all year 2 diabetes
1/545
Earn XP
Description and Tags
excludes the affecting metabolism deck
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
546 Terms
Where is extracellular calcium stored and what is the optimum concentration of it?
In the plasma and extracellular fluid and around 1.25mM.
What hormones control calcium level?
- Parathyroid hormone.
- Calcitonin
- Vitamin D
What happens if Ca2+ conc is low (Hypocalcaemia)?
- Increases muscle excitability.
- Can cause muscle spasms which can be fatal if respiratory muscle spasm.
How does hypocalcaemia increase muscle excitability?
Low Ca2+ levels increases permeability of Na+ which increases the movement of action potentials down a neuron (depolarises neuron)
What happens if Ca2+ conc is high (Hypercalcaemia)?
- Decreases muscle excitability.
- Can cause cardiac arrythmias.
How does hypercalcaemia decrease muscle excitability?
High Ca2+ levels decreases permeability of Na+ which decreases the movement of action potentials down a neuron (hyperpolarises neuron)
Where is most of calcium in the body stored?
99% of calcium is stored in bones and teeth.
Describe the structure of a cortical bone.
- Compact and dense in appearnace.
- Hollow as it contains bone marrow.
- Contain repeating circular units called osteons.
- Blood vessels run through the Central Canal osteons.
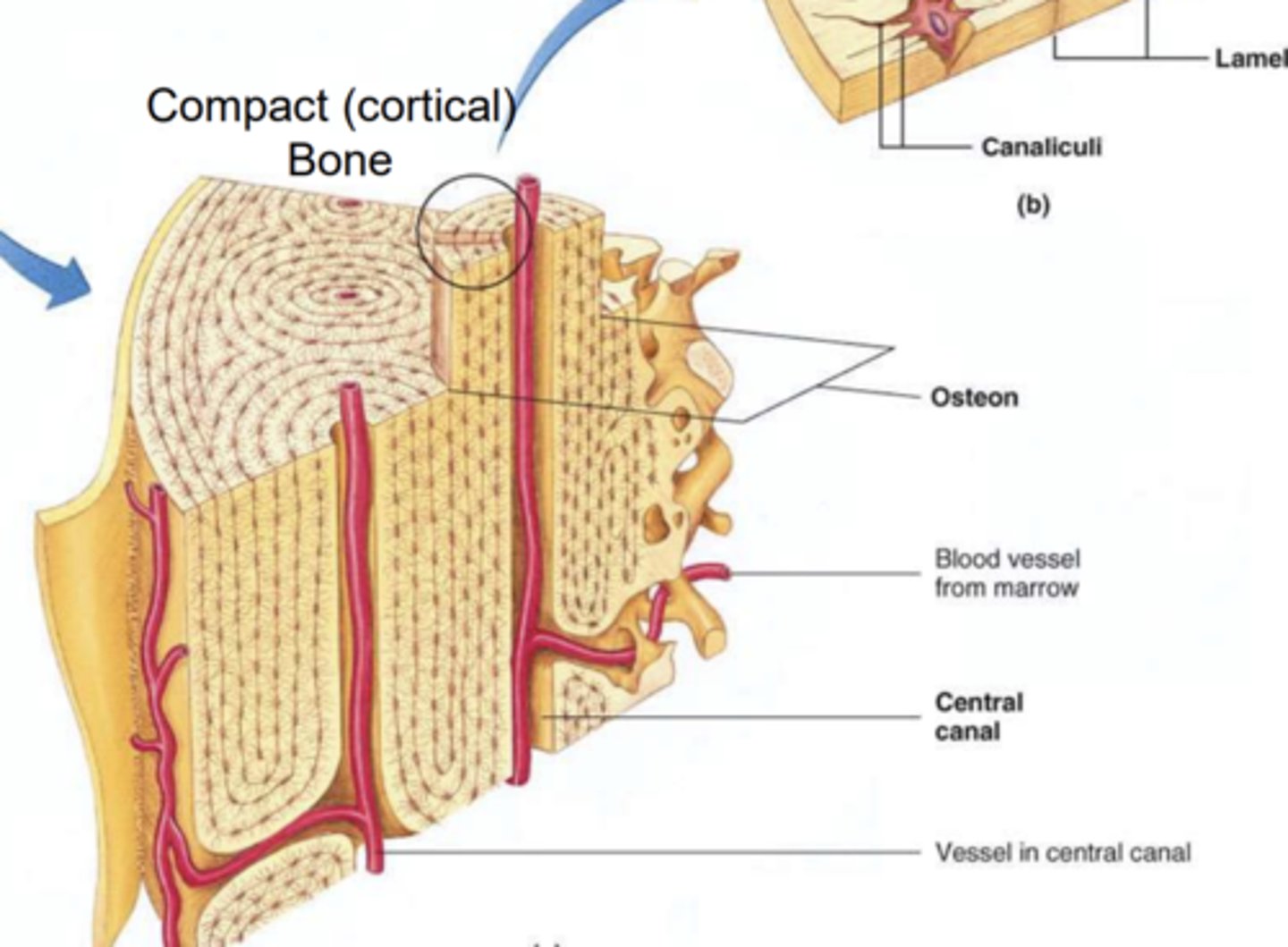
Describe the structure of osteons.
- Made up of osteocytes and network of blood vessels called canaliculi.
- Also contain lamella which are layers of cells that surround the central canal
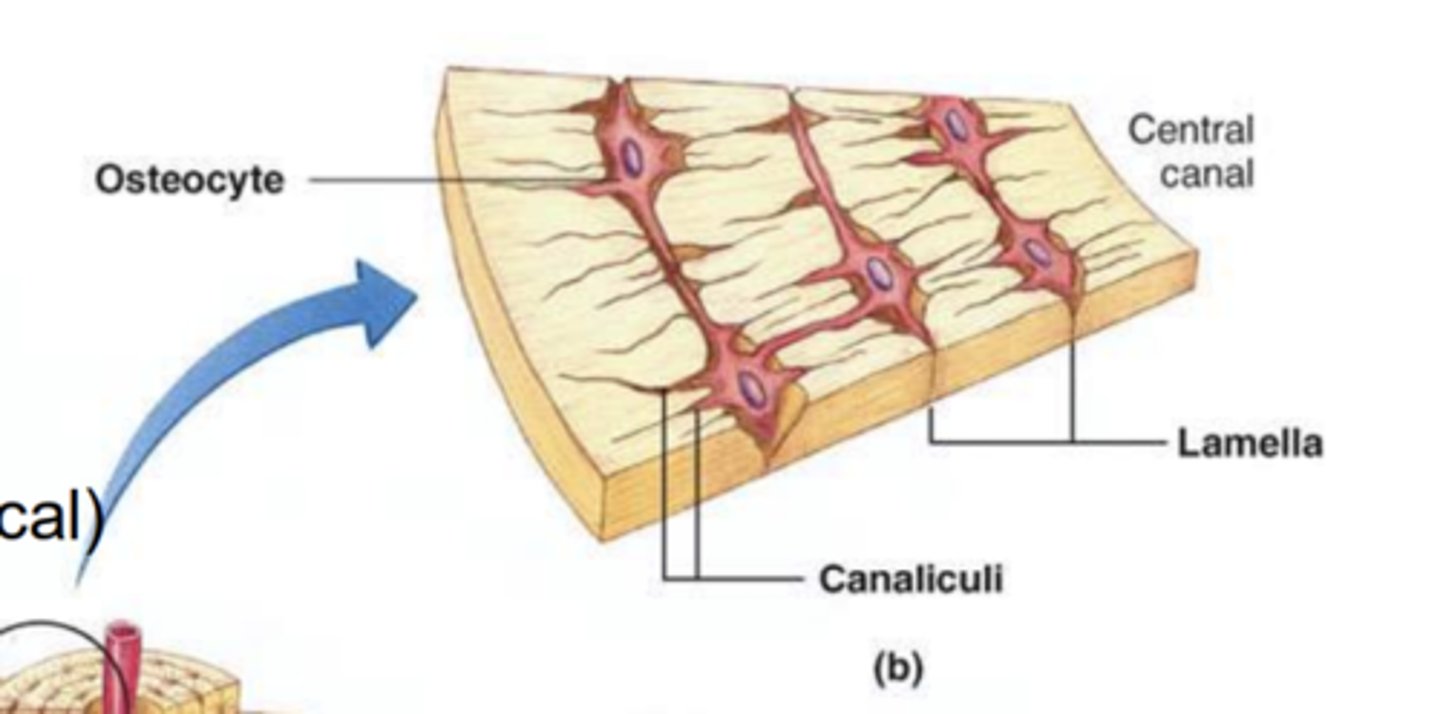
What do the canaliculi transport?
The extracellular fluid AKA bone fluid.
Describe osteoblasts.
- Bone forming cells.
- Form connections with osteocytes (osteocytic-osteoblastic bone membrane).
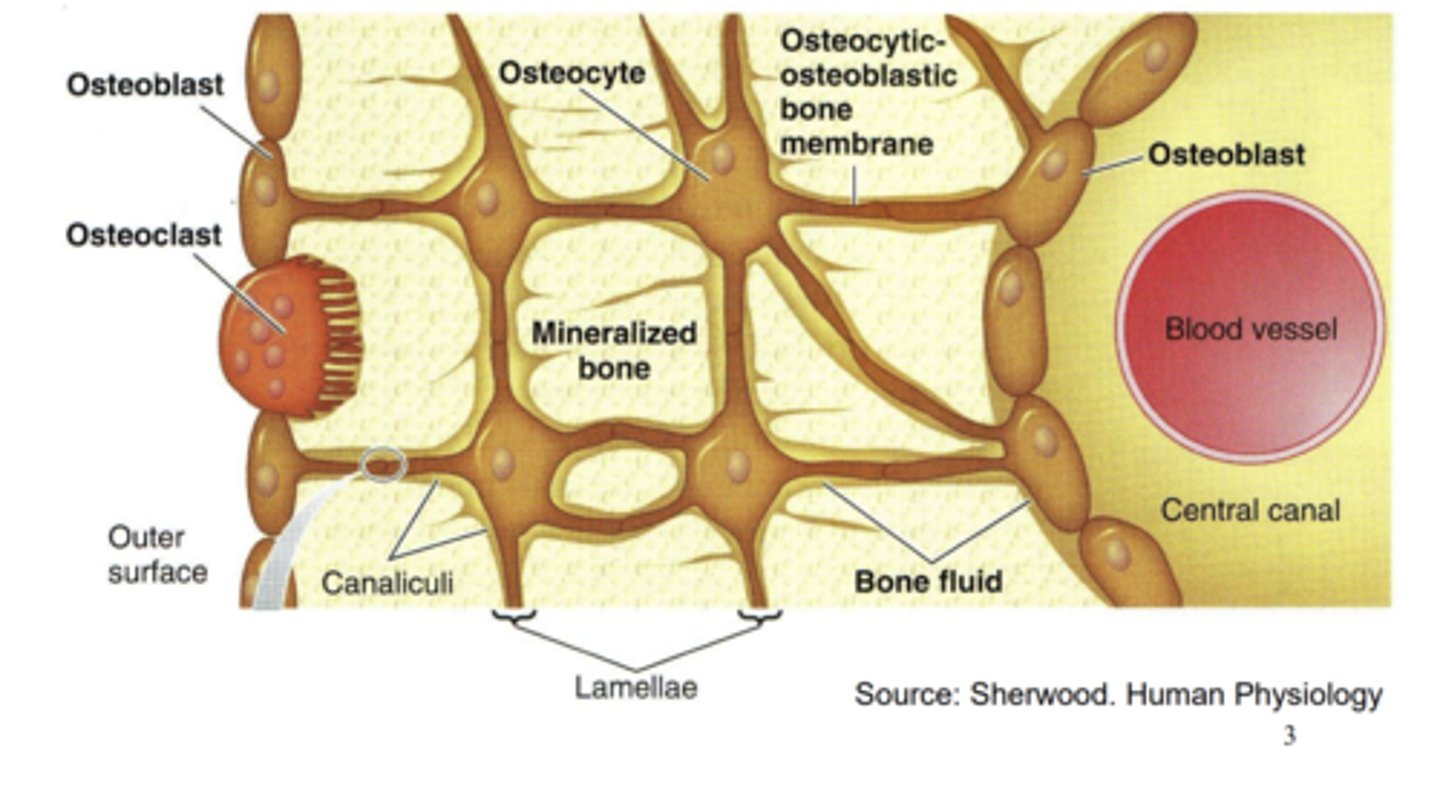
Describe osteoclasts.
- Sit on the outside of the bone and are multinucleated.
- Responsible for breakdown of old bones.
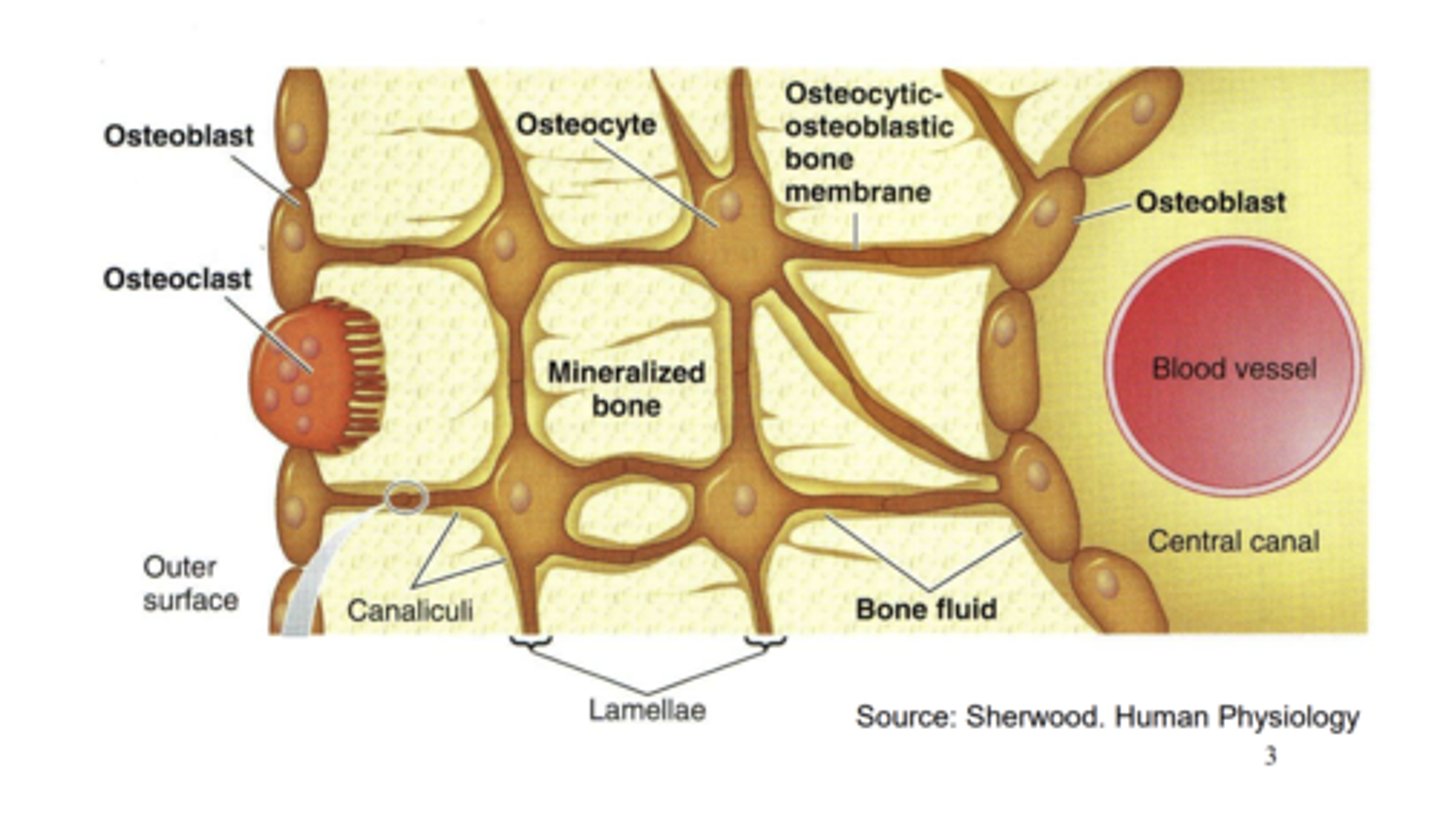
How do osteoclasts dissolve the bone?
- Secrete HCl which dissolves the mineralised bone.
- Secretes Cathepsin K which breaks down collagen.
What is mineralised bone made of?
- Majority is made of hydroxyapatite which contains high levels of Ca2+ and PO43-
- Contain an extracellular matrix of collagen called the osteoid.
Describe bone turnover.
- Osteoclasts dissolve the bone (resorped).
- Osteoclasts then die or migrate which allow osteoblasts to fill the cavity (deposition)
- Bone is deposited and resorped at the same rate.
How do osetoblasts deposit into bone?
- They secrete new osteoid which have lots of Ca2+ and PO43-.
- This helps develop the mineralised bone.
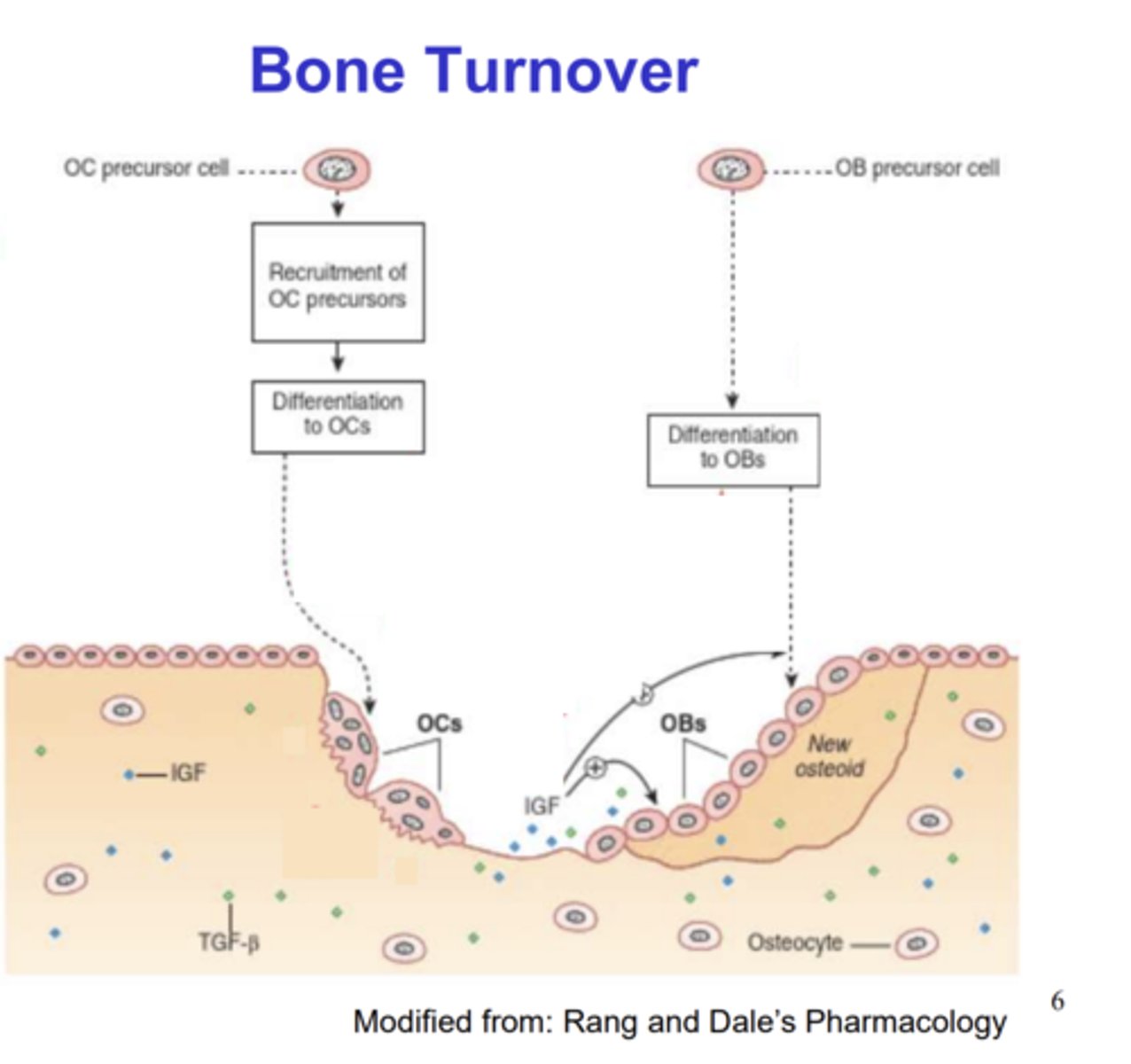
How do osteoclasts (OC) attach to bone?
- OC precursor cells are recruited and then differentiate into OCs.
- OCs then attach to the surface of the bone to dissolve it.
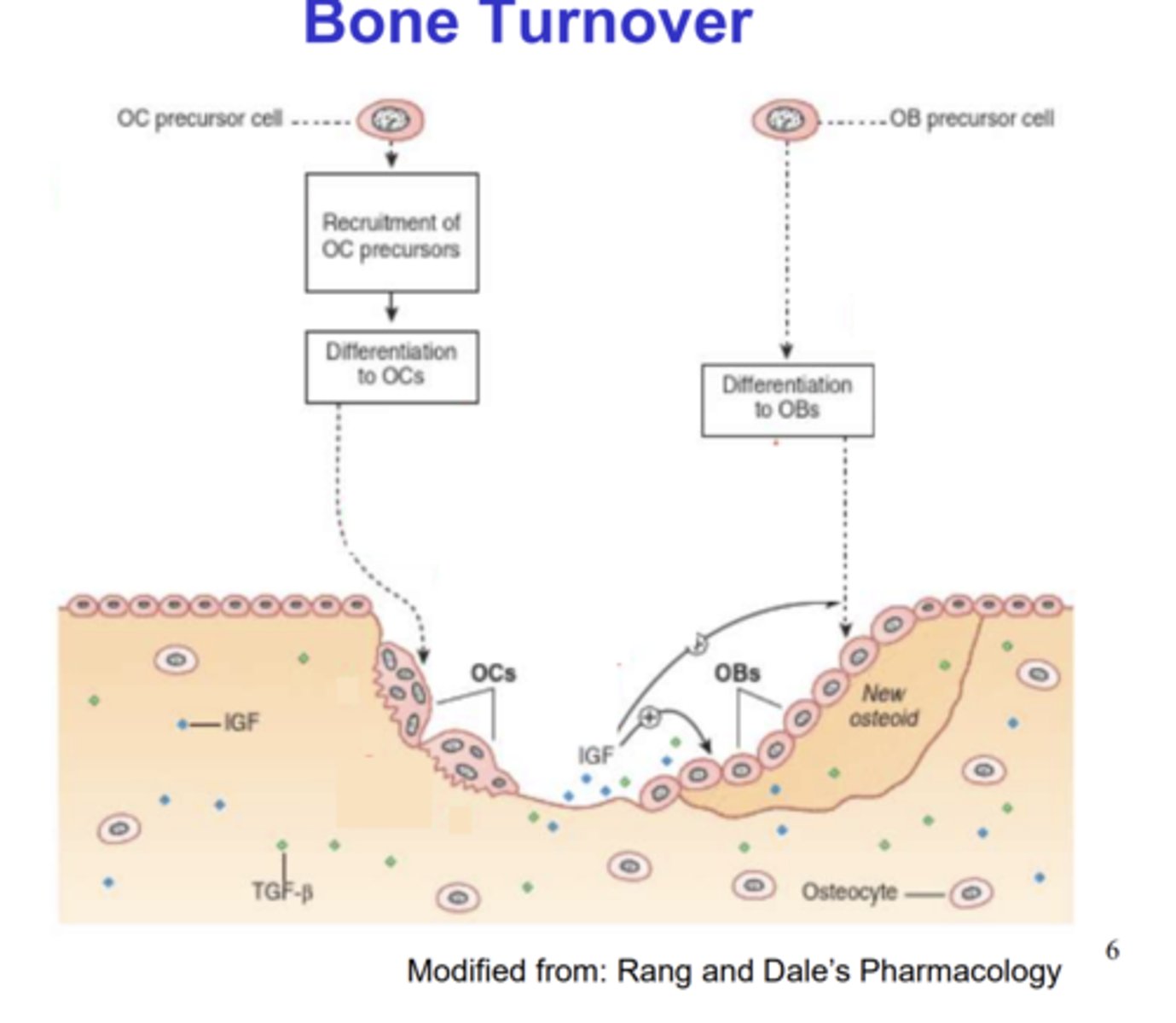
How do osteoblasts (OBs) attach to the bone?
- Sequestered growth factors such as IGF and TGF-β are released from the bone when OCs dissolve the bone.
- IGF and TGF-β encourage differentiation of recruited OBs.
- This allows OBs to attach to the bone and stimulate deposition.
How are osteoclast numbers regulated?
Controlled by RANKL and Osteoprotegrin (OPG).
Balance of these 2 factors is essential for bone density.
How does RANKL regulate osteoclast numbers?
- It attachestes to OCs which increases OC differentiation and decreases OC apoptosis.
- This increases their number and promotes bone resorption over time.
How does Osteoprotegrin (OPG) regulate osteoclast numbers?
- Acts as a decoy receptor for RANKL preventing it from binding to OCs.
- This decreases OC numbers and promotes bone deposition over time.
What hormones are involved in Ca2+ control?
- Parathyroid hormone (PTH)
- Calcitonin
- Vitamin D
Describe parathyroid hormone.
- A peptide hormone which is secreted from the parathyroid gland.
- Secretion increases when extracellular Ca2+ decreases.
- Causes Ca2+ conc to increase.
How does the parathyroid gland monitor Ca2+ levels?
- Parathyroid cells monitor directly via GPCRs.
- Negative feedback to maintain levels.
Where is the parathyroid located?
Behind the thyroid
What effect does PTH have on the bone?
- Causes release of Ca2+ from the bone to increase Ca2+ levels.
- Occurs in 2 phases, Fast exchange and Slow exchange.
Describe the fast exchange of Ca2+ release from bone via PTH.
- PTH attaches to PTH GPCRs on the osteoblasts and osteocytes.
- This activates the Gs subunit and increases cAMP conc.
- This causes Ca2+ to move into the osteocytes and osteoblasts from the canaliculi.
- Gap junctions within osetocytes and osteoblasts allow Ca2+ to move into the plasma to increase plasma conc.
- This is a very rapid process.
Describe the slow exchange of Ca2+ release from the bone via PTH.
- PTH activates osteoblasts which increases RANKL expression.
- This increases osteoclast numbers (and inhibits Osteoblast deposition) which therefore increases bone resorption.
- This is a slower process which causes Ca2+ conc to increase.
- Also increases PO43- levels.
When is the fast and slow exchange of Ca2+ activated?
The slow exchange is activated when condition of hypocalcemia is prolonged and fast is when hypocalcemic conditions are temporary.
What effect does PTH have on the kidneys?
- Promotes Ca2+ reabsorption.
- Promotes PO43- excretion.
- Activates vitamin D
Describe calcitonin.
- A peptide hormone which causes Ca2+ conc to decrease.
- It is secreted from C cells in the thyroid gland.
- Attaches to GPCRs on osteoclasts.
How does calcitonin decrease Ca2+ levels?
- Binds to GPCRs on osteoclasts which inhibits osteoclast resorption.
- This prevents Ca2+ being released and therefore decreasing its movement from the canaliculi to the plasma.
- Inhibits reabsorption of Ca2+ in the kidney.
Describe vitamin D.
- A pre-hormone that increases Ca2+ absorption from the food in the GIT.
- Synthesised in the skin in response to sunlight.
- Also absorbed from diet in dair products.
How is vitamin D activated?
- One OH group is added in the liver.
- A second OH group is then added by the kidney, caused by PTH.
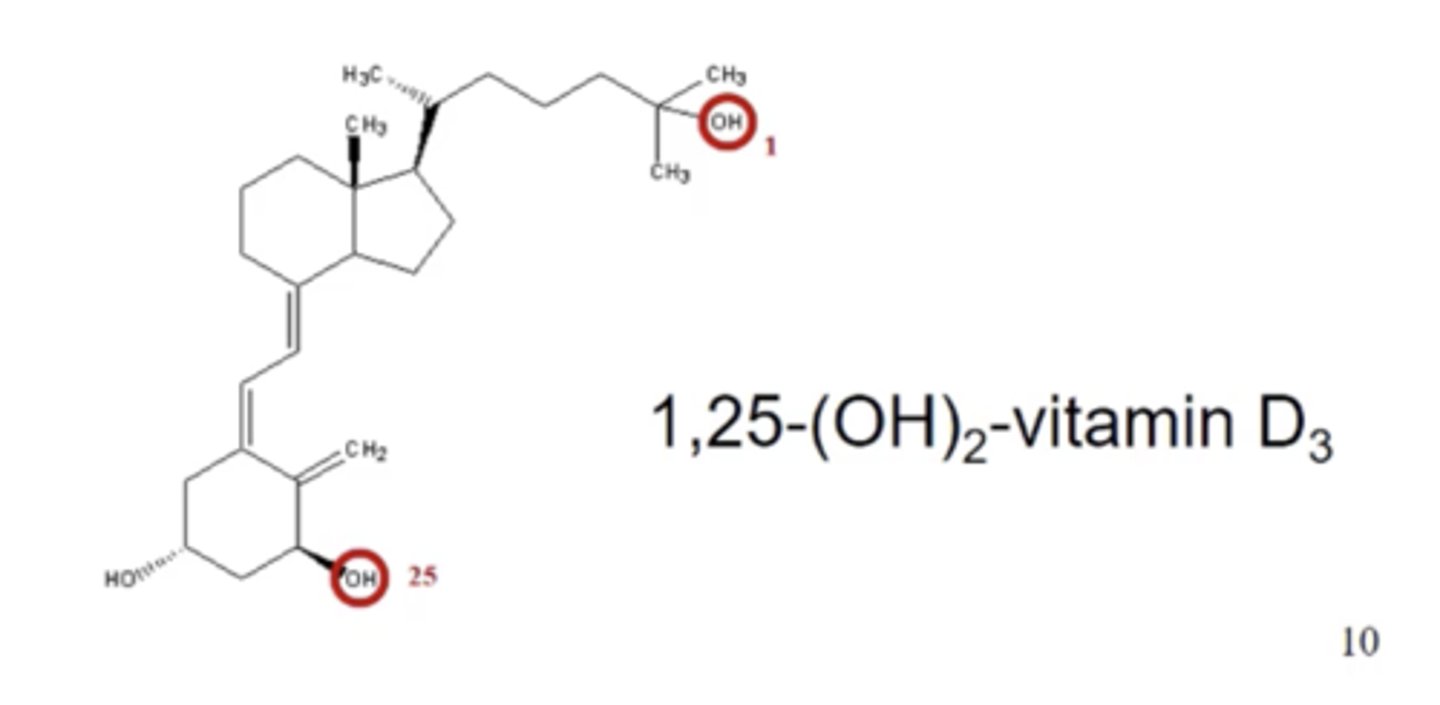
How does activated vitamin D increase Ca2+ conc?
- Binds to nuclear receptors which promote expression of specific genes.
- These genes caused increased absorption of Ca2+ in the GIT.
- It also increases reabsorption of Ca2+ in the kidney and works with the PTH to increase release of Ca2+ from bone.
- All of this helps to increase Ca2+ levels.
Describe the mechanism of action of Acarbose.
- Mimics the oligosaccharides formed from starch as well as the transition state of the enzyme α-glucosidase.
- Acts as a competitive inhibitor of α-glucosidase.
- This inhibits hydrolysis of starch and therefore reduce absorption of glucose.
What is α-glucosidase?
An enzyme which breaks down starch
How is acarbose produced?
Via fermentation from a strain of Actinoplanes.
What are the side effects of Acarbose?
Flatulence and diarrhoea due to build up of oligosaccharides in the colon.
Why are acarbose very useful?
- Effective antidiabetic drug.
- Their effect is independent of pancreatic insulin secretion.
Describe PPAR.
- PPARs are transcription factors which attach to PPAR receptors.
- Upon binding PPAR translocate to the nucleus where they form heterodimers with RXR.
- The dimers bind to DNA and activate transcription of adjacent genes.
- Activation of PPARγ is associated with beneficial outcomes in diabetes.
Describe PPAR agonists.
PPARγ agonists improve insulin sensitivity by enhancing glucose uptake in peripheral tissues such as skeletal muscle and adipose tissue.
Describe Pioglitazone.
- A type of PPARγ agonist.
- Often used in combination with metformin, sulfonylureas and insulin.
- Does not cause hypoglycaemia which is positve.
- Effect is independent of insulin secretion.
What are the side effects of pioglitazone?
Associated with weight gain and fluid retention.
What is SGL2 and how do they work?
- A major transporter that reabsorbs glucose in the kidney (PCT).- Na+ ions are pumped out of PCT whilst K+ ions are pumped in which incerases the conc gradient of Na+ outside the cell.- This allows Na+ and glucose to be co-transported back into the PCT which allows glucose to move into the blood.
How do SGLT2 inhibitors work e.g., dapagliflozin?
- Block the SGLT2 transporter which prevents reabsorption of glucose (and Na+).- This causes glucose to be excreted in the urine therefore plasma concentration of glucose is reduced.
Why can glucose still be reabsorbed if SGLT2 inhibitors are being used?
Only selective to SGLT2 transporters so SGLT1 transporters can still reabsorb glucose in the kidneys and gut.
Why are SGLT2 inhibitors useful?
- Likely to promote some weight loss due to glucose reabsorption.
- Not associated with hypoglycaemia.
- Effect independent on insulin.
What are issues with SGLT2 inhibitors?
- Results in glycosuria >50g/day.
- Potential for side-effects due to glycosuria e.g., tiredness, dehydration, UTIs.
What is a carbohydrate?
A molecule which has a carbon chain with hydroxyl molecules and an aldehyde (aldoses) or ketone group (ketoses).
What are the uses of carbohydrates?
- An energy source, glucose.
- Structure, cellulose cell walls.
- Glycoproteins on plasma membrane.
- Medicine, clarithromycin.
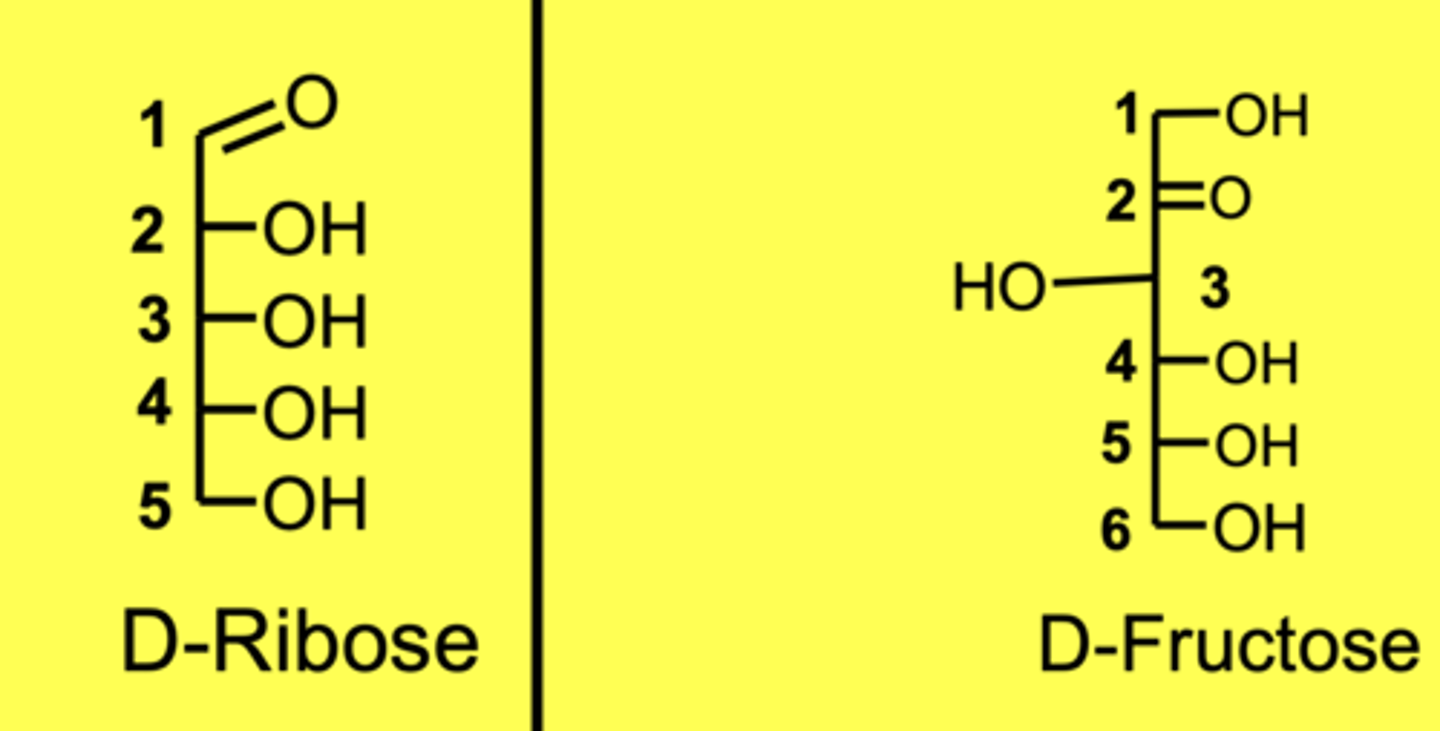
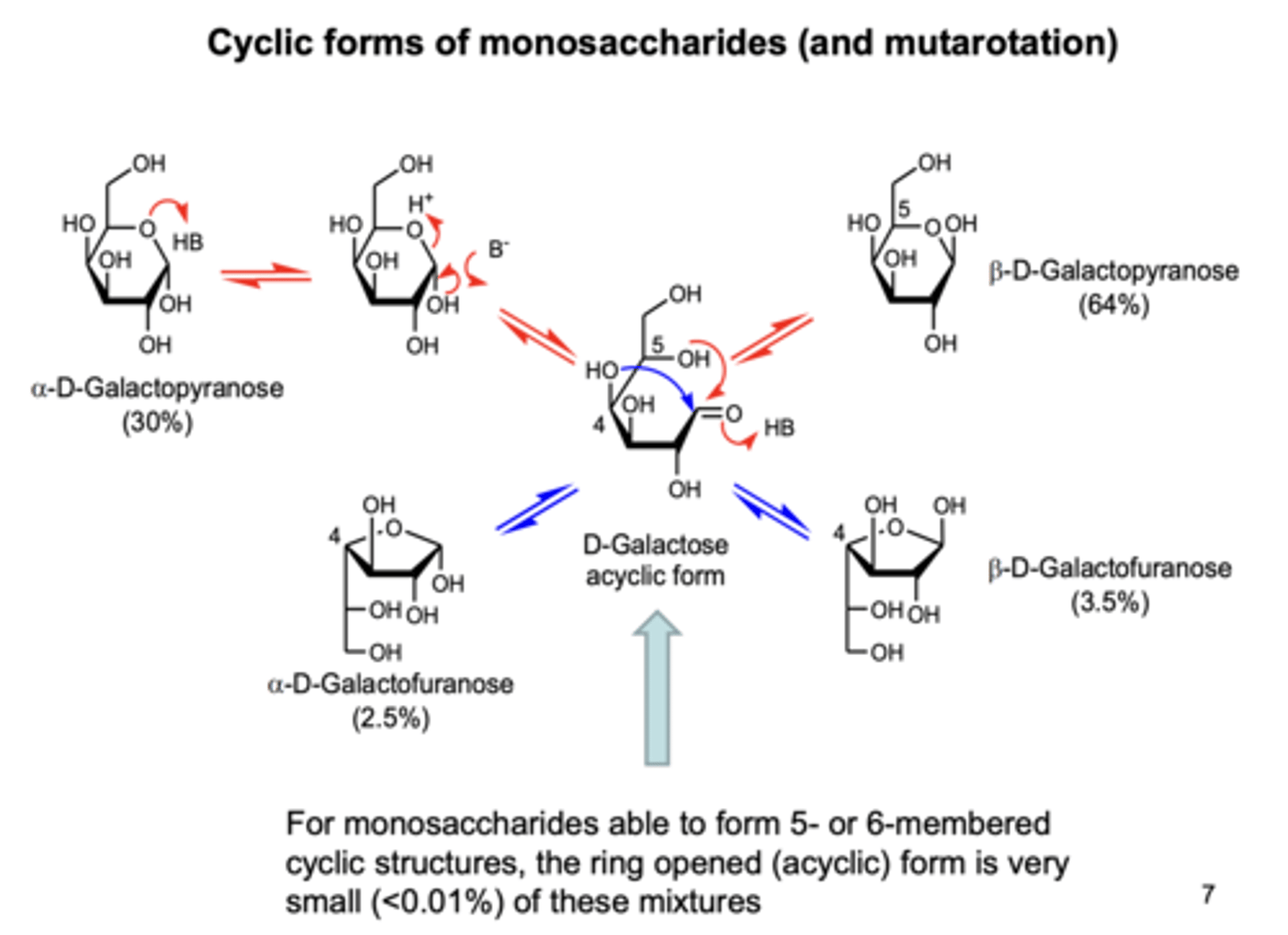
Show the interconversion between cyclic forms of monosaccahrides.
What is a lipid and give an example?
Molecules which can be isolated from organisms via extraction with a non-organic solvent (hexane).
For example, Fats and oils.
What are the two classes of lipids and give examples of each?
- Hydrolysable e.g., TAGs
- Non-hydrolysable e.g., steroid hormones
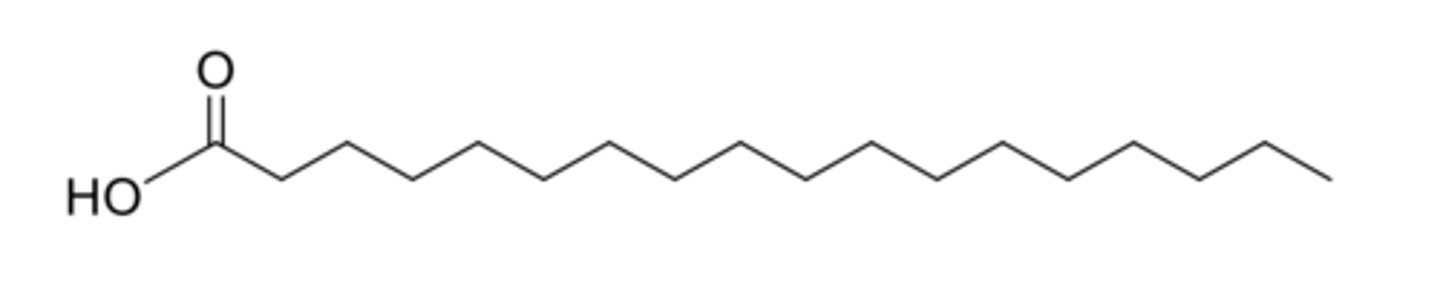
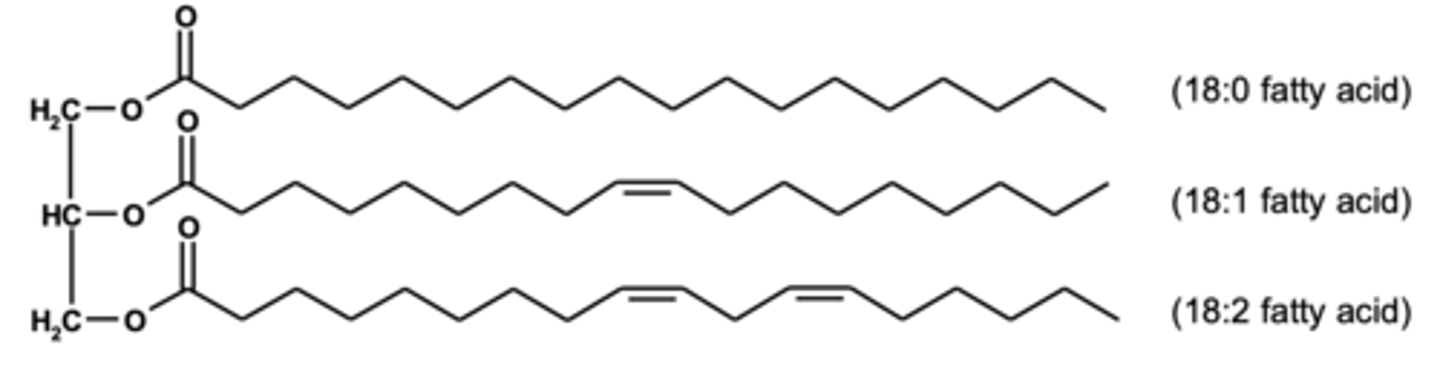
Describe saturated fatty acids.
- Contain no double bonds.
- Generally unbranched.
- Contain an even number of carbon atoms.
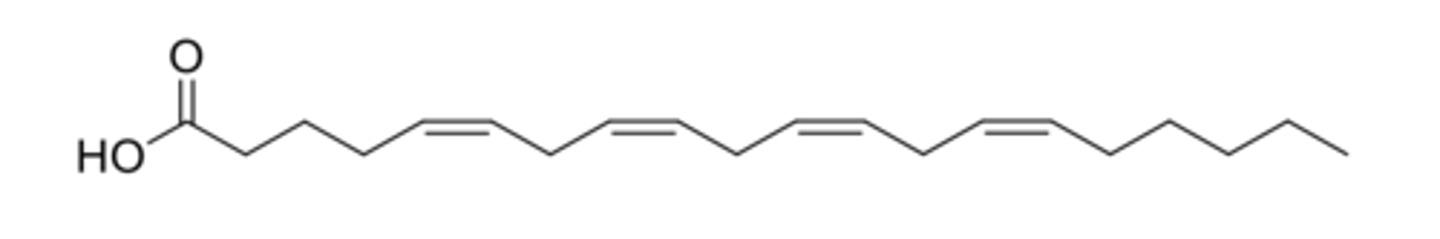
Describe unsaturated fatty acids.
- Contain double bonds.
- Almost exclusively Cis (Z) isomers.
What are the uses of lipids?
- Energy store e.g., triglycerides.
- Phospholipid membranes.
- Messenger lipids e.g., hormones.
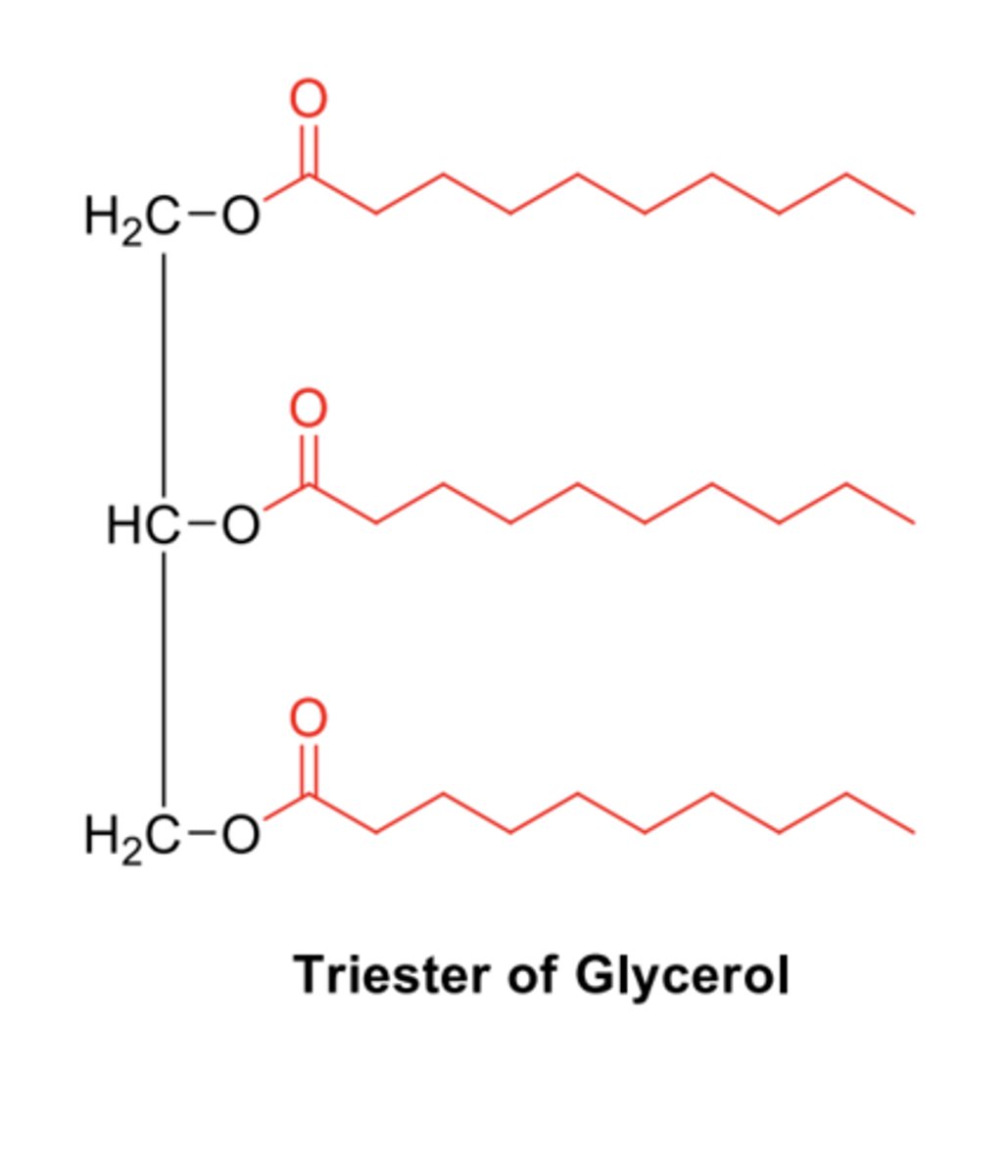
Describe triacylglycerols (TAGs).
- Triesters of one glycerol and three fatty acids.
- Fatty acids don't all have to be the same so can very in length and be saturated or unsaturated.
Describe membrane lipids e.g., phospholipids.
- Contain two fatty acid tails and a polar head.
- The fatty acid chains can interact with each other via van der waals.
- Fatty acids and polar heads do not need to be identical.
Describe cholesterol in membranes.
- Fatty acid tails in phospholipids are flexible which allows cholesterol to embed in between them.
- This increases rigidity of the membrane.
How can cholesterol cause harm?
If cholesterol builds up it can cause arteries to be blocked and the development of gallstones.
What are insulin regimes?
- Regimes of insulin use which mimic insulin release in the body.
- For example, a constant low levle to maintain body tissues and a dramatic increase in response to rise in blood glucose after food intake.
What are the types of insulin regimes?
- Multiple
- Twice Daily
- Once
- Sliding scale
- Starting insulin
- Adjusting doses
Describe the multiple use regime for insulin use.
- Intermediate/long acting insulin as well as multiple short acting insulin at meal times.
- Advantages is that it is flexible if adjustments to day is required.
- Disadvantages is that it requires more injections.
Describe the twice daily insulin regime.
- Short + intermediate acting insulin is used.
- Advantages is that it is simple, provides good control and uses few injections.
- Disadvantages is that it is not flexible and timing of injections as well as time of food intake is very strict.
Describe the once insulin regime.
- Intermediate/long acting inuslin used once at night.
Often used in combination with with oral hypoglacaemics.
Describe the sliding scale regimen.
- IV used for medical emergencies e.g., unstable diabetes.
- Given by continuous IV infusion and rate is adjusted frequently according to blood glucose levels.
Describe the starting insulin regime.
- Starts with very low doses & increase very slowly.
- 6-10IU BD.
- Patients can be taught to adjust dose according to pre-prandial blood glucose.
Describing adjusting doses insulin regime.
- Pre-meal blood glucose relates to previous dose of insulin so don't change next insulin dose.
- Manage blood glucose by food intake.
How is insulin stored?
- Long-term storage in fridge.
- Current use such as a vial or pen should be outside of fridge.
- Avoid freezing as it decreases activity.
What are sick day rules for insulin use?
During illness, diabetics should:
- Not stop taking diabetes medication or insulin.
- Test blood more often as well as ketones (ketostix)
- Drink plenty of fluids.
- Replace normal meals with carbohydrate containing drinks.
What are triacylglycerols?
- Storage form of fatty acids.
- Broken down to form energy.
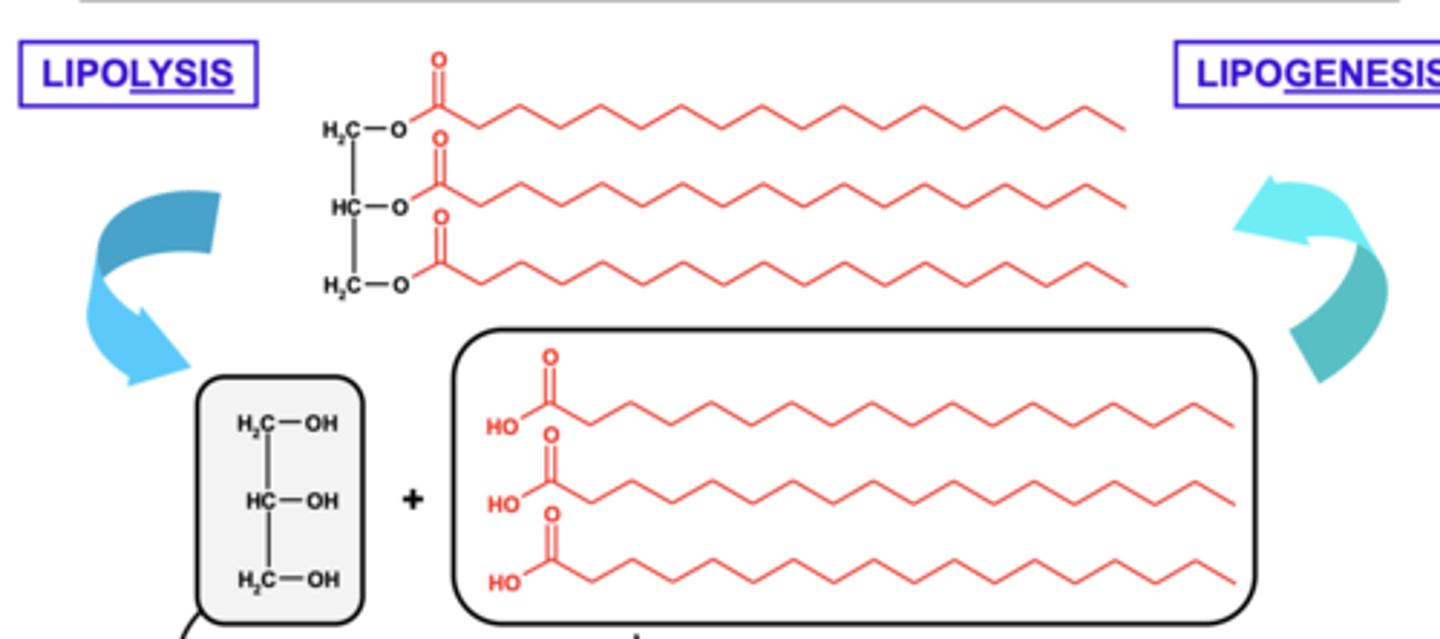
What is lipolysis?
The breakdown of triacylglycerols into fatty acids and glycerol.
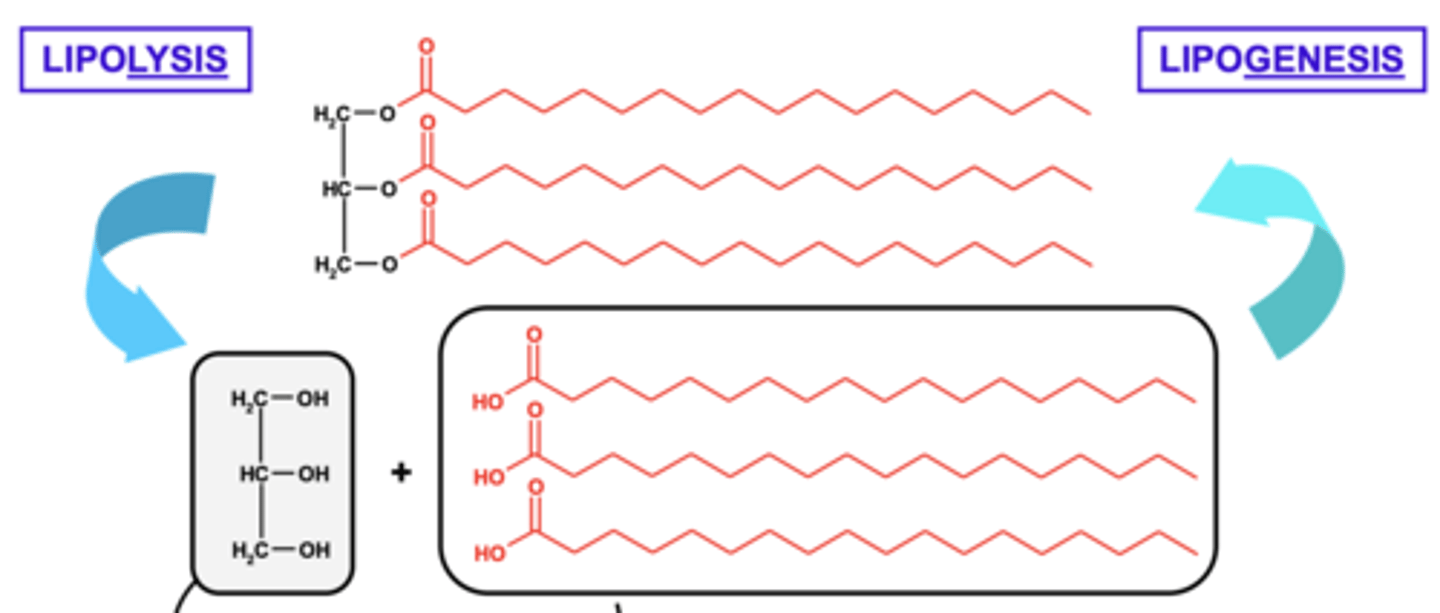
What is lipogenesis?
The formation of triacylglycerols from glycerol and fatty acids.
What are the two main ways energy is produced from triacylglycerols (TAGs)?
- Via production of dihydroxyacetone phosphate.
- β-oxidation of fatty acid chains.
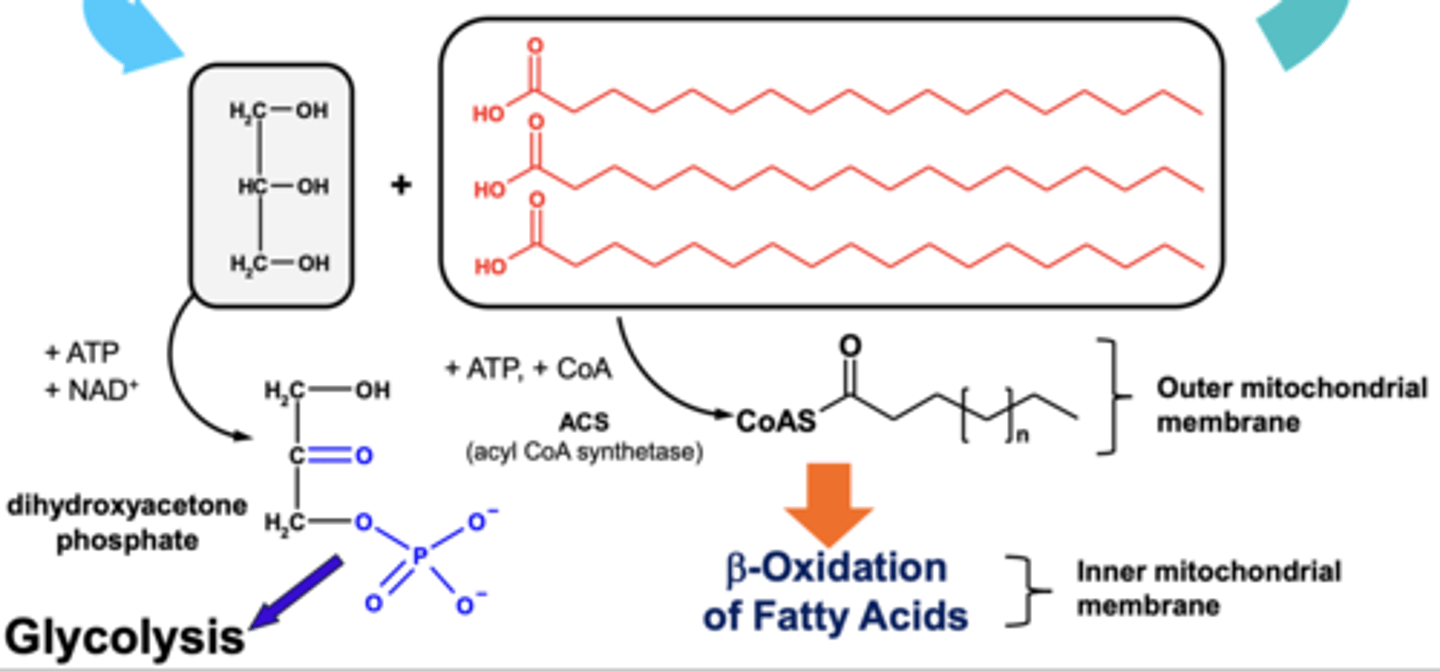
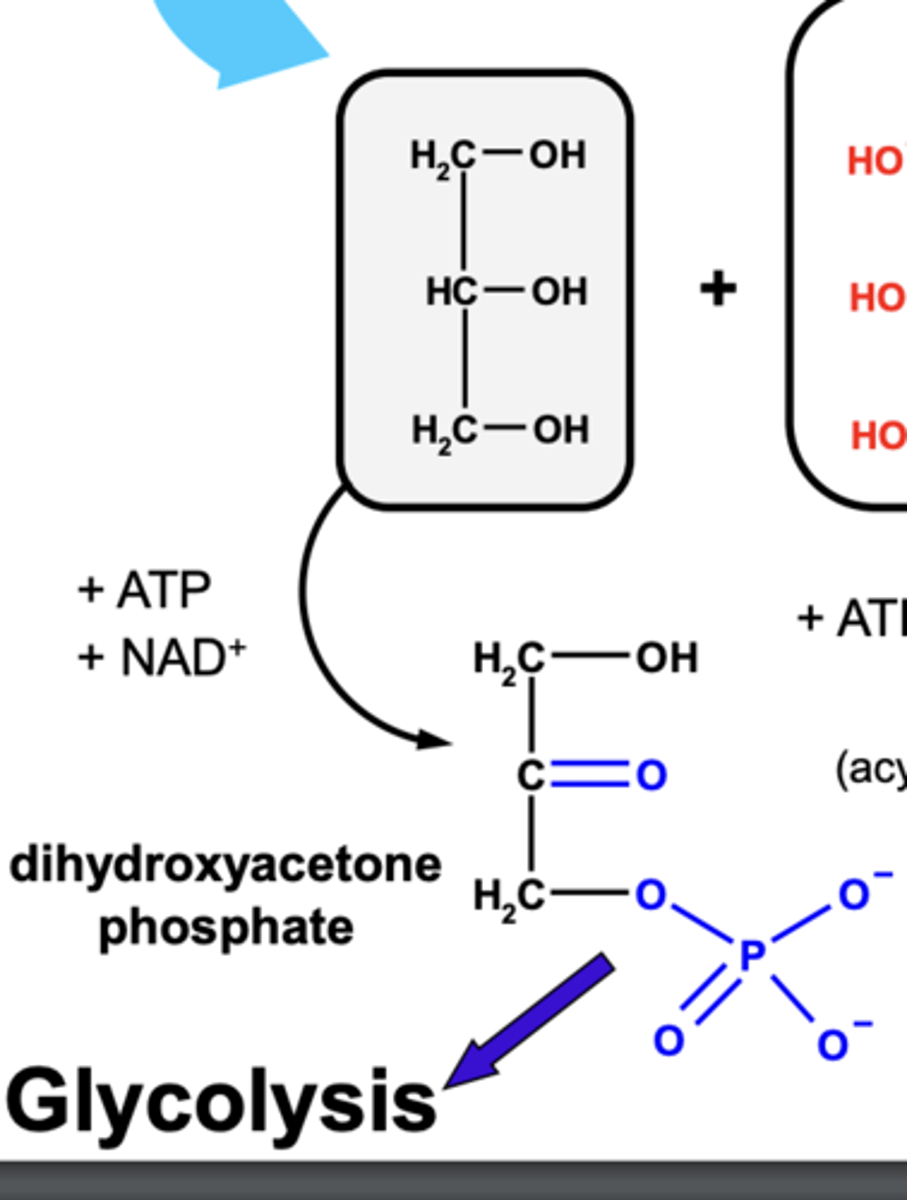
How is energy produced from TAGs via production of dihydroxyacetone phosphate (DHAP)?
- TAG's are broken down into glycerol and fatty acids.
- Glycerol is then oxidised to form a carbonyl group.
- ATP is then dephosphorylated to add a phospahte ion to the glycerol molecule.
- This form dihydroxyacetone phosphate (DHAP)
- DHAP is an intermediate in glycolysis which can be converted into pyruvate so cabohydrate metabolism can continue to produce ATP.
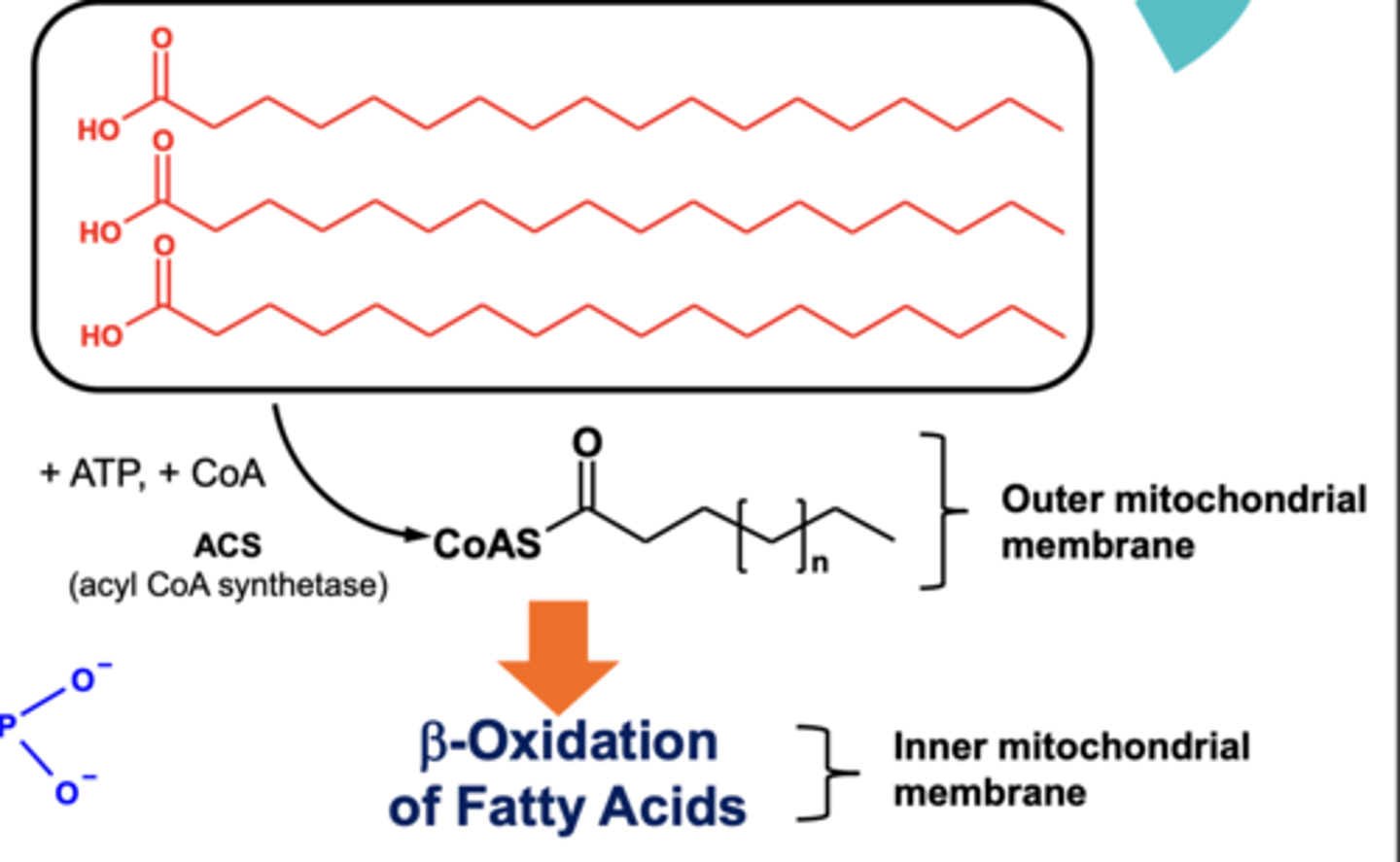
How is energy produced from TAGs via β-oxidation of fatty acids?
- TAGs are broken down into fatty acids and glycerol.
- The fatty acids are combined with CoA, using energy from ATP) in the outer mitochondrial membrane to produce a thioester.
- The thioester can now be oxidised in inner mitochondrial membrane via β-oxidation to produce ATP.
Describe β-oxidation of CoA-thioesters formed from TAGs?
- The CoA-thioester is dehydrogenated via FAD+ (FADH2).
- It is then hydrated adding an alcohol group to the molecule.
- The alcohol is then dehydrogenated to form a ketone via NAD+ (NADH).
- CoA is then removed from the molecule.
- β-oxidation repeats until the fatty acid chain no longer has 2 free alkyl carbons.
What are the products of β-oxidation of TAGs?
As the cycle repeats for the entire length of fatty acid, it produces lots of:
- Acetyl CoA that can join the link reaction of carbohydrate metabolism to produce ATP.
- FADH2 and NADH which can contribute to oxidative phosphorylation to produce ATP.
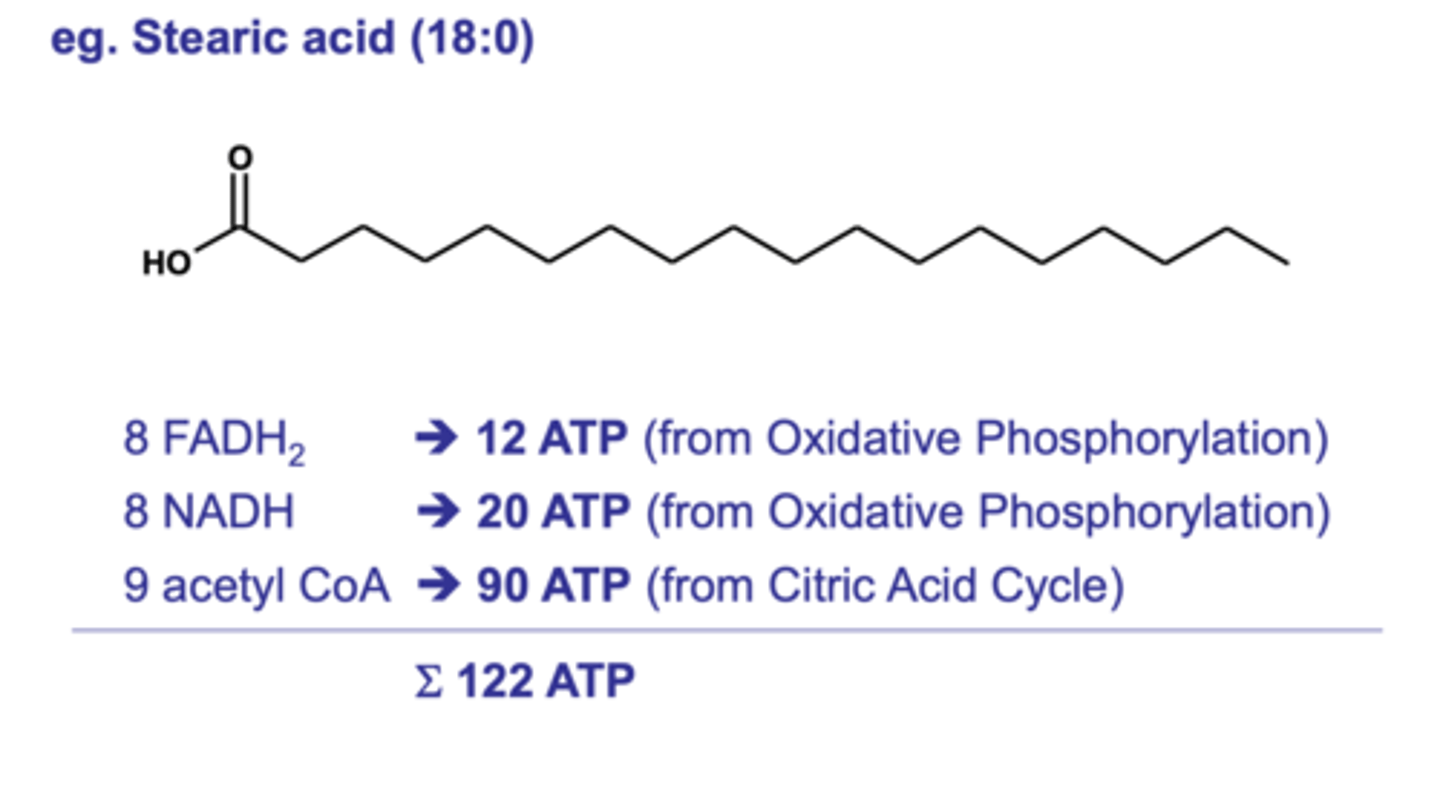
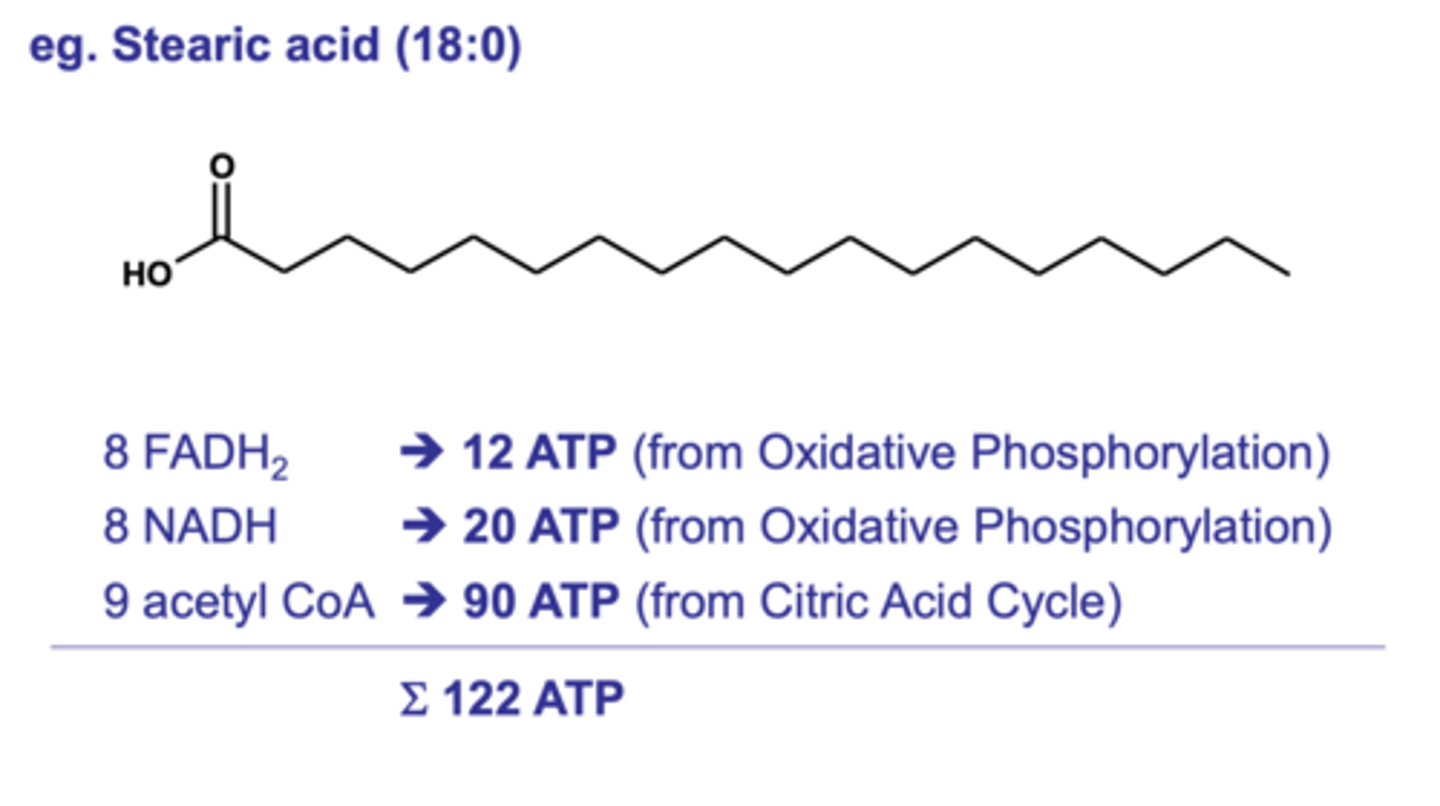
How much ATP is made from a single fatty acid molecule e.g., stearic acid?
122 ATP compared to 30 ATP from a single glucose molecule.
What fuel source (glucose or fatty acids) does skeletal, cardiac, liver and brain tissue?
- Skeletal muscle uses glucose when active and fatty when resting.
- Cardiac muscle uses fatty acid but may use glucose.
- Liver tissue uses fatty acid.
- Brain tissue uses glucose as fatty acids can cross the BBB.
What anabolic reactions is acetyl CoA involved in?
- Lipogenesis.
- Cholesterol biosynthesis.
- Ketogenesis.
What is ketogenesis?
Metabolic pathways where ketone bodies are formed from excess acetyl CoA which cannot be used in the citric acid cycle due to low oxaloacetate levels.
How is cholesterol synthesised from acetyl CoA?
- Two acetyl CoA are combined to form acetoacetyl CoA.
- It is then hydrated to forma precursor for cholesterol.
Describe how ketogenesis takes place.
- Two acetyl CoA molecules are combined to form acetoacetyl CoA.
- It is then hydrated and a single acetyl CoA molecule is removed with produces acetoacetate (ketone).
- Acetoacetate can be altered to form other ketone bodies.
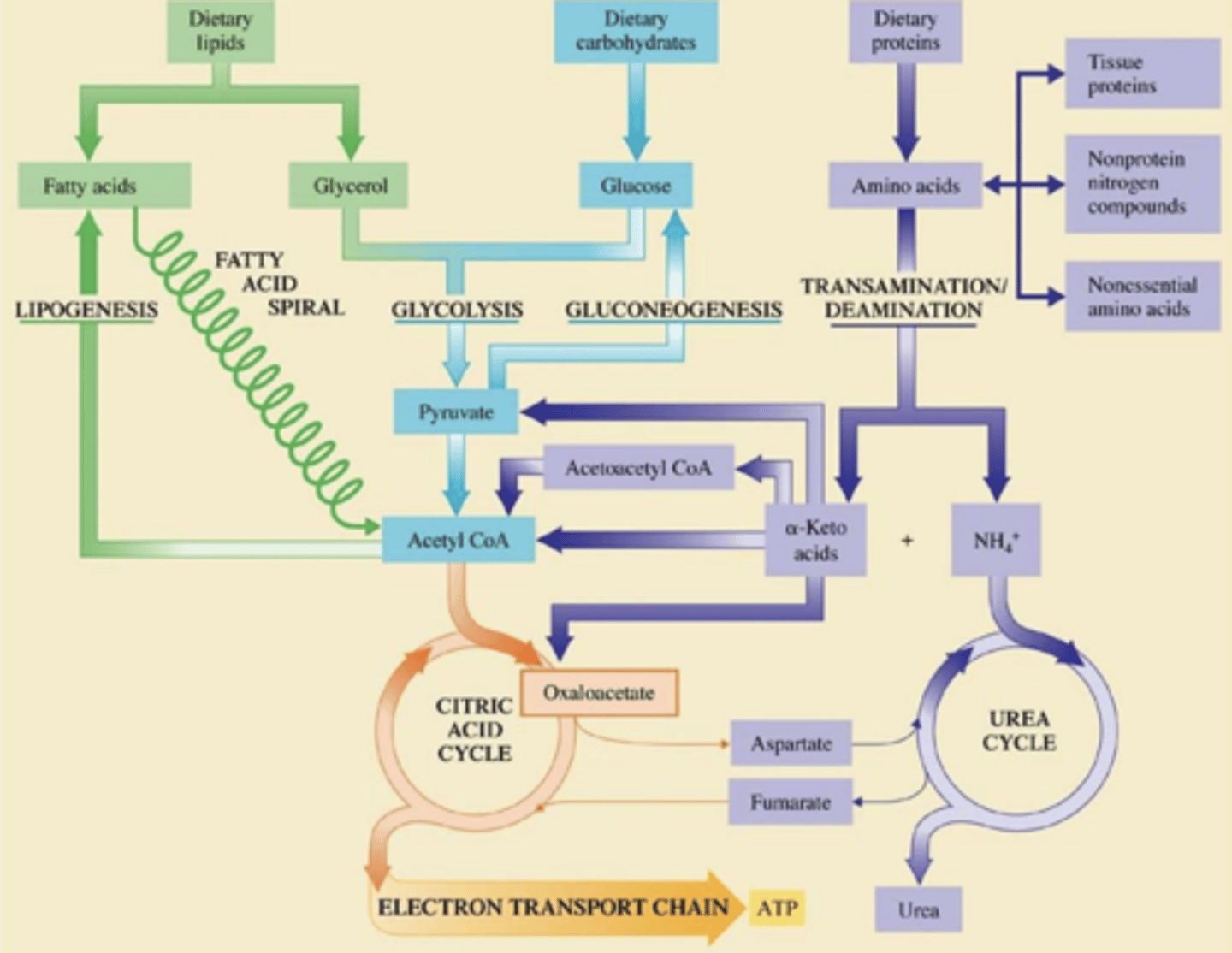
Lipid, Carbohydrate and protein metabolism overview.
What affects does diabetes drugs have?
They cause an increase in secretion of insulin from pancreatic β-cells.
What are the main types of drug that treat diabetes and give an example?
- Sulphonylureas e.g., gliclaszide
- Meglitinides e.g., nateglinide
- GLP-1 agonists e.g., exenatide
- DPP-4 inhibitors e.g., alogliptin
Why are diabetes drugs only effective in type 2 diabetes?
Because they require functioning β-cells which are not present in type 1 diabetes.
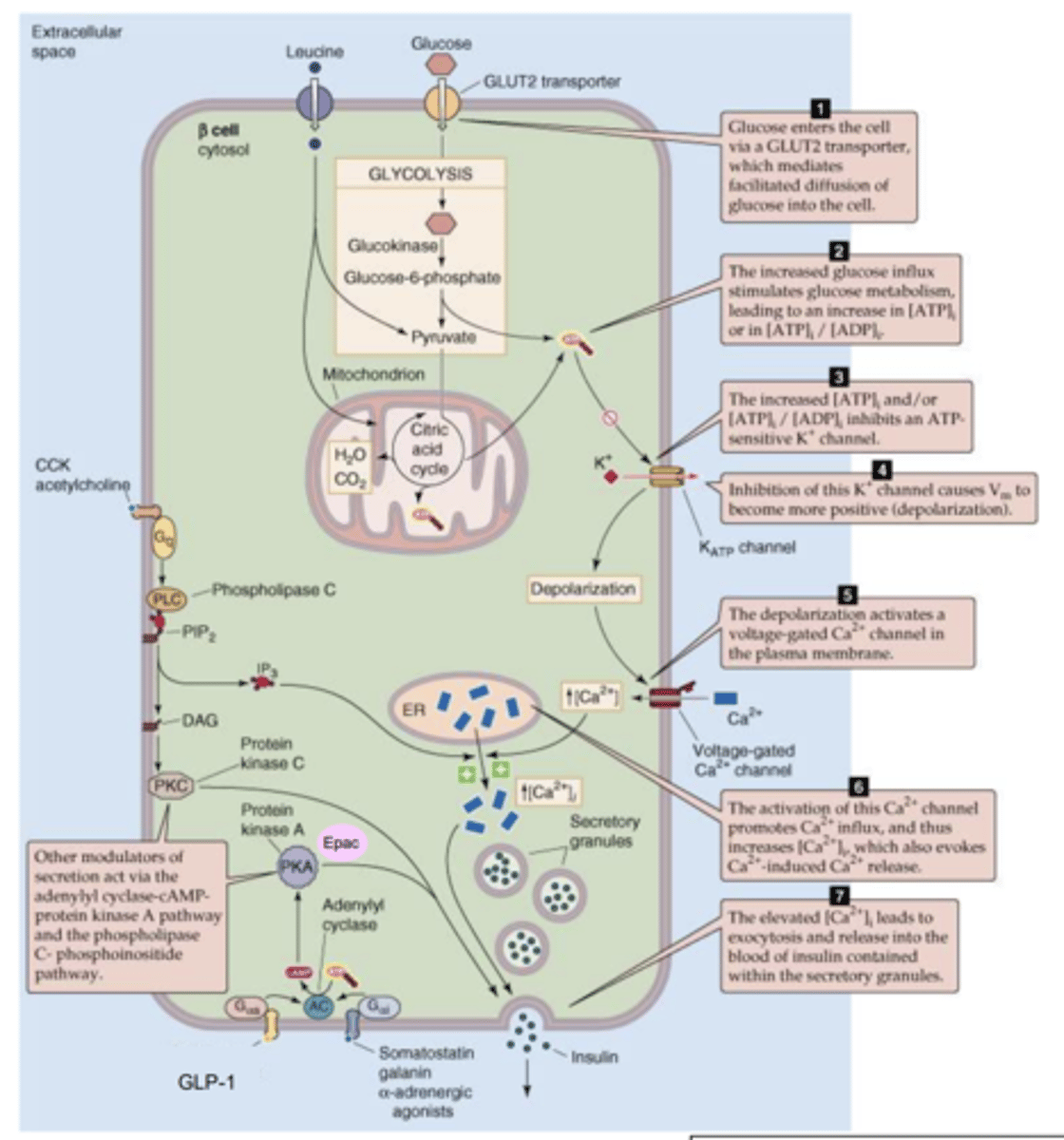
How is insulin secreted from β-cells?
- Aerobic respiration of glucose takes place producing ATP.
- ATP can then close K+ channels by binding to them which prevents them from leaving the cell
- This depolarises the inside of the cell which causes voltage gated Ca2+ channels to open allowing CA2+ into the β-cell.
- Depolarisation also causes release of Ca2+ from ER.
- The increase in Ca2+ in the β-cell causes vesicles containing insulin to be secreted vis exocytosis.
Describe sulphonylureas e.g., gliclazide.
- They block K+ channels which cause β-cells to depolarise and therefore release insulin.
- Can cause hypoglycaemia as insulin secretion is increased even in absence of glucose.
SAME AS MEGLITINIDES8
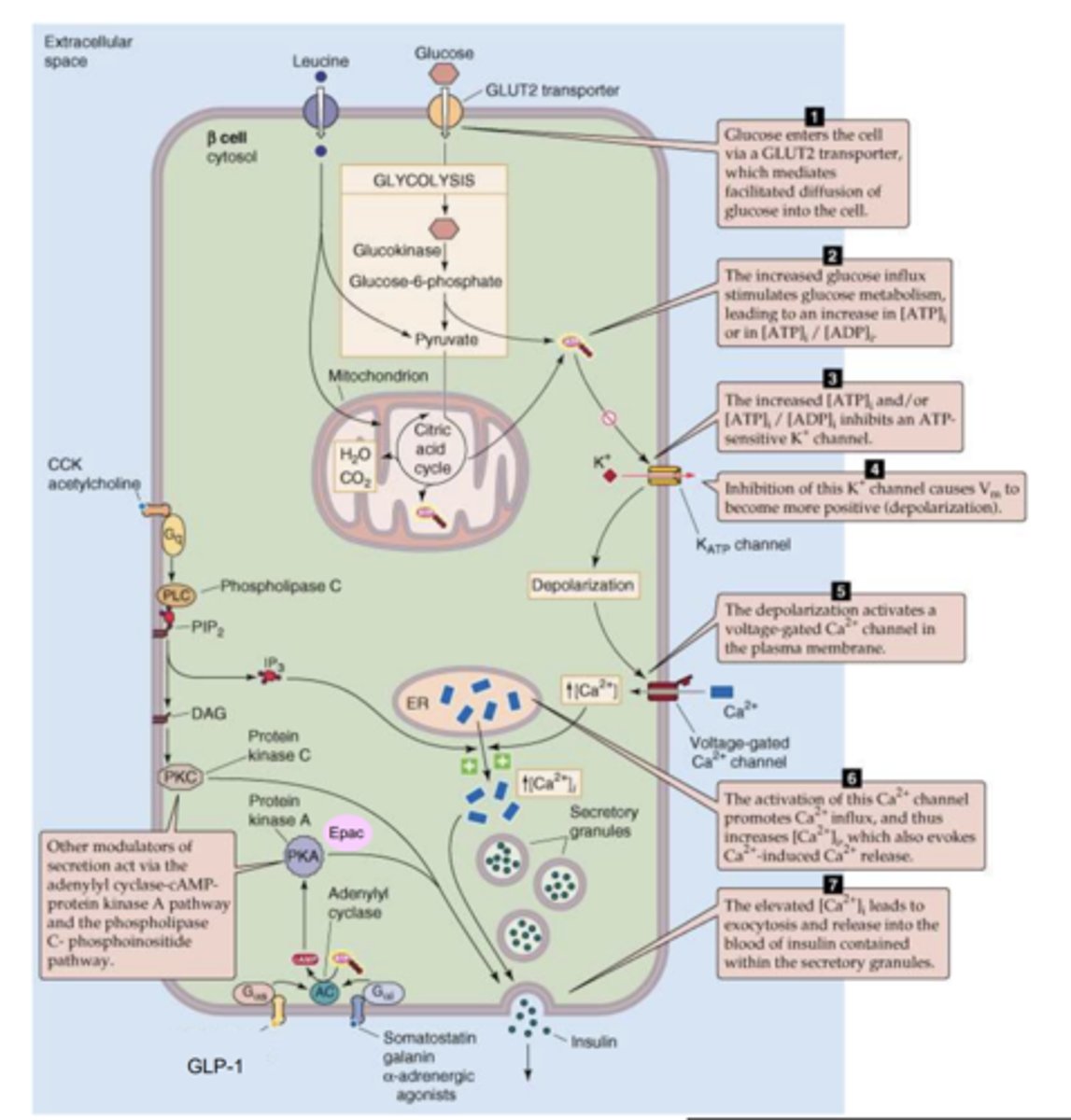
Describe Meglitinides e.g., nateglinide
- They block K+ channels which cause β-cells to depolarise and therefore release insulin.
- Can cause hypoglycaemia as insulin secretion is increased even in absence of glucose.
SAME AS SULPHONYLUREAS
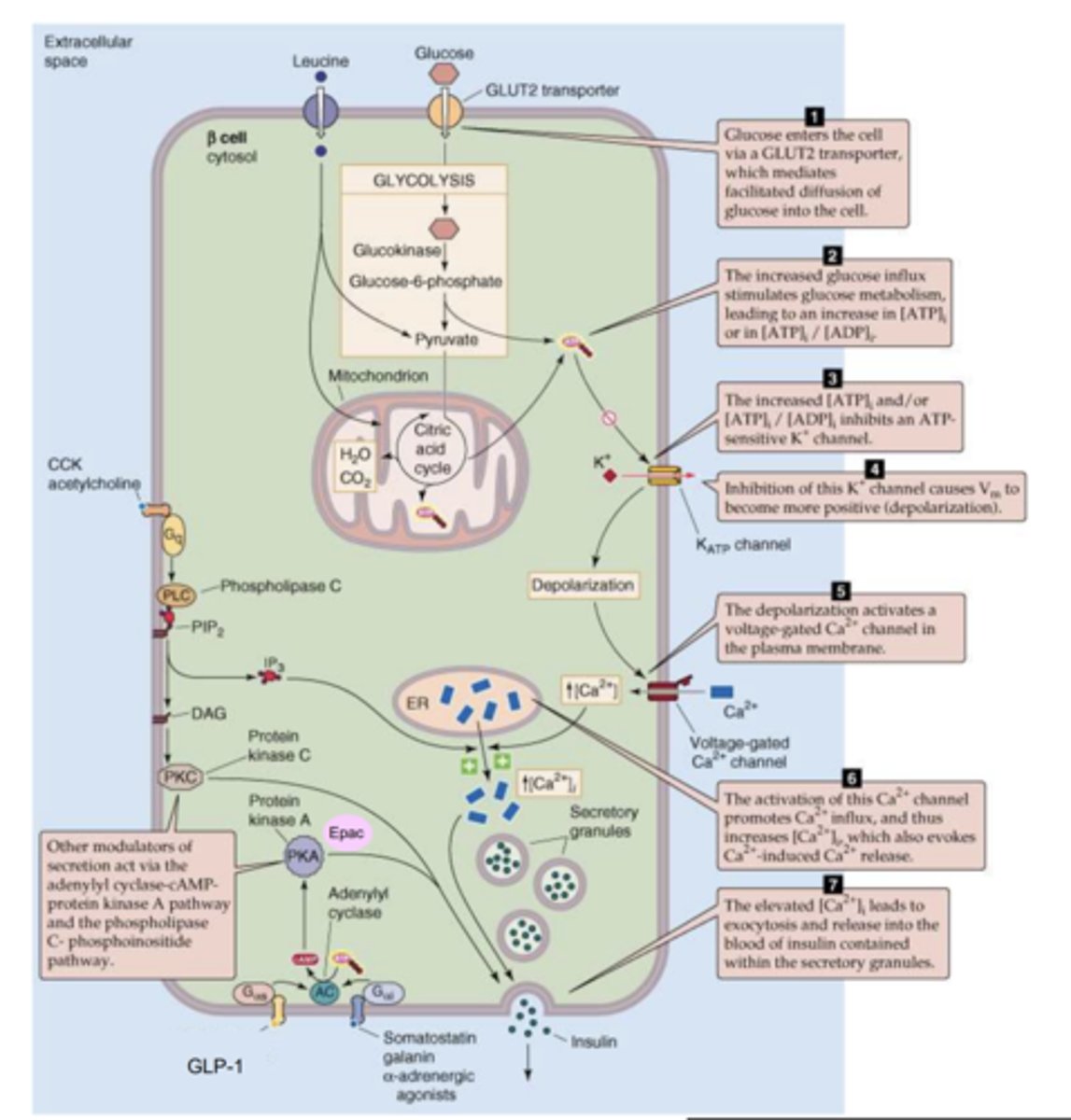
What is GLP-1 and what does it do?
- A peptide hormone which acts on GLP-1 receptors in β-cells.
- It causes an increase in glucose-dependant secretion of insulin.
- Increases the concentration of insulin released per glucose so if 1 molecule of insulin is released from one glucose molecule normally, when GLP-1 is present, 5 insulin molecules will be released from 1 glucose molecule.
Describe the mechancism of action of GLP-1 agonists e.g., exenatide
- The agonist binds to a GPCR on surface of β-cell which activates the Gαs subunit.
This then activates adenlylyl cyclase which produce cAMP from ATP.
- cAMP then activates protein kinase A and Epac which further increase the intracellular Ca2+.
- This causes more insulin vesicles to be secreted.
What other effects do GLP-1 agonists have?
- Decreases gastric emptying (slows down absorption of glucose which can be beneficial for diabetes).
- Decreases glucagon secretion (decreases blood glucose levels).
- Increases satiety (don't feel as hungry).
What is DPP-4 and what does it do?
An enzyme that breaks down GLP-1 and therefore decreases insulin secretion.
Describe how DPP-4 inhibitors (alogliptin) increase insulin secretion.
- They inhibit DPP-4 which prevents GLP-1 from being broken down.
- GLP-1 can then continue to increase glucose-dependent insulin secretion.
- This decreases blood glucose levels.
What does blood glucose monitoring show?
A diabetic patients diabetic control to help us adjust:
- Insulin requirements.
- Avoidance of hypoglycaemia.
- Requirements for exercise.
When do patients monitor their blood glucose?
- Before meals (4-7mmol/l)
- 90 min after meals (5-9mmol/l)
- On waking (4-7mmol/l)
Should record readings in diary.
How often should type 1 diabetics check their blood glucose?
4 times a day typically before meals, at bedtime and when waking.