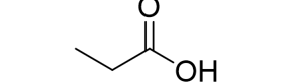
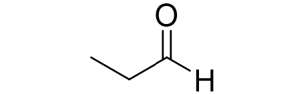
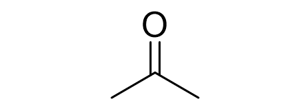
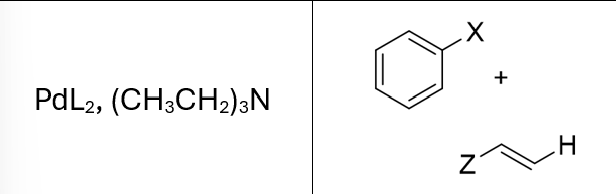
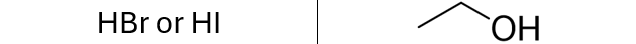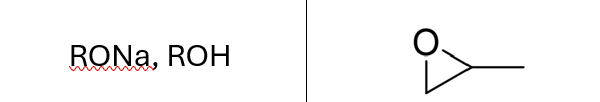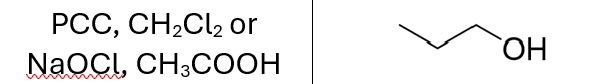Reactions of Alkenes/Alkynes
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
Two-Step Reaction
Markovnikov’s Rule
Carbocation Rearrangements Possible
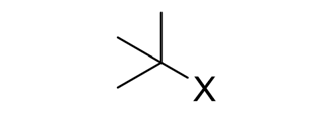
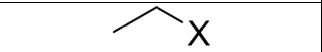
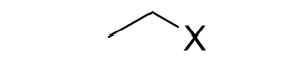
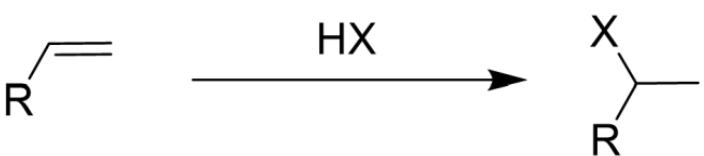
Addition of Hydrogen Halide (HX) to an Alkene
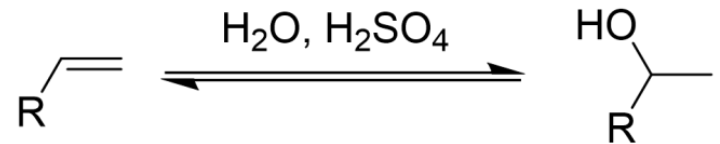
Two-Step Reaction
Acid-Catalyzed
Proton Transfers
Markovnikov’s Rule
Carbocation Rearrangements Possible
Addition of Water (H2O) to an Alkene (Hydration)
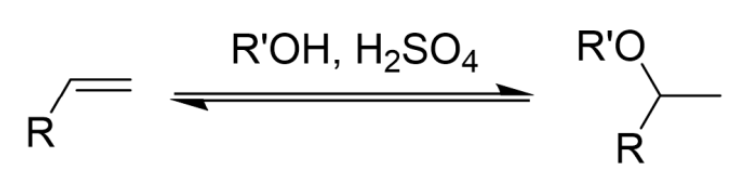
Two-Step Reaction
Acid-Catalyzed
Proton Transfers
Markovnikov’s Rule
Carbocation Rearrangements Possible
Addition of Alcohol (R’OH) to an Alkene
Two-Step Addition
Concerted Addition, Followed by Oxidation
Anti-Markovnikov
Syn Addition
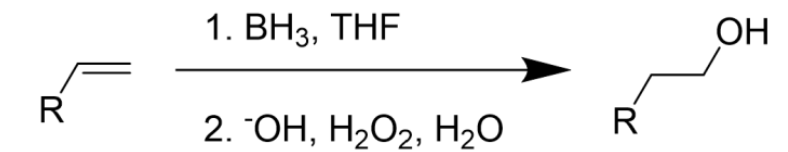
Hydroboration (BH3) of an Alkene
Anti-Markovnikov
Electrophile moves to the more hindered carbon (less substituted)
Anti Addition
Two substituent groups added to opposite faces of the double bond (wedge/dash, dash/wedge)
Syn Addition
Substituent groups added to same faces of double bond (wedge/wedge, dash/dash)
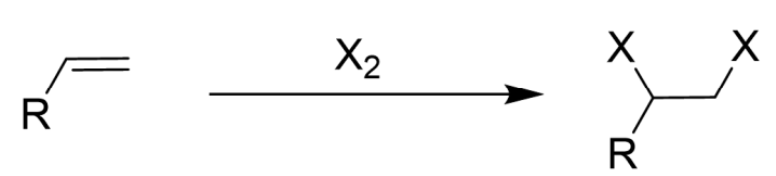
Only Br2/Cl2
Makes Halonium Ion as Intermediate
Anti Addition
Addition of Halogen (X2) to Alkene
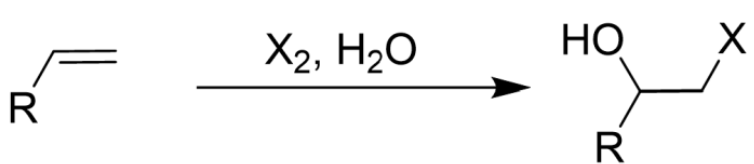
Only Br2/Cl2
Makes Halonium Ion as Intermediate
Anti Addition
Markovnikov’s
OH Group Attack More Substituted Side
Halohydrin (X2, OH) Formation
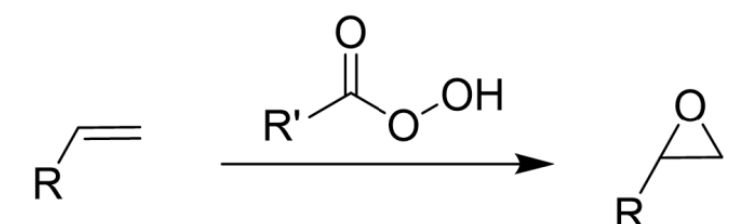
Makes Epoxide
Concerted Mechanism
Syn Addition
Addition of Peroxyacid (mCPBA) to Alkene
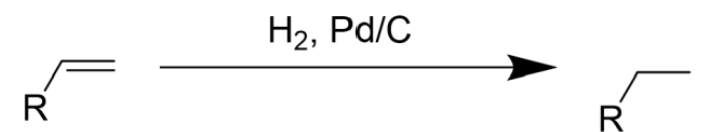
Pd/C Catalyst
Syn Addition
Reduction Reaction
Hydrogenation (H2) of Alkene
Ozonolysis (O3) of Alkene
Double Bond “Cleaved”
Can make Aldehydes and Ketones
Each Side of Cleaved Double Bond has a new double bonded oxygen
Regioselective
Two constitutional isomers could be made, one is made preferentially
Stereoselective
Favors some stereoisomers from all possible stereoisomer products
Stereospecific
Produces Only One Stereoisomer
Racemic
Makes equal amount of both enantiomers
Vinylic Carbocation
Carbocation formed on alkenes after the reaction of an alkyne; More unstable than monosubstituted
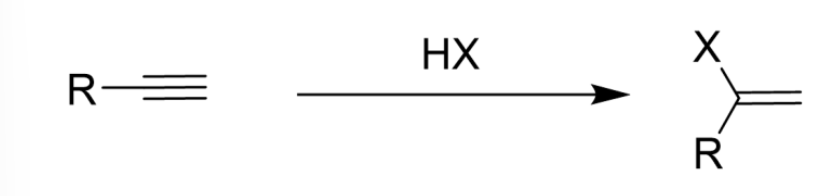
Markovnikov’s Rule
1 Equivalent makes an alkene, excess makes a dihalide
Addition of Hydrogen Halide (HX) to Alkyne
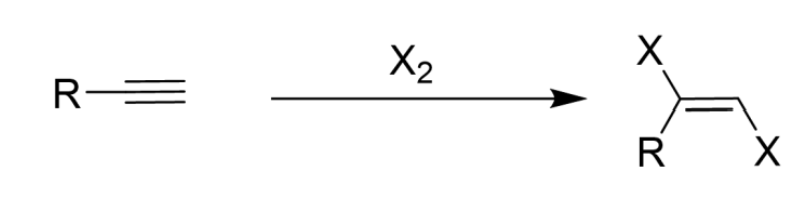
Only Cl2/Br2
Anti Addition
1 equivalent makes a trans alkene, excess makes a tetrahalide
Addition of Halogen (X2) to Alkyne
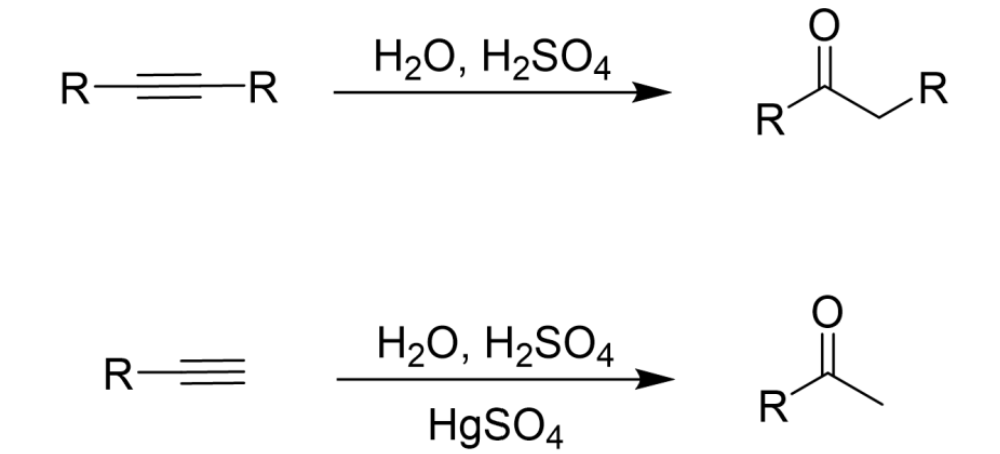
Only one unit of H2O added across triple bond
HgSO4 necessary for terminal alkenes
Tautomerizes to make a ketone
Addition of Water to Alkyne
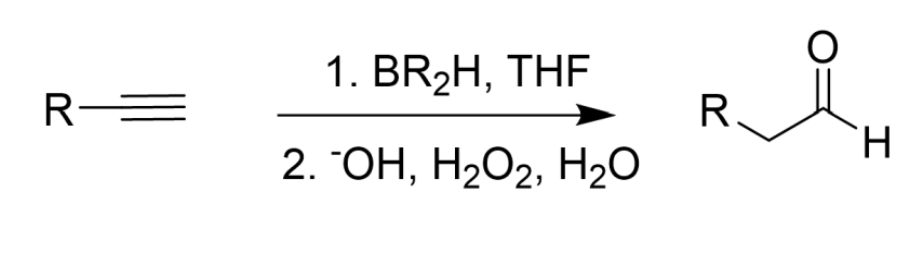
Only 2 Borons
Similar to Alkene Hydroboration (Less Substituted)
Tautomerizes to make an aldehyde
Hydroboration (BR2H/9-BBN, THF) of Alkyne
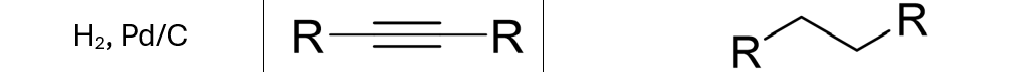
Both pi bonds removed (forms alkane
Hydrogenation (H2) of Alkyne

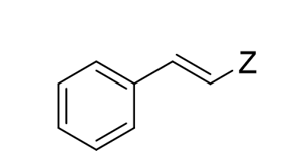
“Cis” hydrogenation (Z)
Only one pi bond removed
Lindlar’s Catalyst of Alkyne

“Trans” hydrogenation
Only one pi bond removed
Adding Na/Li + NH3 to Alkyne