IR Spectroscopy more detailed thank anki
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What does IR measure?
How much Infrared radiation different bonds in a molecule absorb at different frequencies.
What are the conditions for a bond to absorb IR radiation?
IR frequency must match the natrual frequency of the bond vibration
Bond must undergo a change in dipole moment as it vibrates
Why do different covalent bonds give different peaks in infrared spectroscopy?
Because they vibrate at different frequencies (speeds) due to mass and bond strength.
What determines the frequency (speed) at which a covalent bond vibrates?
Strength of bond (stronger bond = higher vibration frequency eg. C≡C)
Combined mass of bonding atoms. (lower mass = higher vibration frequency)
What does IR spectroscopy exploit?
Vibration of covalent bonds joining atoms to reveal how they’re connected
Types of covalent bond vibrations?
stretching
scissoring
rocking
wagging
twisting
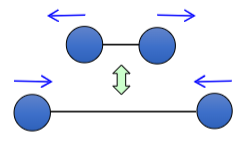
How is this bond vibrating?
stretching
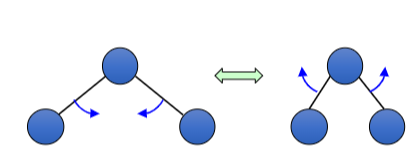
How is this bond vibrating?
scissoring
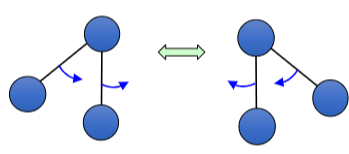
How is this bond vibrating?
Rocking
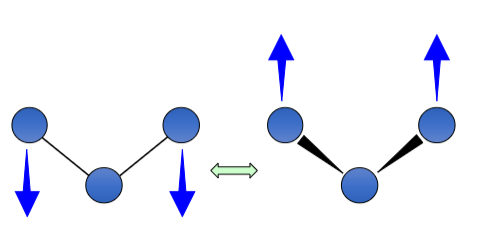
How is this bond vibrating?
Wagging
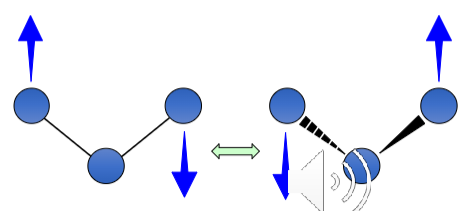
How is this bond vibrating?
Twisting
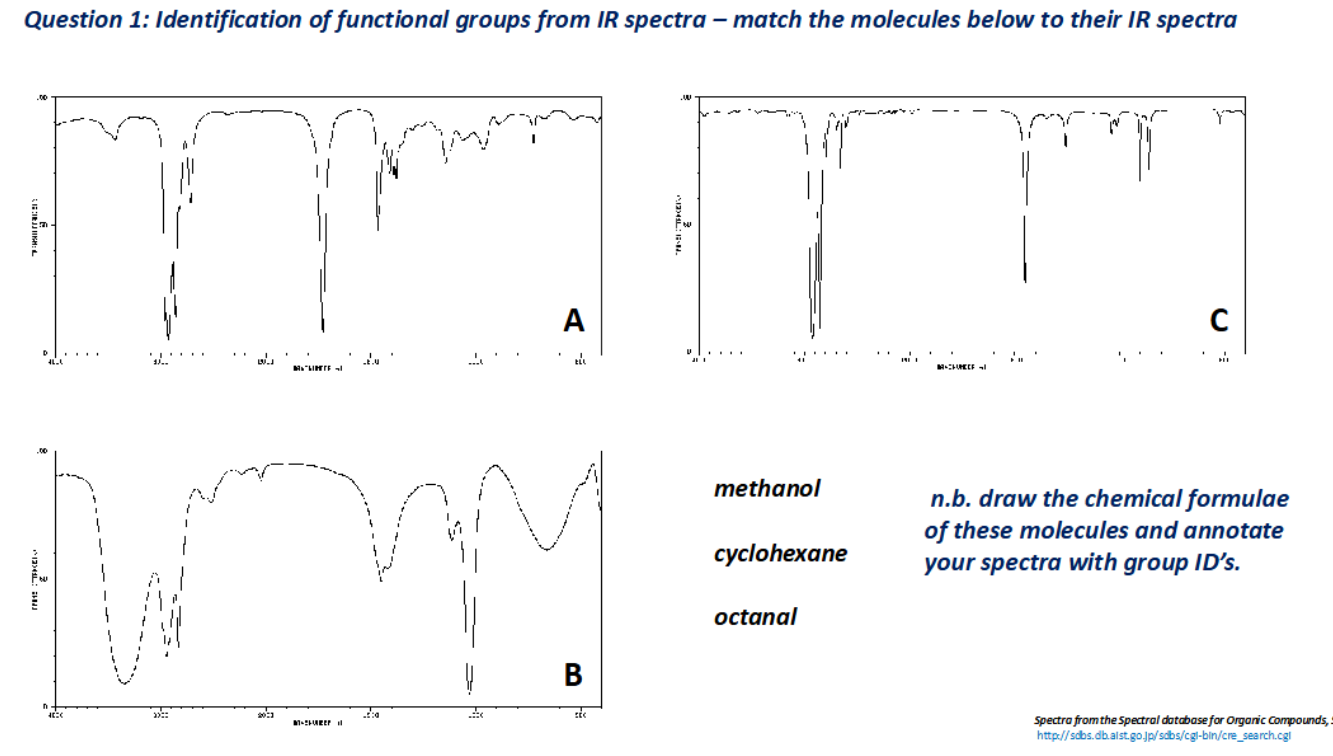
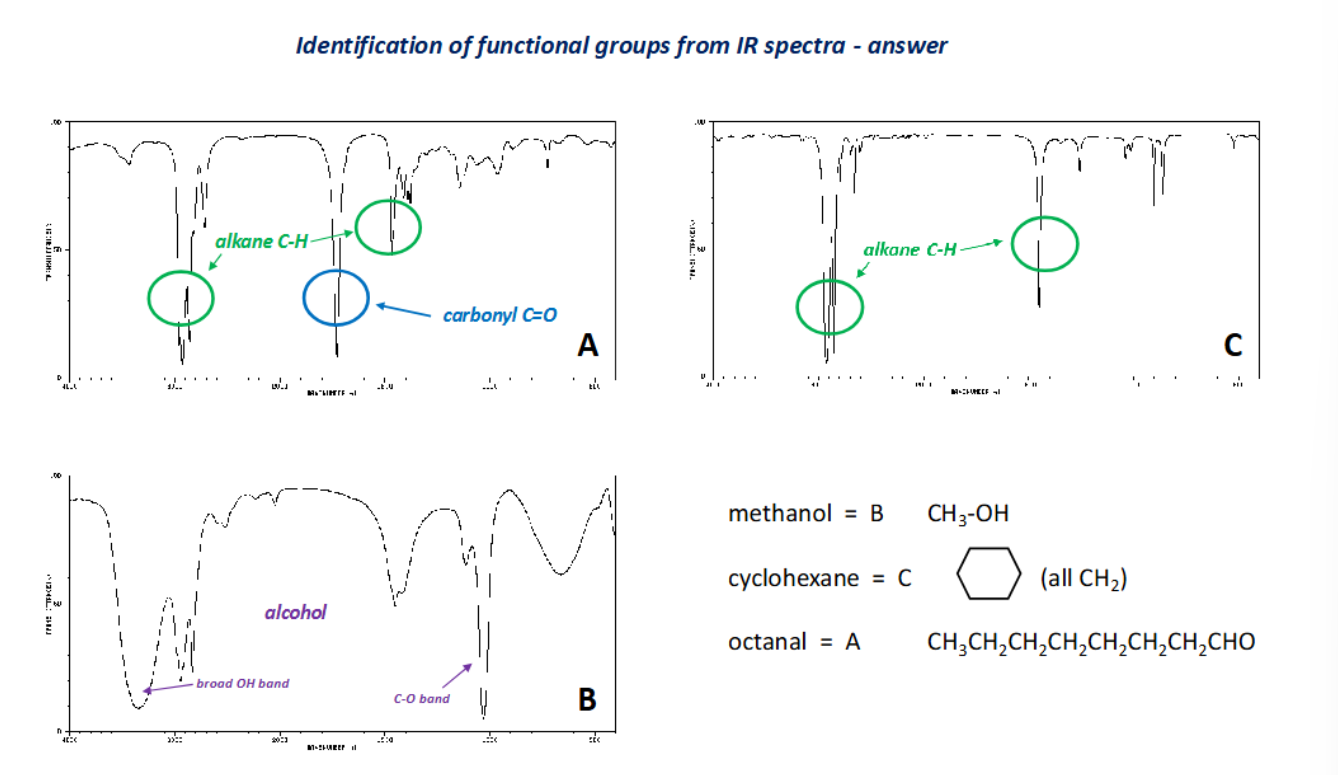
What technique is used to find the identity of functional groups attached to the hydrocarbon framework?
IR spectroscopy
Rank the following bonds according to the wavelength of infrared light that they absorb, with 1 being the shortest wavelength, and 3 being the longest wavelength.:
C-F , C-I , C-Cl
C-F
C-Cl
C-I
(Vibration frequency depends on mass of atoms and bond strength. Low frequency = longer wavelength. C-F is lightest so has shortest wavelength + highest frequency)
Rank the following wavenumbers according to the energy they represent, with 1 = highest energy, and 3 = lowest energy.
2300 cm-1
1200 cm-1
4000 cm-1
4000 cm-1
2300cm-1
1200cm-1
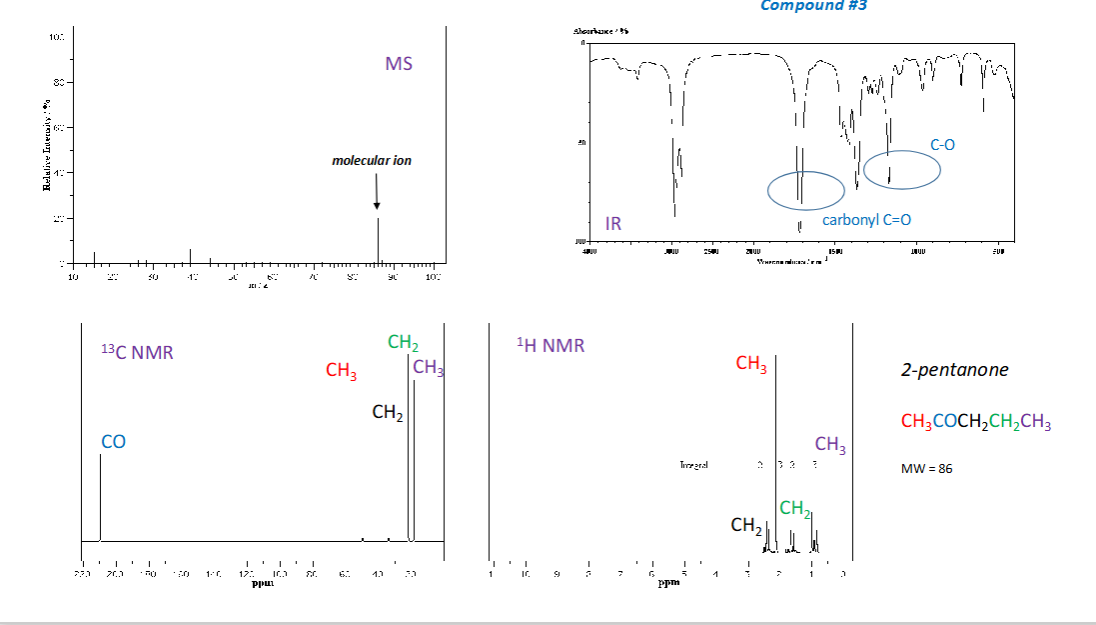
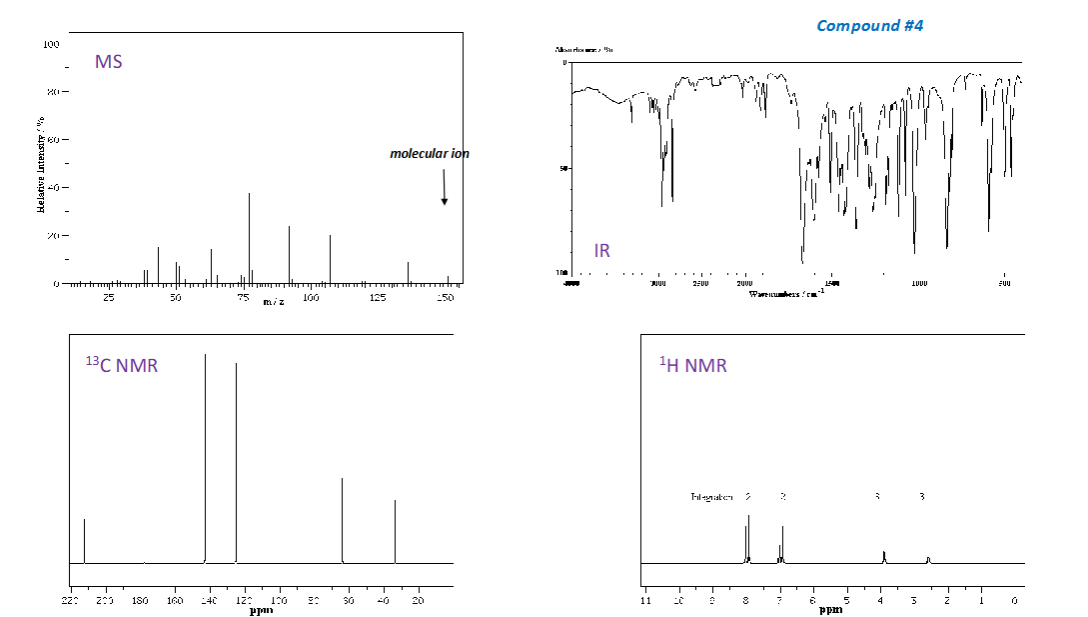
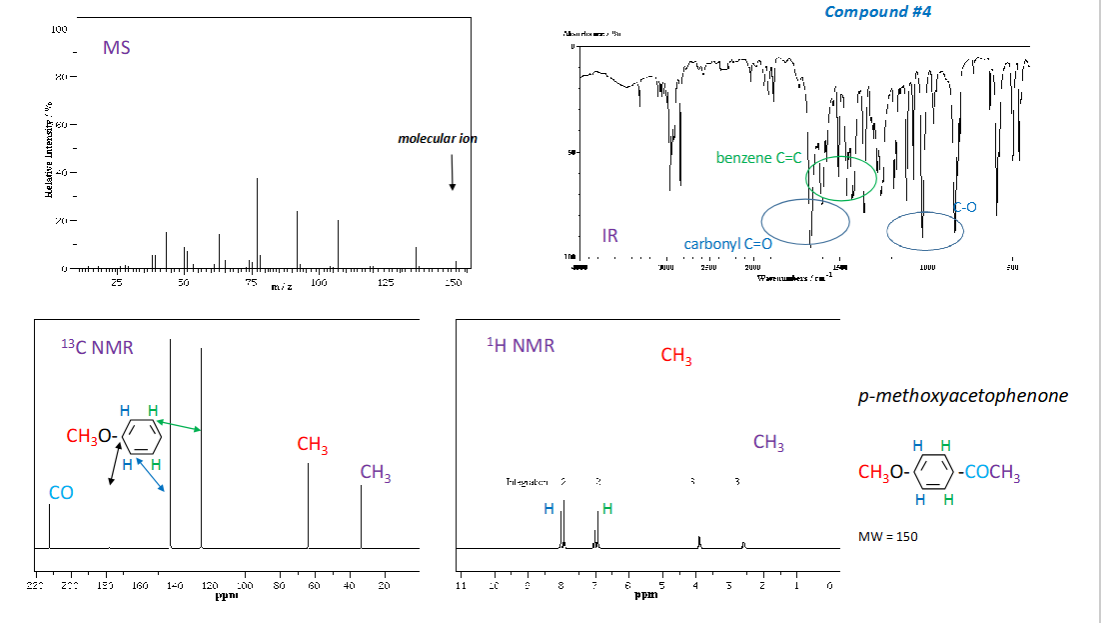
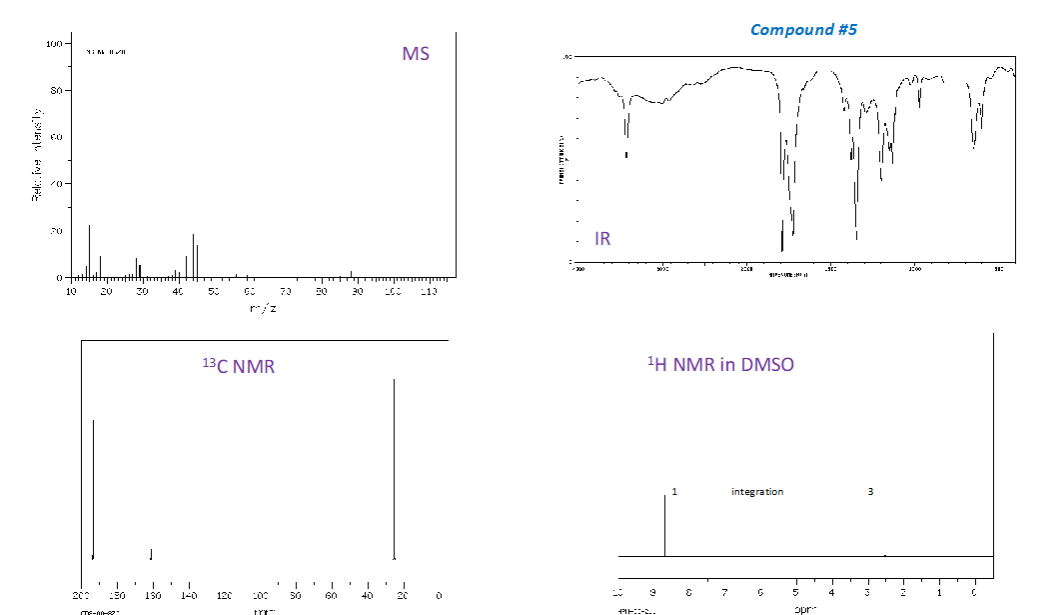
How are wavelength and wavenumber related?
wavelength = 1/wavenumber.
What is on the X axis of an infrared radiation spectroscopy graph?
Wavenumber (cm-1)
does a higher wavenumber mean higher or lower energy?
High wavenumber = high energy