proteins and enzymes 2
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
why do proteins have diverse structures and functions
different amino acid sequences with different side chains
common features of ⍺-amino acids
⍺-carbon (chiral centre) with attachments:
an amine group (-NH2) which can be protonated
a carboxyl group (-COOH) which can be deprotonated
a hydrogen atom
a side chain of R-group
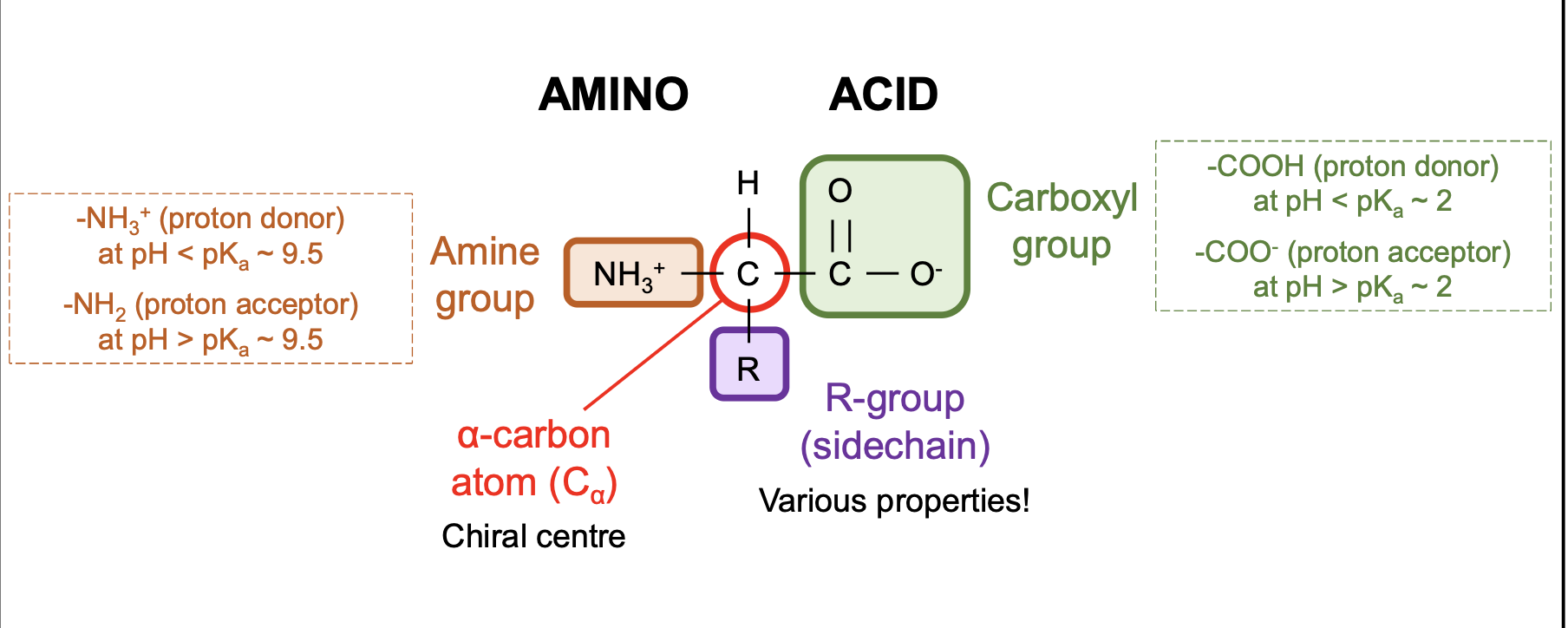
what determines amino acid properties
the R group (side chain)
why is the ⍺-carbon usually chiral
it is attached to four different groups
which amino acid is not chiral
Glycine
what is unusual about proline
its side chain forms a ring with the amine group
at low pH what form does the amino group take
protonated NH3+ (and COOH)
at high pH what form does the carboxyl group take
deprotonated COO- (and NH2)
zwitterion
at neutral pH, amino acid: NH3+ and COO-
enantiomers
non-superimposable mirror images - stereoisomers
which stereoisomer is used in proteins
L amino acids
why are ⍺-helices usually right handed
proteins contain L amino acids
Fischer projection
drawn with carboxyl group on top and side chain on the bottom
how to identify L amino acids in Fischer projections
amine group on the left
where were D amino acids originally thought to only exist in
bacteria - but are now known to occur across plants, invertebrates and vertebrates including mammals
what important free D amino acid acts in mammalian brains
D-serine
D-serine
found in mammalian brains
important signalingi molecules - could serve as early biomarkers
what receptor does D-serine act on
NMDA receptor
how do D-AA-containing peptides (DAACPs) arise
spontaneous isomerisation - certain amino acids, especially aspartic acid, can convert non-enzymatically into heir D form in aging, this can disrupt protein structure and function, D-Asp has been detected in ⍺A crystallin from cataract patient’s eye lenses, D-Asp also occurs in β-amyloid in the brains of Alzheimer’s patients
enzyme-catalysed post-translational modifications (PTMs) - Dermorphin, an opioid peptide from frog skin, contains D-Ala even though its gene encodes L-Ala, peptide isomerase activity has been found in frog skin secretions
non-ribosomal peptide synthesis - bacteria incorporate D-Ala and D-Glu onto their cell walls using specific enzymes, many antibiotics with D-AAs, such as penicillin (contains D-Val) and gramicidin S (contains D-Phe) are made by step by step specialised non-ribosomal peptide synthesases
which bacterial cell wall amino acids are D forms
D-Ala and D-Glu
what disease-associated protein contains D-Asp
β-amyloid
number of common amino acids
20
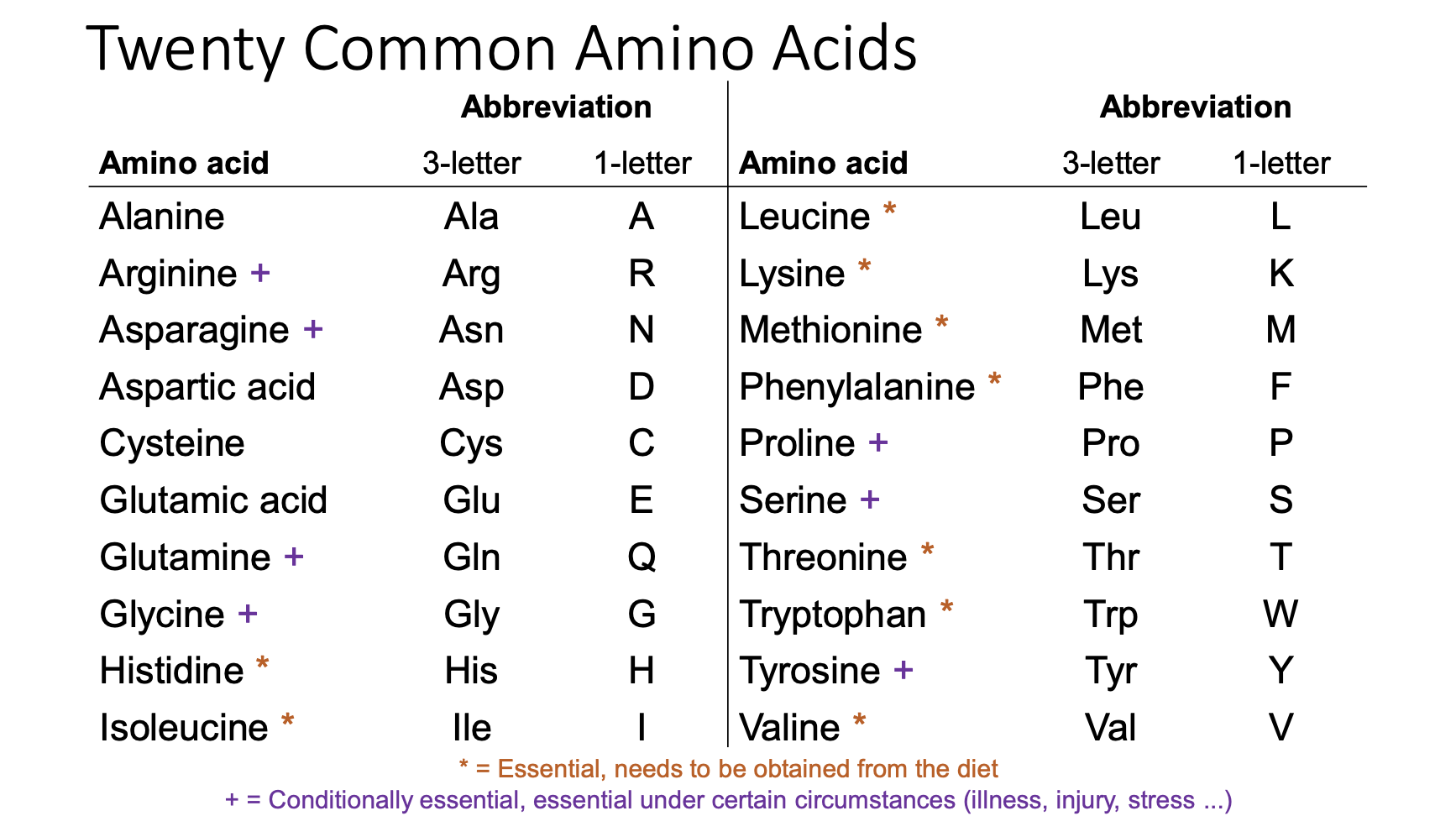
essential amino acids
must be obtained from diet:
Histidine
Isoleucine
Leucine
Lysine
Methionine
Phenylalanine
Threonine
Tryptophan
Valine
conditionally essential amino acids (essential under certain circumstances - illness, injury, stress)
arginine
asparagine
glutamine
glycine
proline
serine
tyrosine
non-essential amino acids
this enrich the body is capable of synthesising
phenylketonuria
an inherited metabolic disorder where ether body cannot properly convert the amino acid phenylalanine into tyrosine due to a deficiency in the enzyme phenylalanine hydroxylase (so tyrosine is conditionally essential)
this leads to a deficiency in the enzyme phenylalanine and low levels of tyrosine
the main treatments is a lifetime diet with very limited intake of foods with phenylalanine
non-proteinogenic amino acids (npAAs)
amino acids not incorporated into proteins
what percentage of total amine acids known to exist in nature do the 20 in proteins (plus selenocysteine and pyrrolysine) make up
~2%
what is L-DOPA derived from
tyrosine (and then L-DOPA is converted to dopamine in neurones)
what neurotransmitter is produced from L-DOPA
dopamine
what disease is treated with L-DOPA
Parkinson’s disease
felinine
found in urine of cats - associated with the characteristic cat urinary odour
derived from 3-MBCG with the help of cauxin
amino acid properties categories
hydrophobic
polar
positively/negatively charged
aromatic
aliphatic
small
tiny
C-β branching
negatively charged amino acids
glutamic acid - Glu - E
aspartic acid - Asp - D
positively charged amino acids
Histidine - His - H
Lysine - Lys - K
Arginine - Arg - R
small amino acids
Aspartic acid - Asp - D
Proline - Pro - P
Asparagine - Asn - N
Threonine - Thr - T
Valine - Val - V
tiny amino acids
Serine - Ser - S
Alanine - Ala - A
Glycine - Gly - G
Cysteine - Cys - C
small and tiny amino acids
C-β branching
Threonine - Thr - T
Valine - Val - V
Isoleucine - Ile - I
aliphatic amino acids
Valine - Val - V
Isoleucine - Ile - I
Leucine - Leu - I
essential amino acids
Histidine - His - H
Isoleucine - Ile - I
Leucine - Leu - L
Lysine - Lys - K
Methionine - Met - M
Phenylalanine - Phe - F
Threonine - Thr - T
Tryptophan - Trp - W
Valine - Val - V
(Hot Wet MILF Takes Kielbasa)
hydrophobic amino acids
Methionine - Met - M
Isoleucine - Ile - I
Leucine - Leu - L
Phenylalanine - Phe - F
Valine - Val - V
Alanine - Ala - A
Glycine - Gly - G
Proline - Pro - P
Tryptophan - Trp - W
(MILF VAG Pisses Water)
E484K mutation
Glu → Lys subsitituoin at positon 484
which amino acids contain sulphur
M and C
amphipathic (have polar and non-polar character) amino acids
C
T
W
Y
K
H
(tiny wet cunts have yellow knots)
which amino cid is tiny and flexible and hwy does that make it structurally important
Glycine - fits into tight turns inaccessible to other residues
which amino acids are branched-chain amino acids (and also aliphatic)
V
L
I
(M not classified as aliphatic as it is very small)
which amino acids contain hydroxyl groups (polar, uncharged)
S
T
which amino acids contain amide groups (polar, uncharged)
N
Q
polar, uncharged amino acids
S (hydroxyl group)
T (hydroxyl group)
N (amide group)
Q (amide group)
acidic amino acids (negatively charged)
D
E
basic amino acids (positively charged)
K (second amino group)
R (guandino group)
H (imidazole group)
where are polar and charged amino acids found
usually on surface of proteins, when buried in the protein they are involved in salt bridges
why is histidine special
pKa near physiological pH - this means it is important in enzymes as it can serve as a proton donor/acceptor
aromatic amino acids (relatively non-polar and participate in hydrophobic interactions)
F
Y
W
Y & W are significantly more polar than F because of the hydroxyl group in Y and the nitrogen of the W indole ring
Y & W and to a much lesser extent F, absorb ultraviolet lights - accounts for the characteristic strong absorbance o flight by most proteins at a wavelength of 280nm
wavelength of light all AAs absorb due to the peptide bond
190-220nm
proline structure
sidechain forms a cyclic ring with backbone nitrogen - a five-membered nitrogen-containing ring
also has a NH2+ rather than an NH3+ group in its isolated form at physiological pH
how many times is the proline side chain connected to the backbone
twice - this ring structure holds proline residues in a rigid conformation, reducing the structural flexibility of polypeptide regions contains proline
effect of proline on flexibility
reduces flexibility
why is proline often found in turns
rigid strutcure favours turns - meaning prolines are often found on the protein surface
which peptide bond configuration is more common generally
trans
which amino acid forms cis peptide bonds
proline - ~6% of the bonds are in cis configuration
what bond forms between two cysteines
disulphide (cysteine is readily oxidised to form covertly linked dimeric amino acid called cystine)
cystine
the oxidised cysteine dimer
role of disulphide bonds
covalent stabilisation of protein structure
PTMs
post translational modifications - chemical modifications after translation catalysed by specific enzymes which target specific residues
common PTMs
phosphorylation (on S, T, Y residues) —-
hydroxylation (P, K)
acetylation (S, T, K) —-
methylation (K, E)
carboxylation (E)
AMPylation (T, Y)
N- or O-glycosylation (S, T, N) —-
lipidation (C, S, K)
disulphide bond formation (C)
ubiquitylation (K) —-
SUMO-ylation (K)
proteolysis and deamination (N, Q) - irreversible PTMs —-
enzyme which adds phosphate groups (phosphorylation)
kinase
enzyme which removes phosphate groups (dephosphorylation)
phosphatase
effect of histone acetylation
opens chromatin
enzyme in histone acetylation
acetyl transferases transfer acetyl groups from acetyl-CoA to lysine residues on histones, neutralising their positive charge - this weakens the DNA, leading to a less condensed and more accessible chromatin structure
enzyme which removes acetyl groups from histones
histone acetyl detransferases - removes acetyl groups form lysine,, causing the chromatin to compact
erythropoietin glycosylation
the blood-stimulating hormone erythropoietin has N- and O- linked oligosaccharides contributing to its function and stability
ubiquitylation
involves the addition of a small protein called ubiquitin on to other proteins
involves a large family of proteins, the E2 and E3 ligases
deubiquitinating enzymes reverse this process
most associated with protein degradation
proteases
enzymatically hydrolyse peptide bonds in substrate proteins, resulting in a widespread, irreversible PTM if the protein’s structure and biological function
so the proteins re irreversibly cleaved
proteolysis of prothrombin
cleaved at two sites and converted to active thrombin
prothrombin is rich in 𝛾-carboxyglutamate which can bind to Ca2+ ions, crucial for the activation of prothrombin to thrombin
collagens
proteins that form fibrous structures and are abundant in the body - present in skin, blood vessel walls, bones, teeth and more
collagen molecule
three polypeptide chains with many repeating Gly-X-Y sequences where X is often proline and Y is often hydroxyproline
the three chains coil together in a tight triple helix
every third residue passes though the crowded centre of the hex, where only GLy can fit
the bulky, rigid proline and hydroxyproline residues give the entire structure its strength and stability
main repeating collagen sequence
Gly-X-Y
where X is often porcine and Y is often hydroxyproline
why si glycine essential in collagen
only residue small enough for triple helix centre
hydroxyproline generation
by PTM of proline by the enzyme proline hydroxylase which requires vitamin C (ascorbic acid) for activity
vitamin C deficiency causes
lower levels of hydroxyproline and therefore collagen fibres cannot form, leading to scurvy
scurvy
characterised by fragile blood vessels, skin lesions, poor wound healing and if untreated, death
vitamin C deficiency
21st and 22nd amino acid
Selenocysteine - Sec - U
Pyrrolysine - Pyl - O
stop codon which encodes selenocysteine
UGA
stop codon which encodes pyrrolysine
UAG
where is selenocysteine found
in active sites of a small number of selenoproteins in archea, bacteria and eukaryotes
where is pyrrolysine found
in a small number of row methanogenic archea and bacteria
Pyl synthesis and incorporation
from Lys by three enzymes
ligated directly to tRNAPyl and inserted into proteins in response to UAG codons
Sec synthesis and incorporation
synthesised from Set on its cognate tRNA (tRNASec) by one enzyme in bacteria nd two in archaea and eukaryotes
during translation, Sec-tRNASec is delivered to the ribosome by a specific elongation factor (via another protein in archaea and eukaryotes) that requires a characteristic stem-loop structure in the mRNA to actively recode an inframe UGA from stop codon to Sec sense codon
pKa
pH where acid is 50% dissociated
relationship between pH and pKa
pH = pKa + log10(A-/HA)
the lower the pKa, the … the acid
stronger - and the more easily it dissociates
if pH < pKa, which form dominates
protonated form
if pH > pKa, which form dominates
deprotonates form
zwitterion
molecule with both positive and negative charges but net charge 0
alanine net charge at low pH
+1 - acid dissociation constant at ~2 for the -COOH group so above 2 it becomes COO-
alanine net charge at high pH
-1- acid dissociation constant at ~9.5 for the NH3+ so above 9.5 it is NH2
isoelectric point (pl)
pH where net charge = 0
pKa of side chain of aspartic acid
~4