3.3.11.3 nucleophilic properties
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
what can amines act as?
as nucleophiles
name the mechanism haloalkanes undergo to form amines
nucleophilic substitution
what is the condition to form amines with haloalkanes?
excess ammonia
outline the products that can be formed from a reaction between ammonia + amines with haloalkanes
this reaction can form primary, secondary, tertiary amines and quaternary ammonium salts
eg for the reaction between bromoethane + excess ammonia, give the symbol equation
CH₃CH₂Br + 2NH₃ → CH₃CH₂NH₂ + NH₄Br (quaternary ammonium salt)
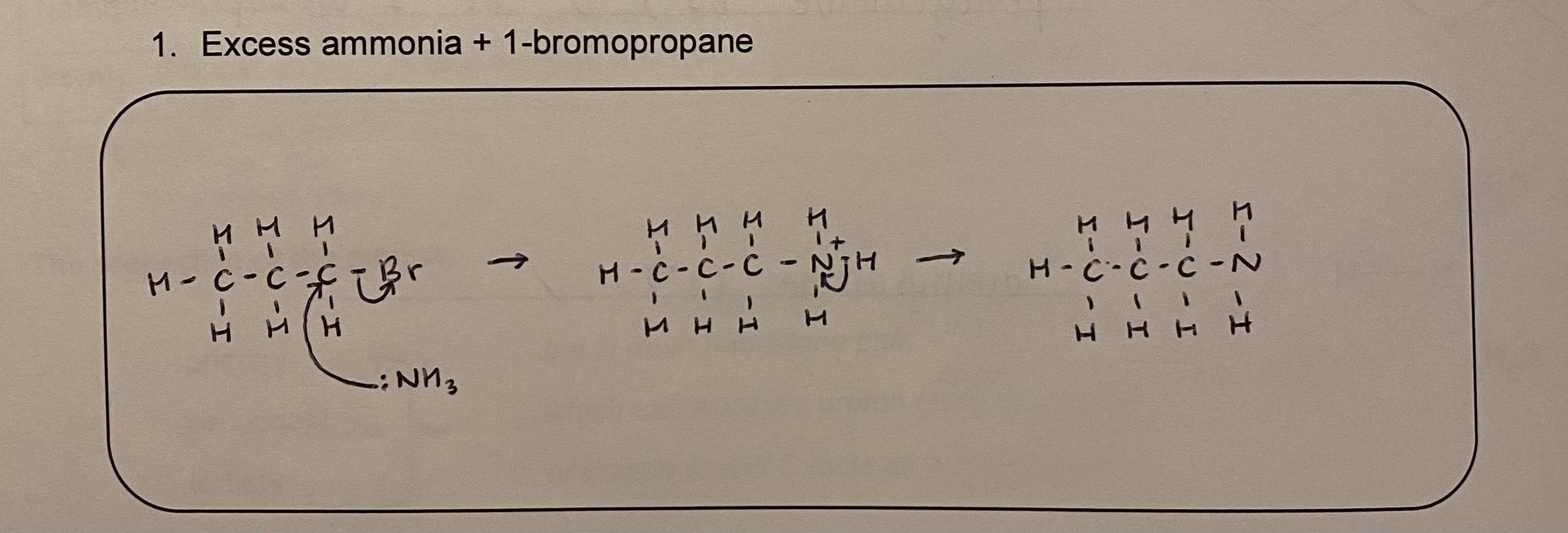
draw the mechanism for eg excess ammonia and 1-bromopropane
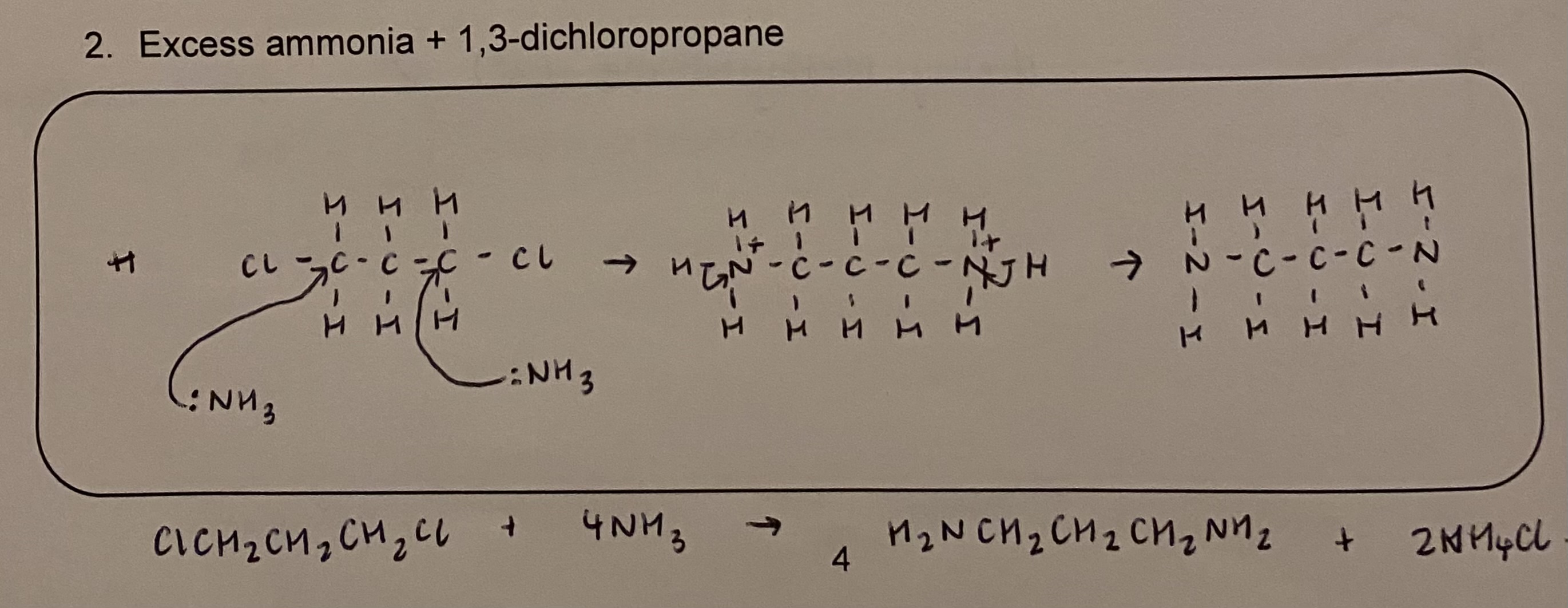
draw the mechanism + give the symbol equation for eg excess ammonia and 1,3 - dichloroopropane
outline the disadvantage of nucleophilic substitution of haloalkanes as a method of preparing amines
a mixture of amine products is formed due to the product also acting as a nucleophile
therefore a LOW YIELD of primary amine is produced due to further substitution
other products include secondary, tertiary amines + quaternary ammonium salts
how can you minimise these further substitutions?
by using a large excess of ammonia as this would make ammonia more likely to react as the nucleophile
outline what further substitution is
the amine product can also react with the halogenoalkane, as the N has its lone pair of electrons + can behave as a nucleophile → this leads to a mixture of haloalkanes being produced
outline the 4 stages/reactions that can take place with ammonia + bromomethane
stage 1: reaction between a halogenoalkane + ammonia produces the primary amine
stage 2: primary amine produced has a lone pair on N + can behave as a nucleophile
stage 3: secondary amine produced can also react with another halogenoalkane to produce a tertiary amine
stage 4: tertiary amine also has a lone pair on N, therefore further substitution is possible, which leads to quaternary ammonium salt
give the equation + draw the mechanism for stage 1 (between ammonia + bromomethane)
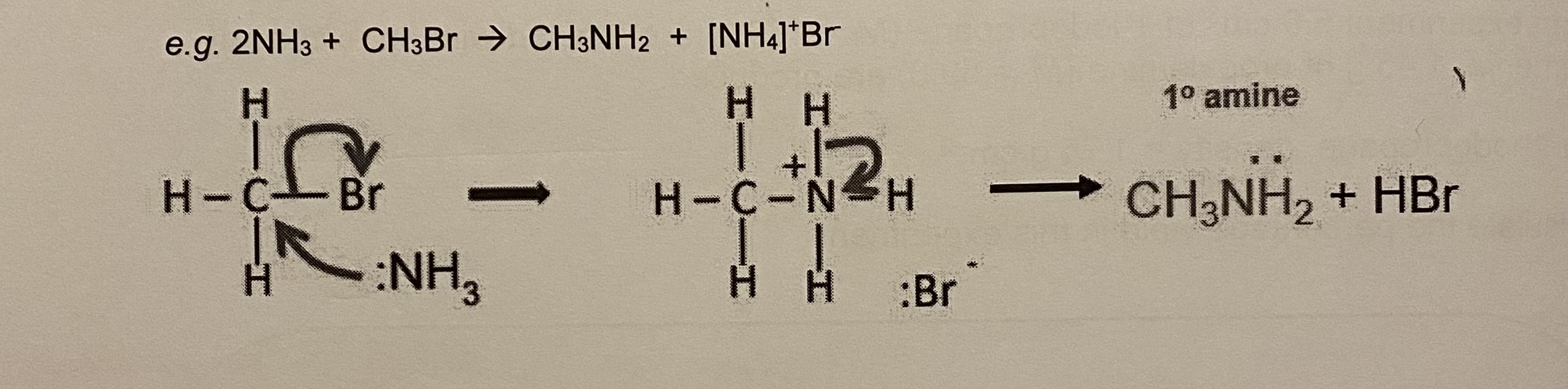
give the equation + draw the mechanism for stage 2 (between ammonia + bromomethane)
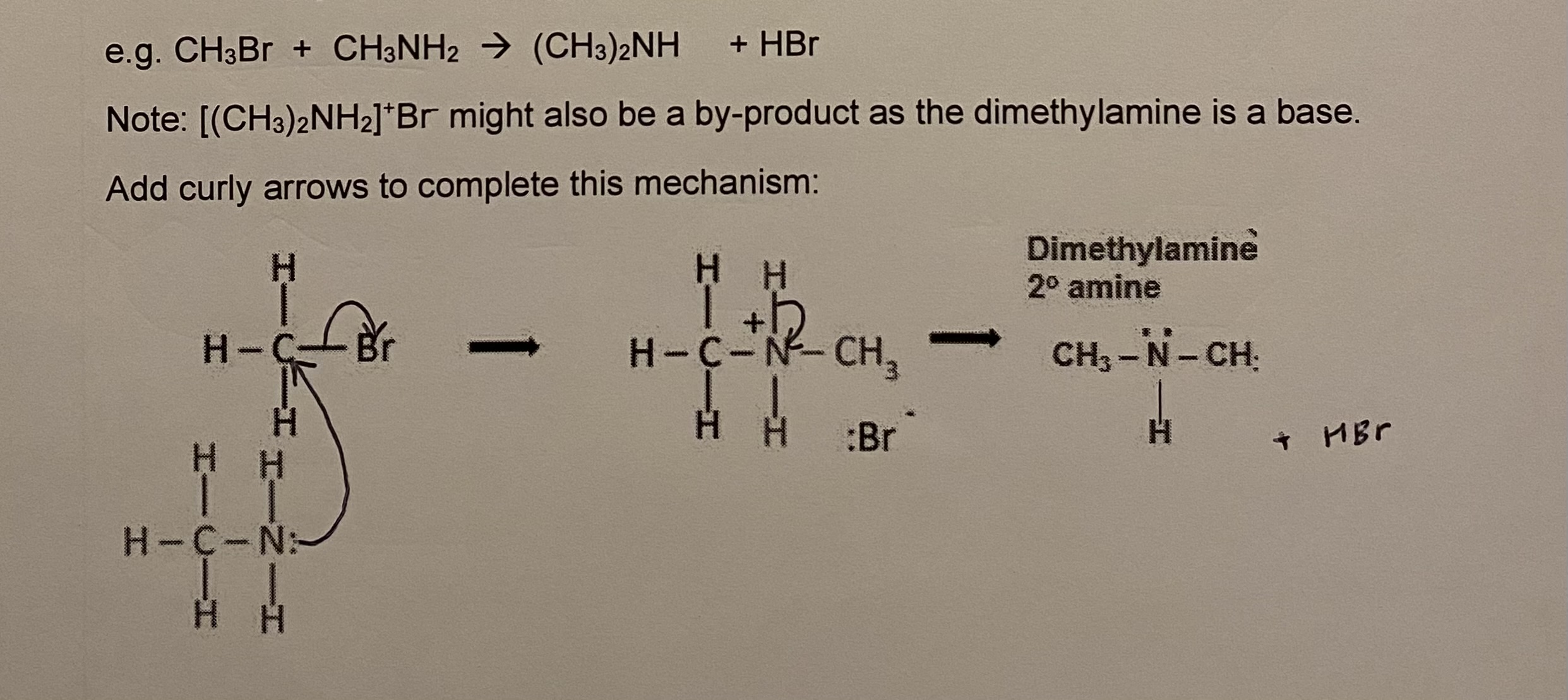
give the equation + draw the mechanism for stage 3 (between ammonia + bromomethane)
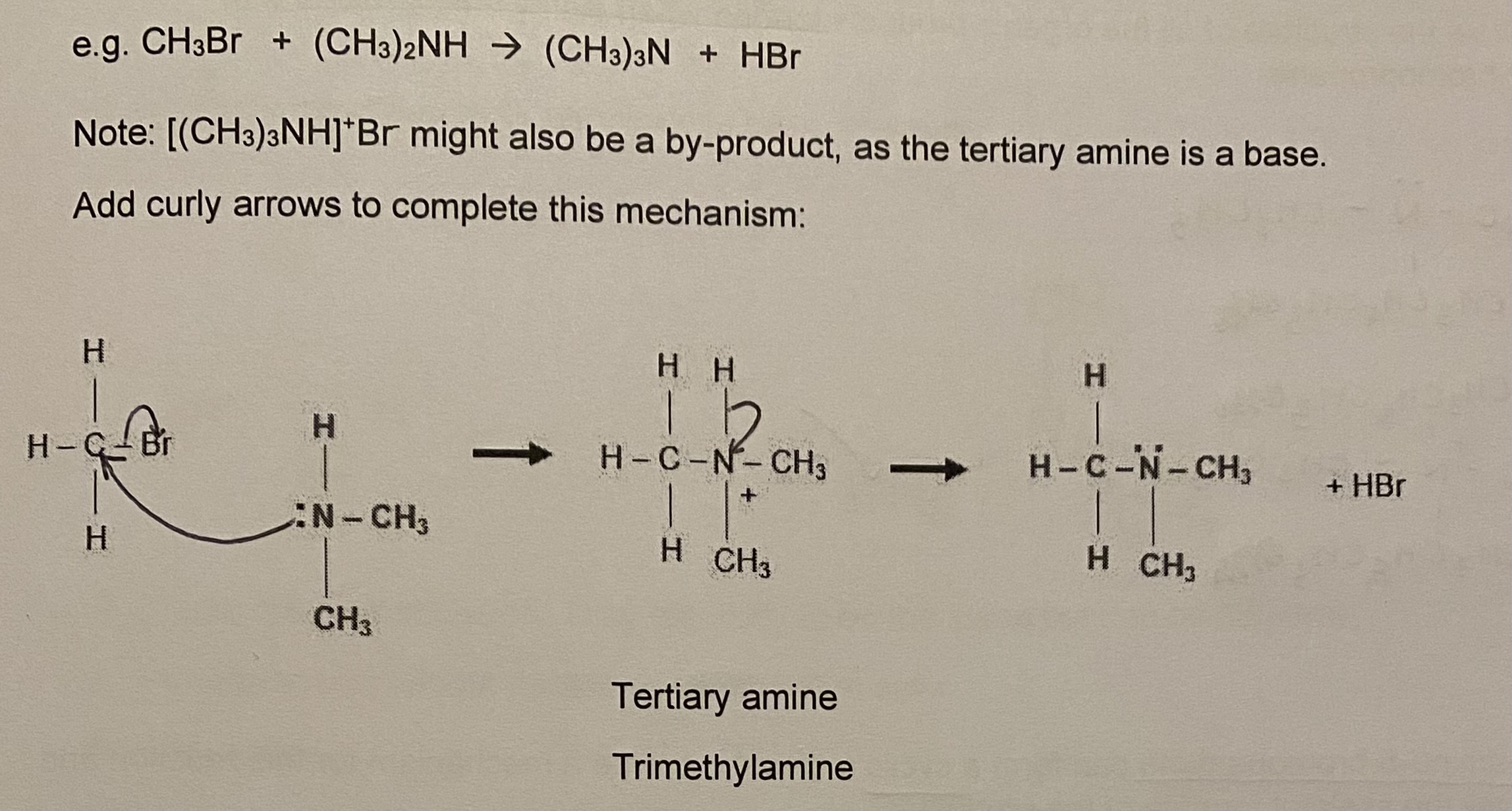
give the equation + draw the mechanism for stage 4 (between ammonia + bromomethane)
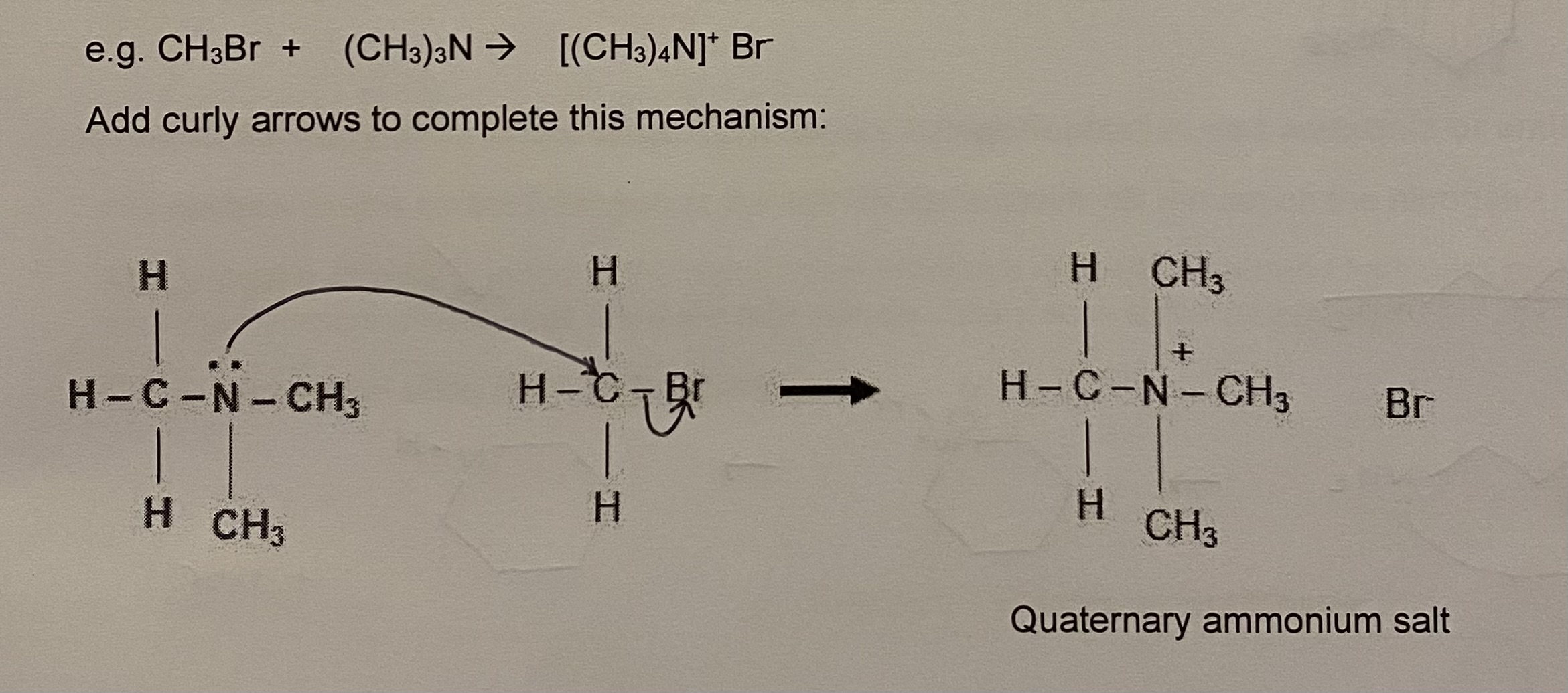
a. what does a large excess of halogenoalkane lead to?
b. what does a large excess of ammonia lead to?
a. high yield of quaternary ammonium salt
b. better yield of primary amine
give the basic structure of quaternary ammonium salts
the R groups are carbon chains → they are likely to be the same but can be different

what are quaternary ammonium salts used as?
as cationic surfactants
how are quaternary ammonium salts formed?
from excess halogenoalkanes + ammonia or amines
outline the changes that occurs to the reactants to form quaternary ammonium salts
any carbon chains on the nitrogen at the start of the reaction will remain on the nitrogen
all hydrogens directly bonded to the nitrogen are removed + extra carbon chains will be added from the halogenoalkanes until there are four carbon chains around the nitrogen
the X- will be the halide from the halogenoalkane
give the structure of the quaternary ammonium salt formed from methylamine + excess chloroethane
CH₃N(CH₂CH₃)₃⁺Cl⁻
ethylamine reacts with an excess of 1-bromopropane, what is the final product?
CH₃CH₂N(CH₂CH₂CH₃)₃⁺Br⁻
outline what amines are used in
in the manufacture of synthetic materials such as nylon, dyes + drugs
what are quaternary ammonium salts used as?
as cationic surfactants in fabric softening + hair products