BS1050 Block 6 - Regenerative Medicine and Gene Therapy
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
How does the UK Medical Research Council define regenerative medicine?
An interdisciplinary field that seeks to develop science and tools to repair or replace damaged or diseased human cells or tissues to restore normal function.
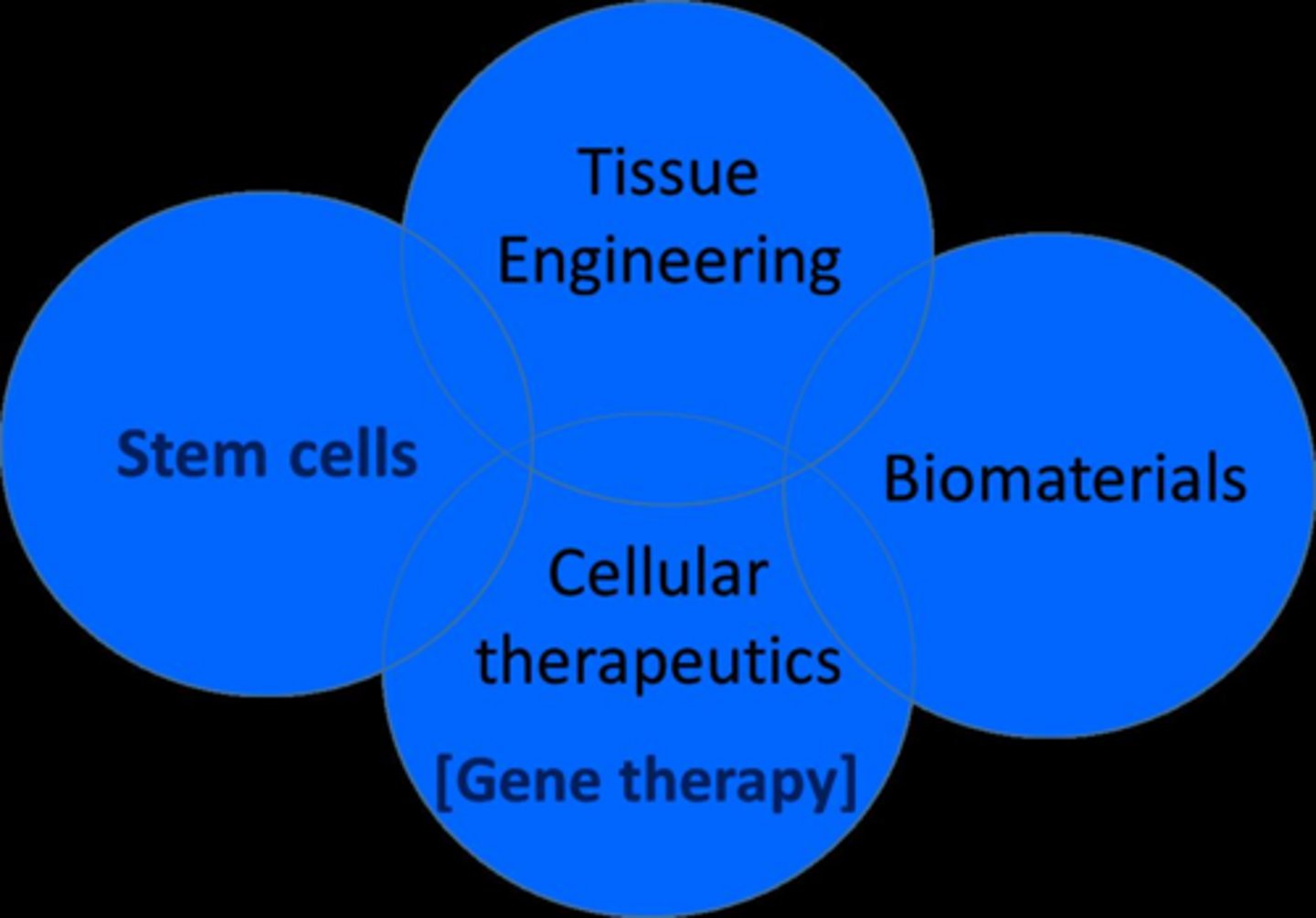
4 main areas of regenrative medicine
tissue engineering,bimaterials,stem cells, gene therapy
What are the two fundamental abilities of stem cells?
Self-renewal and differentiation
divide and the progeny of cells once they divide and produce progeny that differentiates in specifalised cell types= healthy organims s
How does stem cell self-renewal differ from cancer cell proliferation?
Unlike cancer cells, stem cells can produce progeny that differentiate into specialized cell types to contribute to a healthy organism
cancer cells divide continuously and dont differentiate .
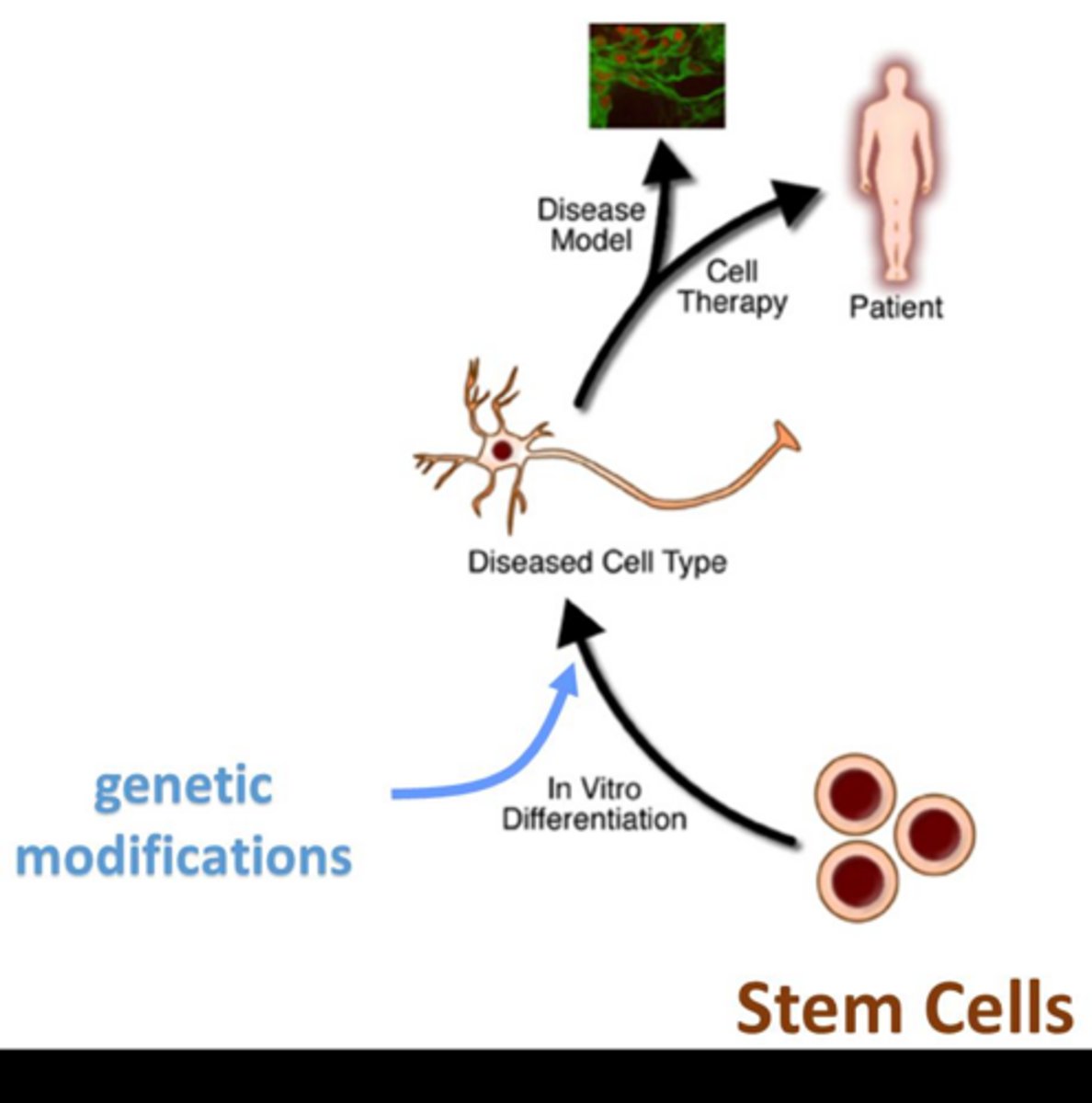
What are the two primary methods for manipulating the stem cell genome in vitro?
Inserting exogenous DNA to create transgenic cells or editing the endogenous DNA.
potency
potential of becoming undifferentiated cells

Define totipotent stem cells and provide an example.
Cells capable of forming all cell types; examples include the zygote and blastomeres up to the morula- (blackberry a ball) stage.
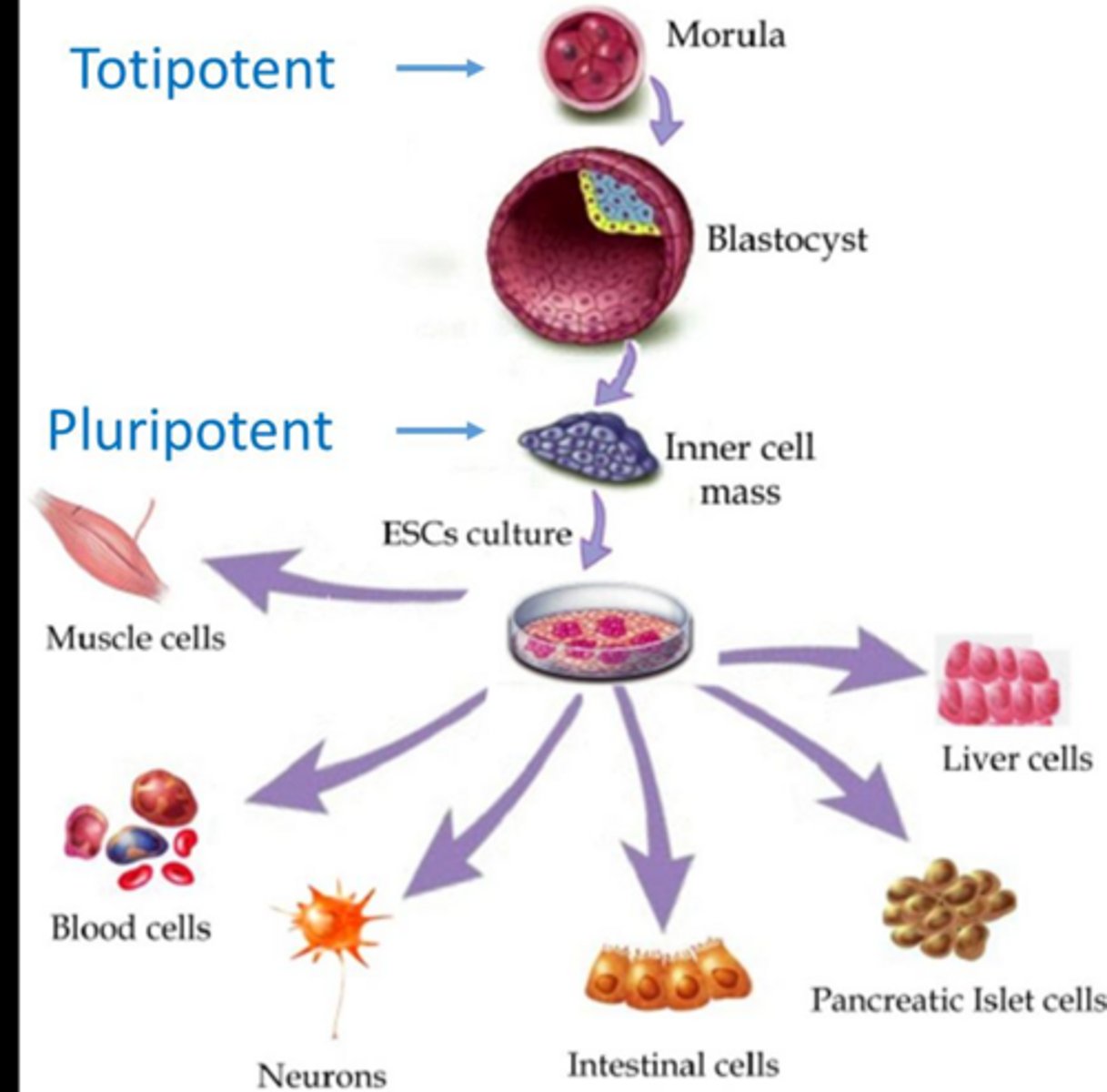
totipotent and pluripotent difference
these cells can be grown and make embryonic stem cells
gastrulation = cells are in ball and scarify in 3 sheet, layers, gut and muscle/heart,can make muscles neurone etc
why they are pluripotent THEY CANNOT DO PLACENTA
What is multipotent?
diff type of cells within one type e.g within mesoderm produce bones,muscles but not neurons
What is oligopotent?
They can produce different types of cells of one defined tissue- less potent
unipotent?
cells of skin -only produce cells part of skin
Which types of stem cells are classified as pluripotent?
Embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs).
multipotent and oligopotent - HSCs
mulitpotent produce both- lymphoid eg t cells and myeloid lineage produce evrrything else blood cells,macrophages
oligopoent example
neuron- self renewing, neural stem cell
neurogenesis - important for our mood, depression so a cure is driving these cells to proliferate
What is the potency level of mesenchymal stem cells (MSCs)?
Multipotent.
Where are epidermal stem cells located and what is their function?
They are found in the basal layer of the epidermis and are responsible for the everyday regeneration of the different layers of the epidermis.
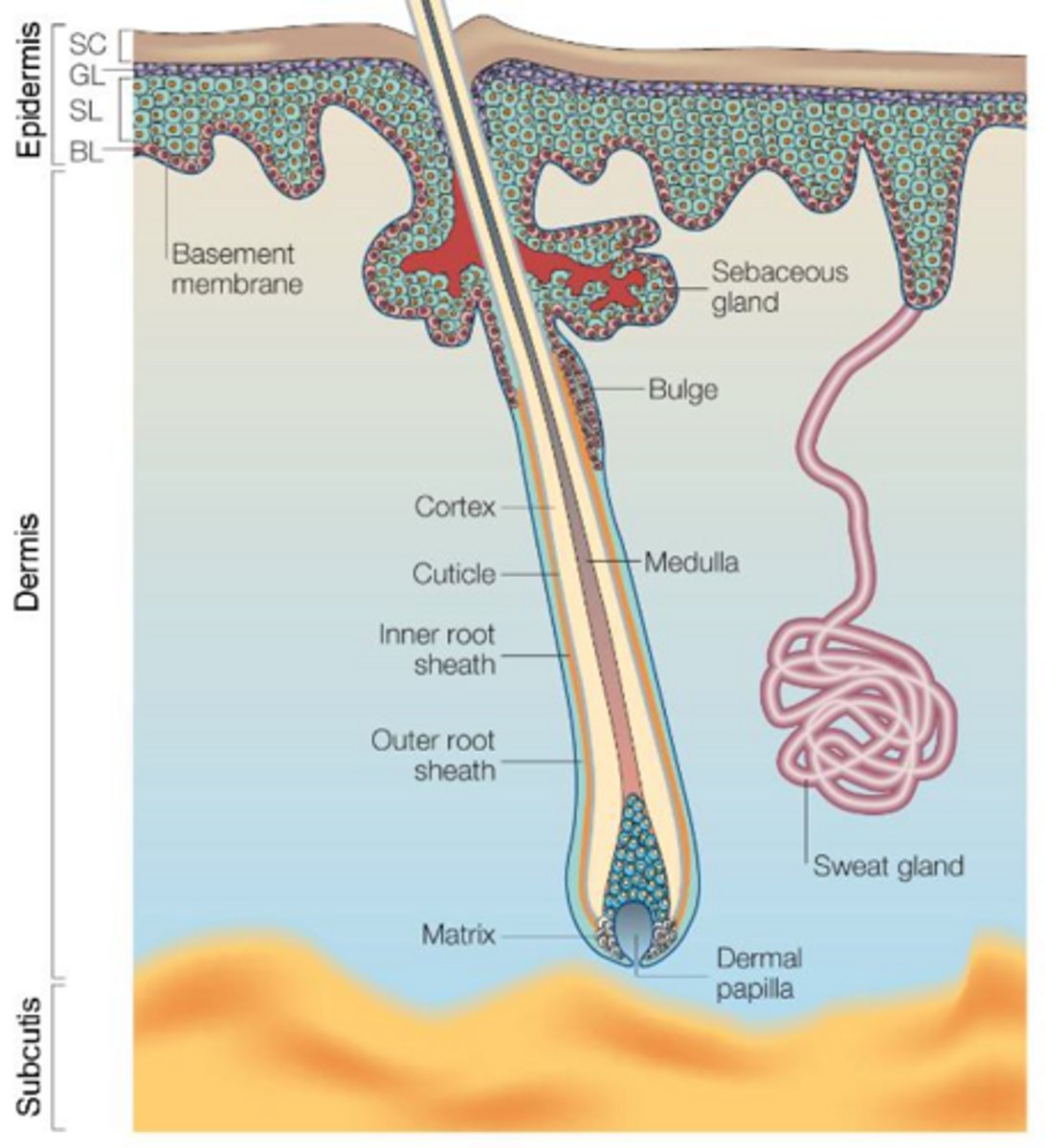
What is the function of hair follicle stem cells?
They ensure constant renewal of hair follicles and can regenerate the epidermis and sebaceous glands if those tissues are damaged.
What is the role of melanocyte stem cells?
They are responsible for the regeneration of melanocytes, which produce melanin for skin and hair pigmentation.
Why are adult stem cells or somantic generally considered inconvenient for therapy?
They are typically scarce, difficult to isolate, and often have limited growth potential in vitro apart from HSCs that already used
Why are pluripotent stem cells (like ESCs) preferred for therapeutic research?
They grow indefinitely in vitro and can be differentiated into any cell type by adding specific signaling molecules.
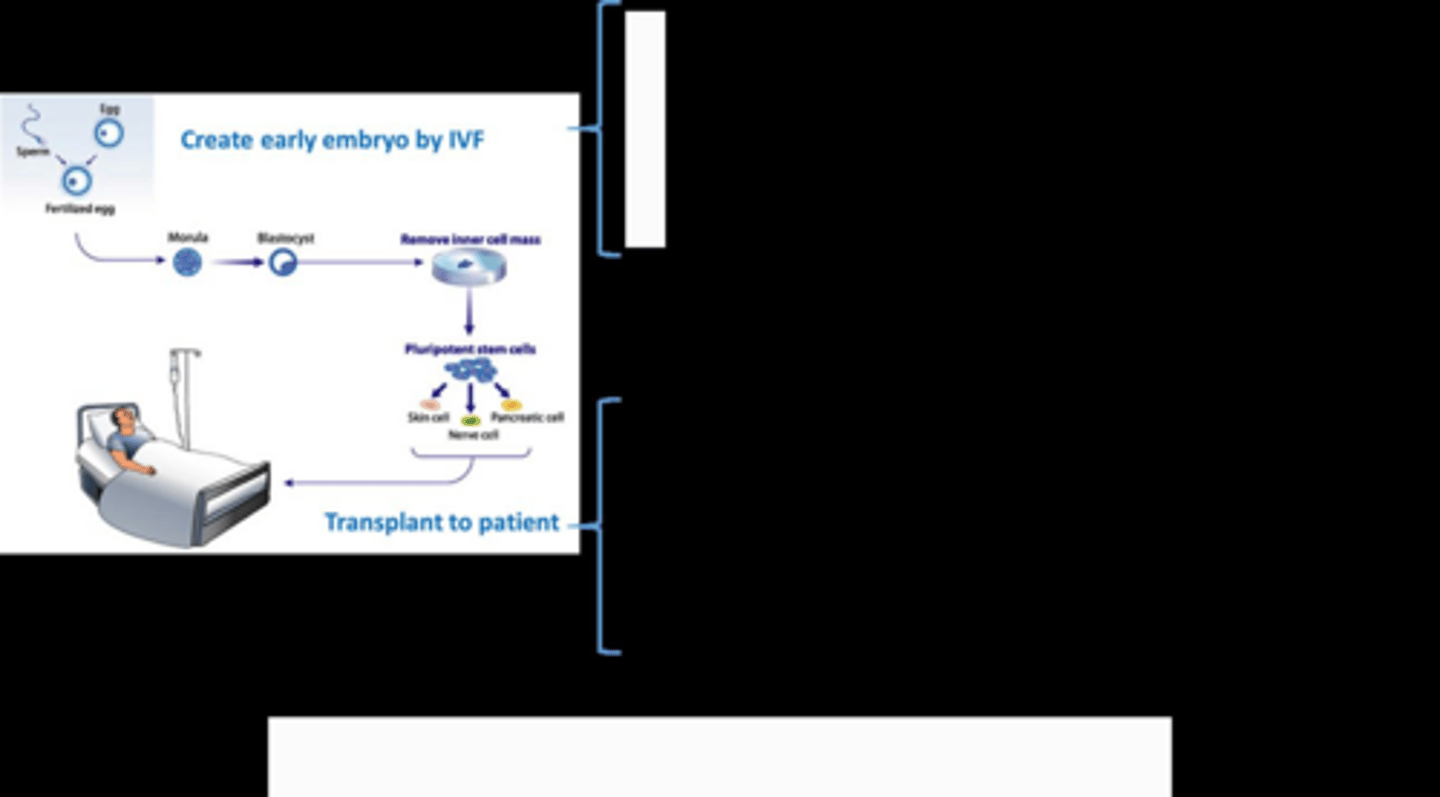
problem for in ECs
egg wont be identical so chance for rejction
not pure- keep growing like cancer so can be dangerous
What are the two main objectives of stem cell transplantation?
To physically replace unhealthy/degenerated cells or to provide nutritional/signaling support to endogenous cells to delay degeneration.
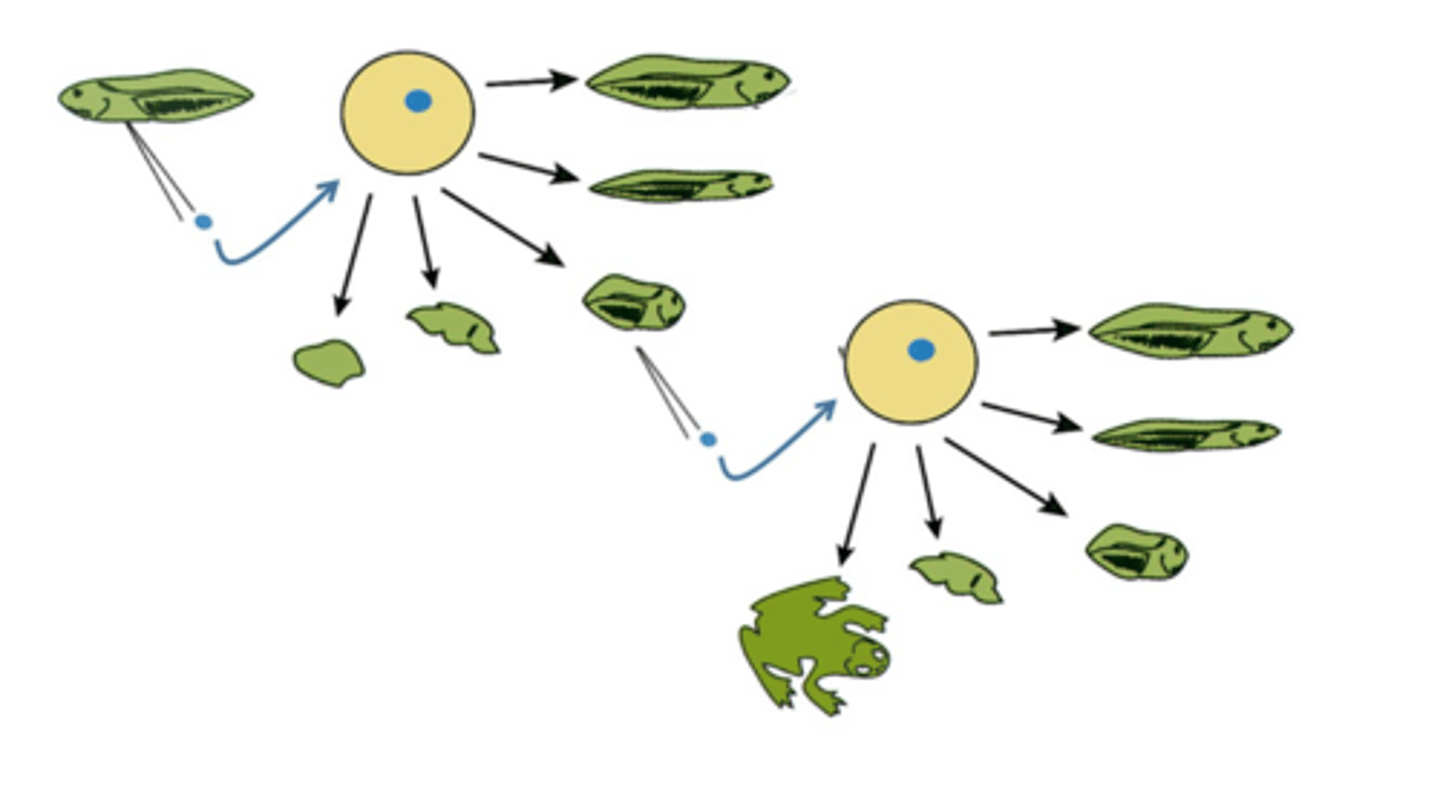
What is somatic cell nuclear transfer (SCNT)?
A technique where the nucleus of a somatic cell is transplanted into an egg cell to create a cloned embryo.
somatic cell nuclear transfer
egg from donor remove nucleus and put another nucelus from paitient and then differentiate e.g in dollynsheep
An embryo capable of generating pluripotent stem cells.
how does SCNT program cells
The egg cytoplasm resets the nucleus.
What is the difference between iPSC and SCNT?
iPSC uses transcription factors; SCNT uses an egg.
What was the significance of the 2012 Nobel Prize awarded to Gurdon and Yamanaka?
It recognised the discovery that mature cells- zygote can be reprogrammed to become pluripotent
Some proteins can open different areas that are compact (heterochromatin) to make it differentiate and found OCT4,KLF4,SOX2,MYC are able to differentiate cells
What are the two main roles of stem cells in therapy?
Replace damaged cells and provide signalling support or nutritional
Why is the eye considered an ideal target for stem cell clinical trials?
It is an immune-privileged environment- antbodies find it hard to penetrate so tissue rejection is less, easily accessible environment that allows for non-invasive monitoring and requires only small numbers of cells
if something goes wrong wont kill paitient
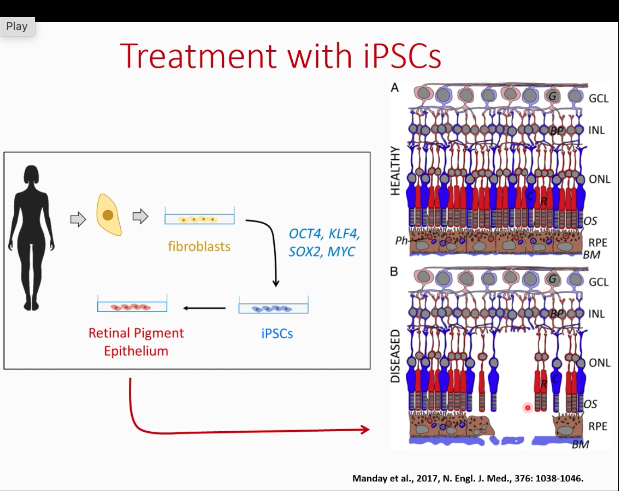
What is the primary cause of blindness in age-related macular degeneration? iPSCs
The loss of the retinal pigment epithelium causes loss of photoreceptors that depend on it
neural circuits are generally intact - fibroblasts induce them with factors and make them inducable using iPSCs and growth factors and put it back in retina
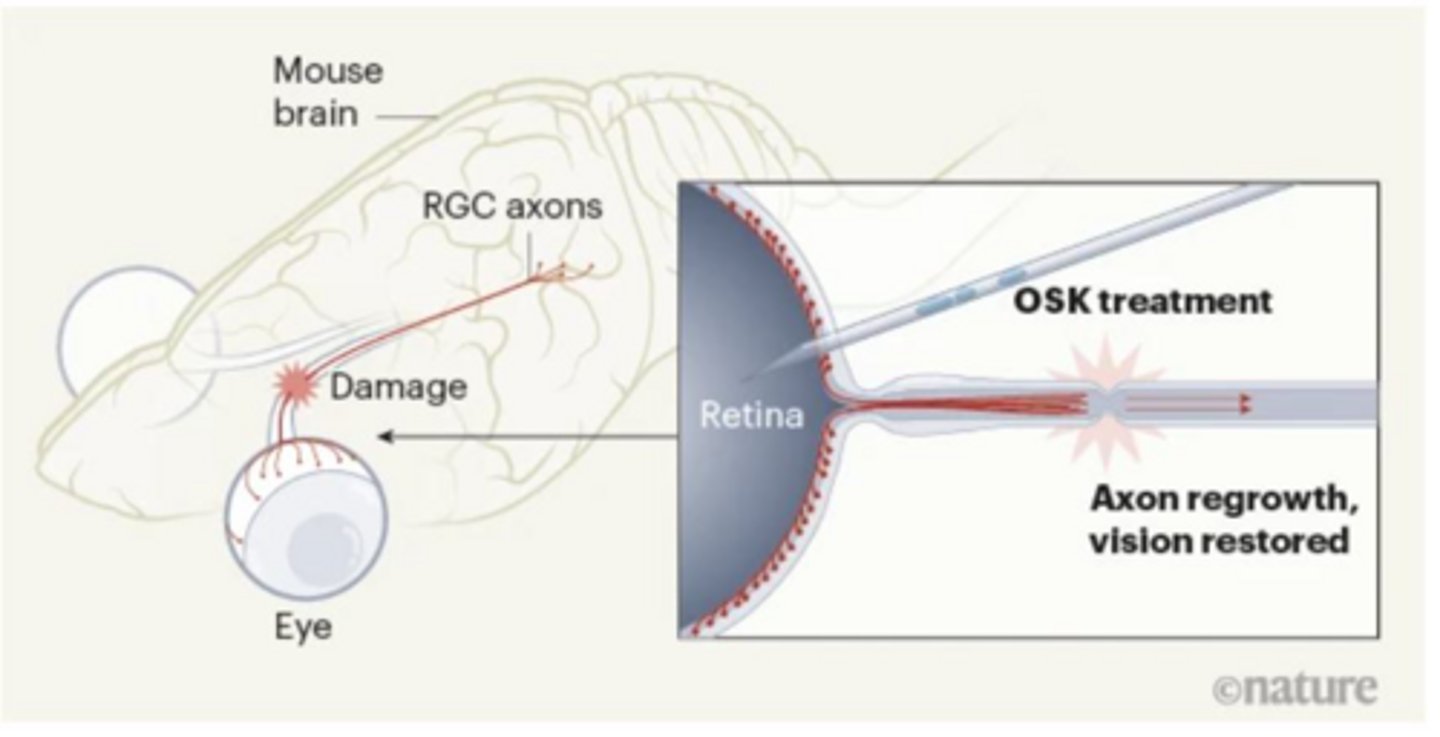
What does 'partial reprogramming' aim to achieve in research?
It aims to reset aging signatures in cells rather than changing their cellular identity.
What is the 'OSK' cocktail mentioned in recent regenerative research?
A transcription-factor cocktail used to restore damaged cells to a youthful state, promoting axon regeneration.
What is the difference between allogeneic and autologous stem cell therapy?
Allogeneic therapy uses cells from a donor, while autologous therapy uses the patient's own cells.
What is the potency of neural stem cells (NSCs)?
Oligopotent.
What is the potency of hematopoietic stem cells (HSCs)?
Multipotent.
What is the potency of unipotent stem cells?
They are restricted to producing only one cell type, such as specific skin or muscle stem cells.
What is the main advantage of using stem cells as delivery vehicles?
They can be used to deliver therapeutic agents like genes and cytokines directly to target tissues.
What is the primary definition of gene therapy?
An experimental treatment that uses gene transfer of genetic material into a patient's cells to cure a disease by modifying the genetic information responsible for the condition.
What are the three main types of gene therapy interventions?
Replacement (adding a functional gene), silencing (blocking a dysfunctional gain-of-function allele), and editing (correcting faulty alleles or related loci).- in that gene or a related gene to correct
What is the difference between in vivo and ex vivo gene therapy?
In vivo involves modifying patient cells directly within the body, while ex vivo involves modifying autologous cells outside the body making it pluripotent and before reintroducing them to the patient.
What are the two main categories of nucleic acid delivery vectors?
Viral vectors and non-viral vectors.
What are the primary advantages of using viral vectors in gene therapy?
High payload capacity- lots of nucleic acid inside , specific cell tropism eg treating liver has portein on capside which binds to receptors in liver etc , high transduction efficiency, and high expression levels.
What are the potential risks associated with viral vectors?
Genotoxicity, cytotoxicity, and unwanted immune responses.
What is a key characteristic of Adeno-associated virus (AAV) vectors? more commonly used RNA virus
They typically persist as episomes without integrating into the host genome and are commonly used for in vivo gene delivery.
injected dna stays in episome and doesnt divide but expresses sp dna will be transcribed and make protein but you need be injected again
Which cell types are commonly targeted by AAV vectors?
Hepatocytes, neurons, muscle cells, and retinal cells.
What is a defining feature of Lentiviral vectors?
They integrate into the host genome, allowing for permanent gene transfer to dividing cells for long term expression
ntegrate in genome- whatever you want to express it will be, insertion is random if by chance break endogenous gene = bad and local gene = canceri
example of lentivirus
HIV, cold sores
Why are blood cells considered appealing targets for gene therapy?
They are easily accessible for collection and manipulation.
How does gene therapy for Haemophilia A function? x linked recessive
An AAV vector delivers a cDNA expression cassette for Factor VIII into hepatocytes, where it persists as an episome to provide continuous clotting factor release.
nucleic acid deliery molecules
dna or rna have a negative charge bc of phosphate so if we want to insert it into the cell it’s surrounded by lipid membrane that’s not too neg
What are Lipid Nanoparticles (LNPs) used for in the context of gene therapy?
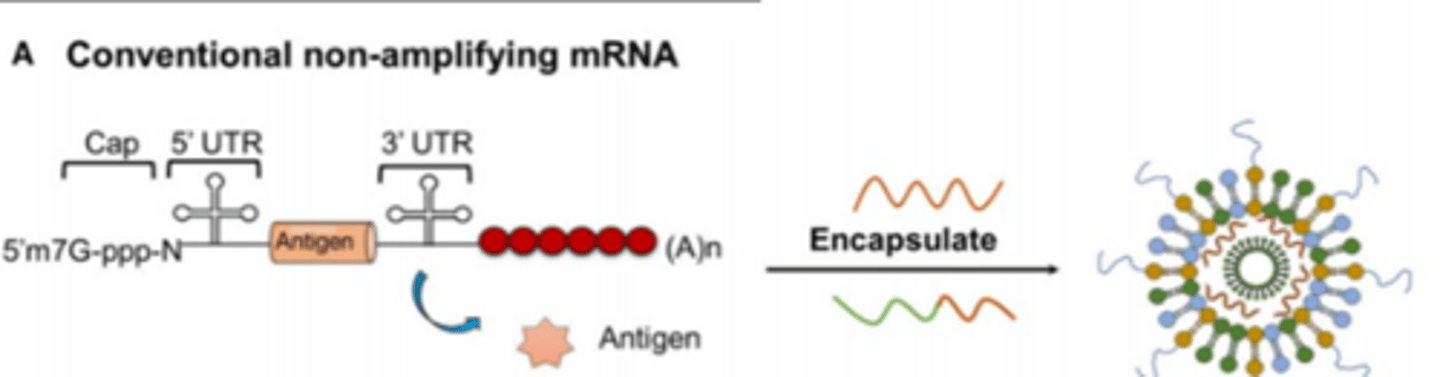
They act as non-viral delivery molecules for nucleic acids, such as in mRNA vaccines.
What is the fundamental mechanism of RNA interference (RNAi)?
It is a regulatory mechanism used to modify gene expression by silencing specific genes.
What is an example of a non-viral system?
Lipid nanoparticles.
LNPs
lipid simialr to membrane hydrophobic and hydrophilic part keeping messenger RNA inside and cholesterol - making membrane less fluid (age enzyme makes too much and it sticks to arteries - why cholesterol is bad)
PEG- polymer which is hydropholic which coats in water so prtoeinc annot stick by electrostatic interaction
What is the function of the CRISPR/Cas9 system in genome editing?
It uses a synthetic guide RNA to direct the Cas9 enzyme to a specific DNA sequence, allowing for precise genome editing.
mRNA vaccines
encapsualted and injected into us
What is RNA interference (RNAi)?
Gene silencing by degrading mRNA. esp in retroviruses they way they integrate make double stranded rna which is used to make dna
dicer- dsRNA recognises it and cuts it
RISC ecapsualte fragments and release some
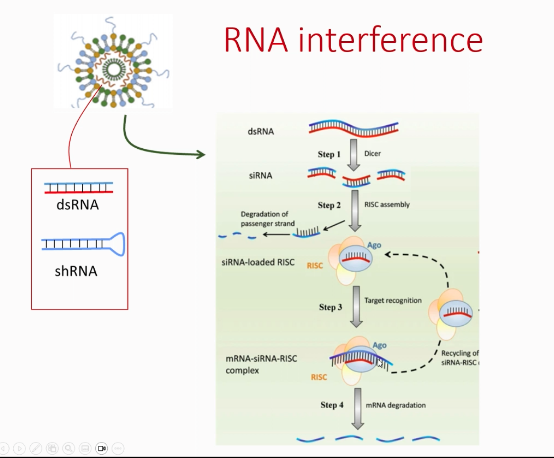
What happens to double-stranded RNA?
Cut into siRNA by Dicer.
What is the role of RISC?
Uses siRNA to bind and degrade mRNA.
What is the result of RNAi?
no protein is produced.
At what level does RNAi act?
Post-transcriptional.
What is CRISPR-Cas9?
A gene editing tool that cuts DNA at specific sequences.
What determines the target site in CRISPR?
Guide RNA (gRNA).
What enzyme cuts DNA in CRISPR?
Cas9.
What are the two repair mechanisms in CRISPR?
NHEJ and HDR.
What does NHEJ cause?
genetic alterations, chromosomal translocations, and potential genomic instability
What does HDR allow?
Precise DNA repair.
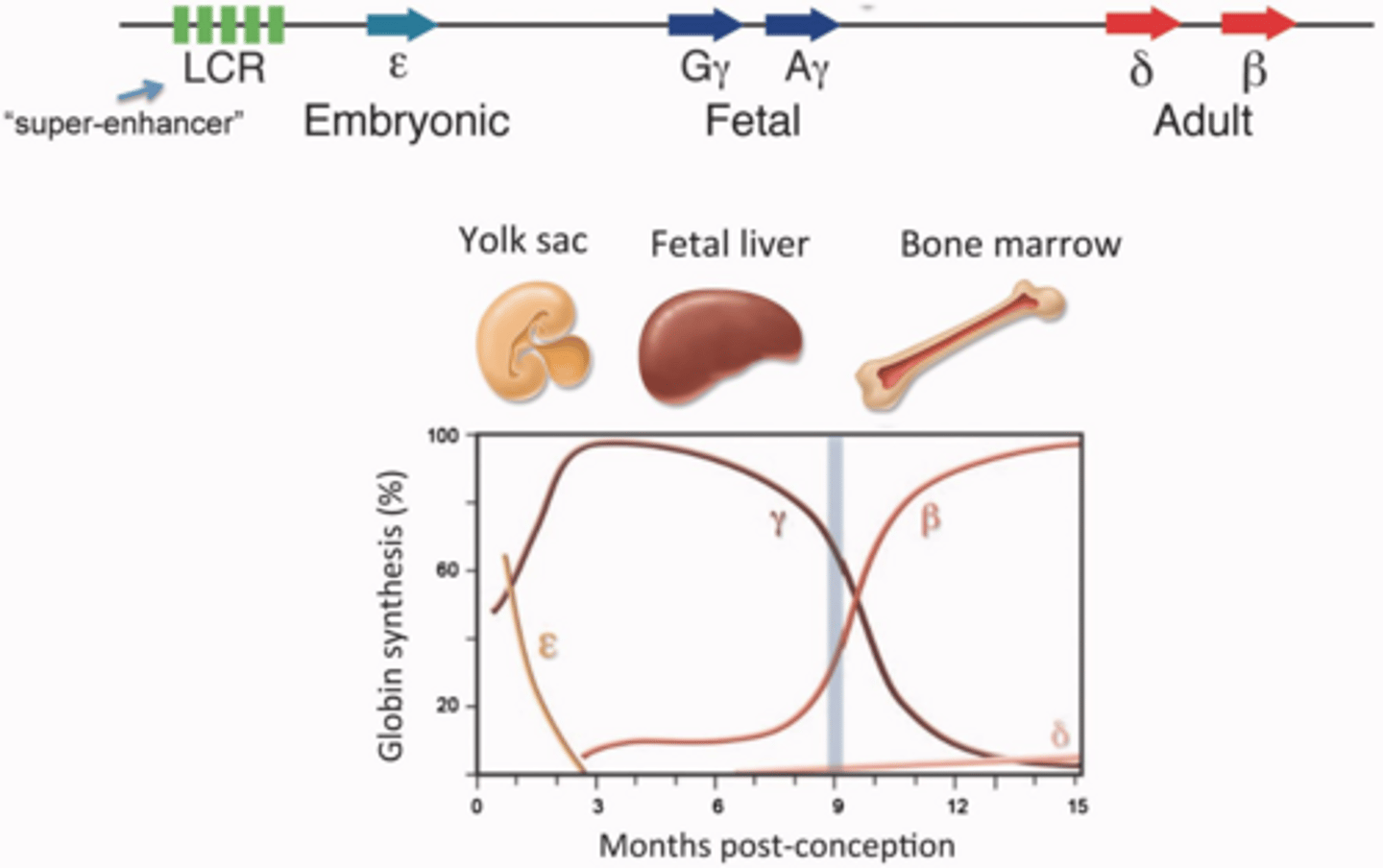
Why is gene therapy for beta-thalassemia considered challenging?
It requires achieving high yet balanced levels of beta-globin production within specific cell lineages.
What is the primary goal of targeting BCLA11 in gene therapy?
It is a target for modulating hemoglobin expression, particularly relevant in treating hemoglobinopathies like sickle cell anaemia and beta-thalassemia.
What does 'transduction efficiency' refer to in viral vector delivery?
The ability of a viral vector to successfully enter a target cell and deliver its genetic payload.
What is the significance of episomal persistence in AAV vectors?
It allows the therapeutic gene to be expressed without the risk of insertional mutagenesis associated with genome integration.
What is the role of autologous cells in ex vivo gene therapy?
They are the patient's own cells (stem or differentiated) that are removed, genetically modified, and then returned to the patient to avoid immune rejection.
What is the primary difference between silencing and editing in gene therapy?
Silencing blocks the expression of a harmful gene product, whereas editing physically alters the DNA sequence to correct the mutation.
haemoglobin expression
alpha and beta must balanced
expression of each chain are controlled
on diff chromosomes beta 11 alpha 16
type we use go through changes- decide what it’s expressed
adult- 2 alpha and2 beta