Organic
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
CFC-11 is a greenhouse gas that can contribute to global warming. State and explain how CFC-11 is able to contribute to global warming. (2 marks)
absorbs infrared radiation
molecule has polar bonds
Catalytic Cracking
High temperature - 450oC
Zeolite (aluminosilicate) catalyst
Produces mainly branched alkanes and cyclic compounds with very few alkenes
Used primarily in motor fuels as branched compounds reduce ‘knocking’
Thermal Cracking
High temperature - 700oC
High pressure - 7000 kPa
Produces a high proportion of alkenes - the high temperature causes chain to break close to the end forming small alkenes such as ethene
Free radical process - homolytic fission of carbon chain
Explain why there is an attraction between a C=C double bond and Br2 (3 marks)
C=C electron rich / area of high electron density
Br-Br becomes polarised
δ+ Br attracted to C=C
Why a plasticiser is added (2 marks)
make it more flexible
this is because they get between the polymer chains and reduce intermolecular forces.
Chemists provided evidence that was used to support a ban on the use of chlorodifluoromethane as a refrigerant. Many refrigerators now use pentane as a refrigerant.
State the environmental problem that chlorodifluoromethane can cause.
Give one reason why pentane does not cause this problem. (2 marks)
Causes ozone depletion/decomposition/damage
Accept hole in the ozone layer
Pentane does not have C-Cl bonds
Accept does not produce Cl radicals
Accept does not contain chlorine
State a simple chemical test that distinguishes the propanoic acid from the propan-1-ol. Give one observation for the test with each substance. (3 marks)
add sodium carbonate/hydrogencarbonate
Propanoic acid: effervescence / bubbles not gives off (CO2) gas
Propan-1-ol: no (visible) change/reaction
Use evidence from the rate equation to explain why Step 1 is the rate determining step. (1 mark)
Step 1 contains all the species in the rate equation / Step 1 includes CH3COCH3 and OH- and these are also in the rate equation
The overall order of the reaction can be deduced from a piece of information in one of the column headings in the table. Identify this piece of information and deduce the overall order. (2 marks)
Unit of k (is s-1)
Order 1/first
This question is about simple test-tube reactions to identify organic liquids.
Silver nitrate solution can be used to distinguish between propanoyl chloride and 1-chloropropane. Give the observations you would expect when a few drops of silver nitrate solution are added to separate samples of propanoyl chloride and 1-chloropropane. (2 marks)
propanoyl chloride:
Misty / white / steamy fumes
(immediate) White precipitate forms
1-chloropropane:
No visible change
White precipitate forms slowly
Three unlabelled bottles are known to contain either propan-1-ol, propanal, or propanone.
A sample of each liquid is warmed with a few drops of Fehling’s solution.
Identify the liquid that reacts with Fehling’s solution and give the expected observation.
Suggest a further simple test-tube reaction that can be used to distinguish between the remaining two liquids.
Give the expected observation with the liquid that reacts. (3 marks)
Propanal AND blue solution gives a brick red precipitate
(Warm with) acidified potassium dichromate (VI) | add Na | warm with a named carboxylic acid with conc H2SO4 |
Propan-1-ol/alcohol AND (orange solution) goes green | Propan-1-ol/alcohol AND effervescence | Propan-1-ol/alcohol AND fruity smell |
The figure below shows enthalpy of hydrogenation data for cyclohexene and benzene. It also shows predicted data for the theoretical molecule cyclohexa-1,3,5-triene.
Compare benzene and the theoretical molecule cyclohexa-1,3,5-triene in terms of:
stability
shape
carbon–carbon bond lengths.
For each of these properties, suggest reasons for any differences. Use data from the figure above in your answer. (5 marks)
Benzene is more stable than cyclohexa-1,3,5-triene
The enthalpy of hydrogenation of benzene is (152 kJ mol-1) less/less exothermic
Due to the delocalisation of electrons in benzene
Both are planar/hexagonal
Benzene has equal C-C bond lengths or regular hexagon whereas Cyclohexa-1,3,5-triene has bonds of different/varied length or the hexagon is distorted/irregular
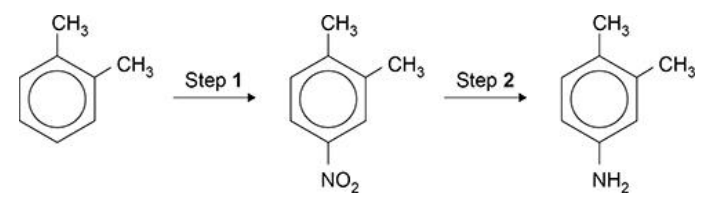
State a possible use for the amine formed in Step 2. (1 mark)
Manufacture of dyes/(cationic) surfactants/fabric softener
Allow to make hair/fabric conditioner
Benzene reacts with methanoyl chloride (HCOCl) in the presence of a catalyst. Give an equation for the overall reaction when benzene reacts with methanoyl chloride. Name the organic product. (2 marks)
C6H6 + HCOCl → C6H5CHO + HCl
Benzaldehyde
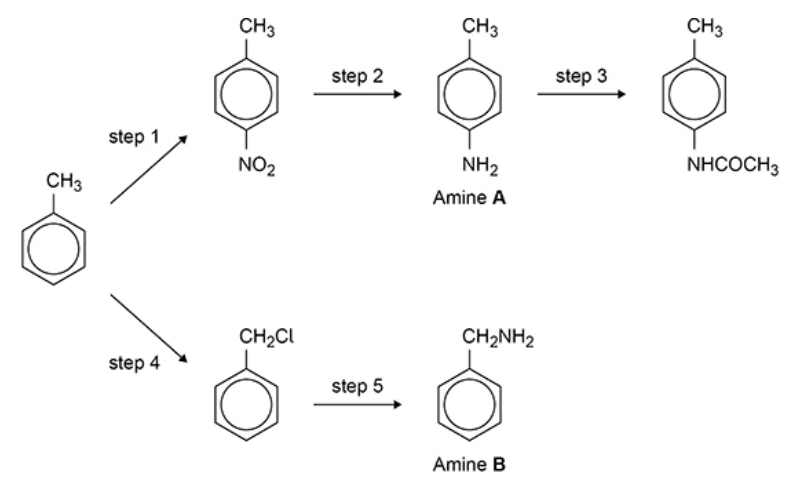
Amine A is formed in step 2 and amine B is formed in step 5. Explain why the yield of B in step 5 is less than the yield of A in step 2. (2 marks)
In Step 5 further substitution / gives other amine products
In Step 2 only one amine
Explain why amine B (previous flashcard) is a stronger base than amine A. (2 marks)
In B Alkyl group is electron donating or positive inductive effect
Or In A lone pair on N partially delocalised
Lone pair on N more available
Lone pair on N less available
A student hydrolyses a sample of endomorphin-2 (a peptide) to break it down into its constituent amino acids. The student analyses the resulting mixture by thin-layer chromatography, TLC.
State a reagent and the conditions needed for the hydrolysis. (2 marks)
(aqueous) HCl/hydrochloric acid
reflux/heat
Name a suitable developing agent. State why the developing agent is needed. (2 marks)
ninhydrin
amino acids are colourless / to make the amino acids visible
R represents a group that varies between different amino acids. R groups can interact and contribute to protein structure.
Explain why the strength of the interaction between two cysteine R groups differs from the strength of the interaction between a serine R group and an aspartic acid R group. Use the Data Booklet to help you answer this question. (4 marks)
Two Cys R groups form a disulfide bridge/link stated or described
Ser and Asp R groups form Hydrogen bonds
Disulfide bridges are stronger than H bonds
Because disulfide bridges are covalent bonds (while H bonds aren’t)
Deduce the type of interaction that occurs between a lysine R group and an aspartic acid R group. (1 mark)
Ionic bond