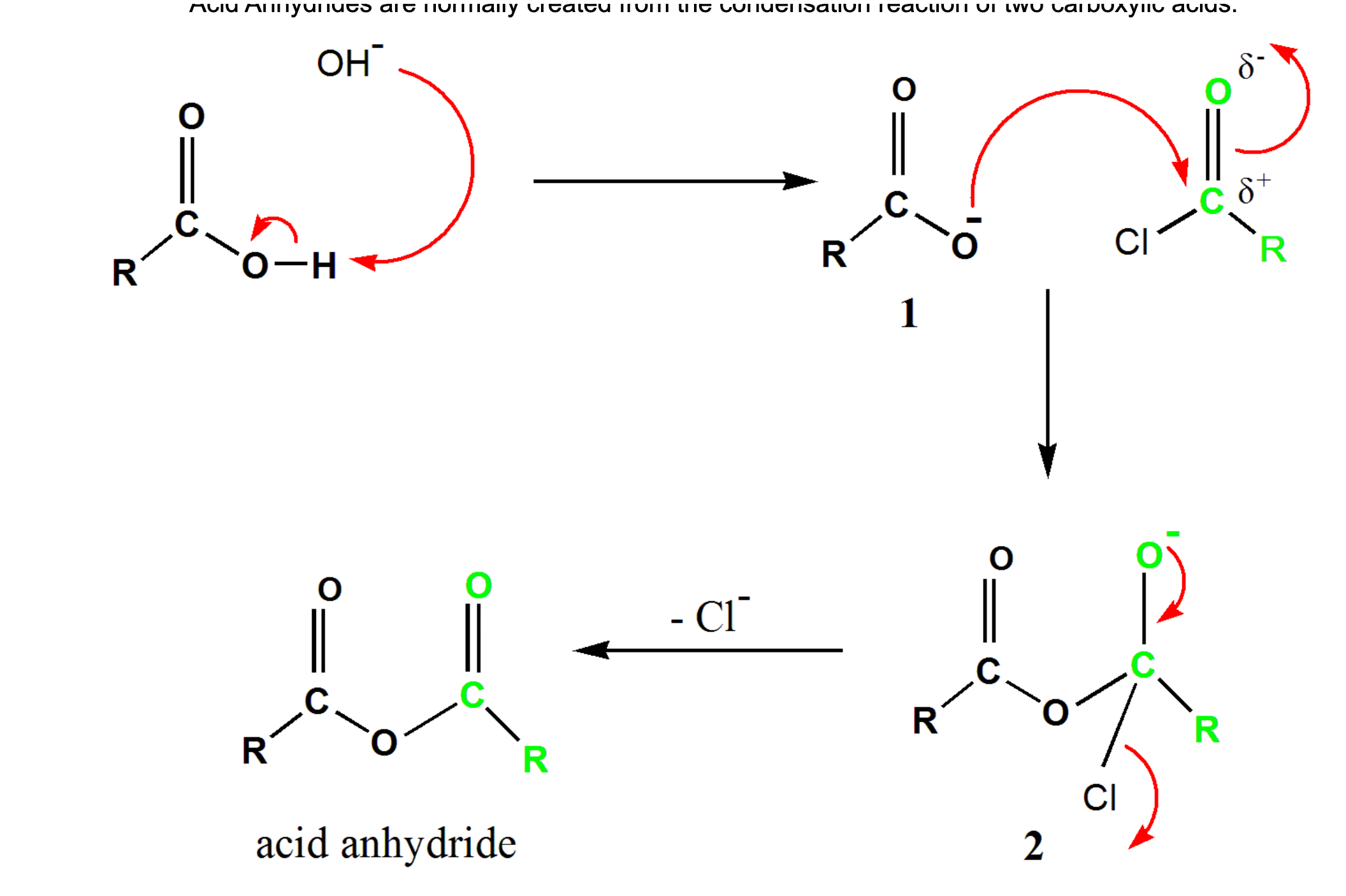
How are acid anyhdrides usually formed?
Acid Anhydrides are normally created from the condensation reaction of two carboxylic acids.
Describe intramolecular anhydride formation.
In molecules with aromatic rings and carboxylic acids, the carboxylic acids can attack one another to form anhydrides. Driven by heat.
How does ring strain affect lactams?
Depending on the number of substituents in a ring (5 and 6 are the only real stable ones) the ring strain imparted on a molecule can severely weaken its stability. Even the amide bond, a very strong bond normally, can be easily cleaved if it is shoved into an awkward ring. This means they are more reactive to hydrolysis or anything that involves breaking the ring
Describe the cleavage of anhydrides.
Anhydrides can be cleaved with various nucleophiles using the standard nucleophilic acyl substitution mechanism. Note alcohols can do this too (which will result in the formation of esters and carboxylic acids). Waters can also split into two carboxylic acids
What is the basic approach to a stoichometry problem?
How much volume does one mole of an ideal gas take up at STP?
22.4 L
how to determine the limiting reactant
In a reaction with two reactants, you can convert the moles of one reactant into the moles of another reactant using normal stoichiometric methods. So what you do is...
1. Identify the moles of both reactants.
2. Pick one of the reactants, it does not matter which.
3. Use the balanced equation to convert the moles of the reactant you picked into the moles of the other one.
4. Now compare the converted moles of the reactant to the original mole of the other reactant. The smaller number is the limiting reactant.
theoretical yield
Theoretical yield is the maximum amount of product that can be generated according to the balanced equation. This number is the yield associated with 100% of the limiting reactant reacting, absolutely no side reactions of any kind occuring, and no accidental loss of product during collection.
percent yield
Percent yield is the ratio of the actual yield to the theoretical yield multiplied by 100%.

In practical terms, a Percent Yield of 90% means that you obtained 90% of the absolute maximum value you could have gotten if you did everything perfectly.
naming ionic compounds
Remember, ionic compounds will always have a cation and an anion.
1. Write the cations name as is (Unless it is an element that can form more than one stable positive ion, in which case you denote what its charge is with parentheses and roman numerals.
2. Write the anion's name. If it is a monoatomic (One type of atom, O2-, F-) anion drop the ending of element and add -ide.
If it's a polyatomic ion, just write the polyatomic anion's memorized name.

Transition metal charges can be identified with roman numerals, but they can also be identified with suffices. What are these suffices?
For transition metals with two stable ions, the suffix -ous is used to refer to the lesser charge ion and the suffix -ic is used to refer to the higher charge ion.

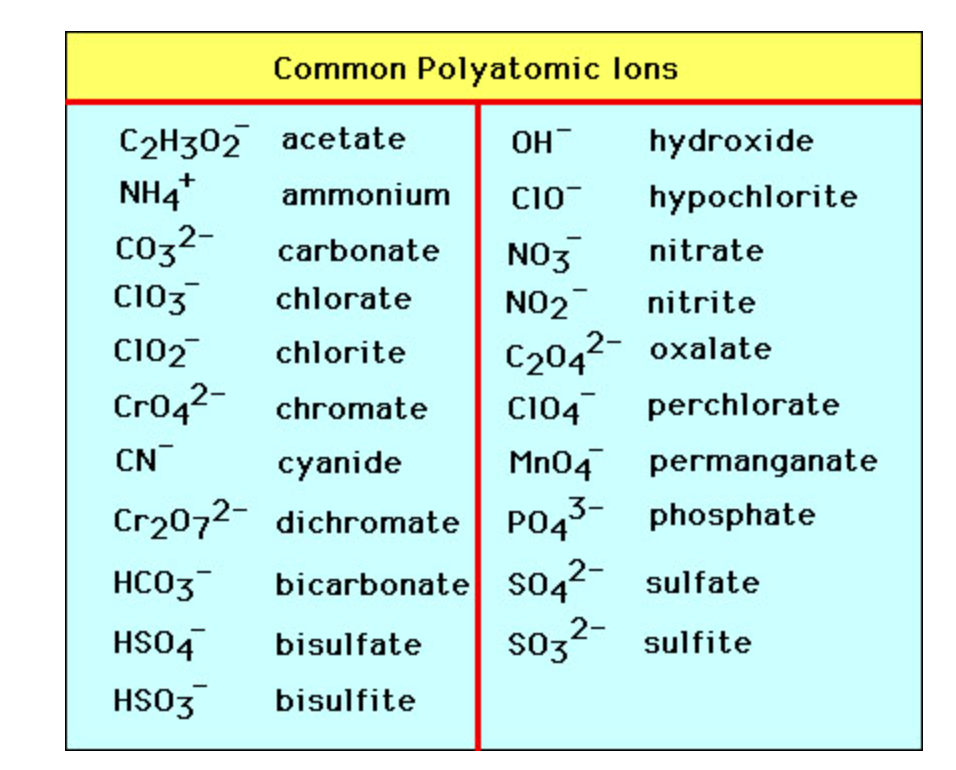
polyatomic ions
oxidation state
Oxidation state is the charge an element will usually take if it is going to form an ion and become part of an ionic compound.
For most elements, this will be the charge assocated with an octet configuration. For example, all of the Alkalie Metals (Na, K, Li) will always have an oxidation state of +1 in ionic compounds. The same is true for some Halogens and their -1 oxidation state.

Then, there are some elements that are usually going to have the oxidation number associated with their octet configuration, but not always.

What does it mean for eukaryotic mRNA to be monocistronic?
mRNA that can only be translated into one protein product. That means every single on of the thousands of proteins we contain has its own mRNA strand.
What does it mean for prokaryotic mRNA to be polycistronic?
Polycistronic mRNA is mRNA that can be translated into more than one protein product. This means that 10 different proteins in a bacteria may all be made from the same strand of mRNA. This is possible because you can get a different protein product if you don't translate all of the mRNA or don't start the translation in the same place.
tRNA
tRNA is responsible for translating the language of nucleic acids into amino acids at the ribosome. Each tRNA molecule contains a folded strand of RNA that includes a three nucleotide anticodon and an amino acid. The anticodon of tRNA binds to the codon of mRNA and brings with it the appropriate amino acid.
What is aminoacyl-tRNA synthetase?
Aminoacyl-tRNA Synthetases are a class of enzymes that attach specific amino acids to specific tRNA molecules (based on the anticodon). Each type of amino acid is activated by a different aminoacyl-tRNA synthetase. Amino Acids are always added to the free hydroxyl at the 3' end of the tRNA.
The new bond is high in energy which is used to make a peptide bond in translation
rRNA
rRNA is synthesized in the nucleolus and functions as an important structural component of ribosomes (and catalyzes the formation of peptide bonds in the growing AA chain). rRna molecules can also act as ribozymes. (detail: can also splice out its own introns)
ribozyme
a strand of RNA with enzymatic properties
3 stop codons
UAA - u are annoying
UAG - u are gone
UGA - u go away
3 types of point mutations
1. Silent Mutation
2. Missense Mutation
3. Nonsense Mutation
octet rule
to have a full octet of electrons in the outermost shell is the most stable state for an atom, and atoms will give/take/share valence electrons in such a way so as to ensure that both (or more) atoms in the compound have noble gas configurations
3 exceptions to octet rule
Incomplete Octet Elements: These elements are stable with less than eight electrons in their valence shell. Hydrogen (2), Helium (2), Lithium (2), Beryllium (4), and Boron (6).
Expanded Octet: Any element in period 3 and greater can hold more than eight electrons. For example, Phosphorus (10), Sulfur (12), Chlorine, (14) and others.
Odd numbers of Electrons: Any molecule with an odd number of valence electrons cannot distribute those electrons to give 8 to each atom, so one of the atoms won't have a full octet in these molecules.
ionic bond
= bond where an electropositive atom donates all its valence electrons to an electronegative atom e.g. NaCl (generally between metals and nonmetals)
Form crystal lattices where each molecule is attracted to many others via electrostatic interactions
covalent bonds
This is usually how nonmetals bind other nonmetals because both of the nonmetals in the bond want to gain electrons.
Dont form crystal lattices, compounds consist of individually bonded molecules
coordinate covalent bond
A coordinate covalent bond is when one atom contributes both of the electrons that the atoms will share to form their covalent bond.
When given a molecule, consider how it was formed. Ex. how many coordinate bonds in NH4+? We know that NH3 has one lone pair so to make NH4+ that lone pair would contribute both electrons to an H+. It wouldnt split the lone pair with a regular H to make a nitrogen with a free radical

characteristics of ionic bonding
1. Ionic compounds have very high melting and boiling points.
2. Ionic compounds dissolve readily in water and other polar solvents.
3. Ionic compounds form crystalline lattice structures so as to maximize the attractive force between atoms of opposite charges and minimize the repulsive force between atoms of same charges.
crystal lattice
the way the atoms of an ionic bond will situate themselves in a group so as to minimize the energy of the system and stabilize the compound.
characteristics of covalent compounds
Covalent compounds contain discrete molecular units with relatively weak intermolecular interactions, and therefore lower boiling points and melting points. They do not break down into constituent ions, and are therefore poor conductors of electricity.
How big must the difference in electronegativity be to be considered a polar bond?
Any electronegativity difference between 0.5 or 1.7 will seperate charge enough to create a polar bond.
Metals ___ electrons to achieve a stable octet
Metals lose to achieve a stable octet