College Biology Test #2
1/39
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Amino Acids
Building blocks and monomers of proteins
Basic Formula for an Amino Acid
R-CNH2-COOH

Number of Amino Acids
Only 20 amino acids are used by living things.
Categories of Amino Acids(based on R group properties)
polar uncharged, electrically charged, and apolar.
Protein
Amino acid polymer.
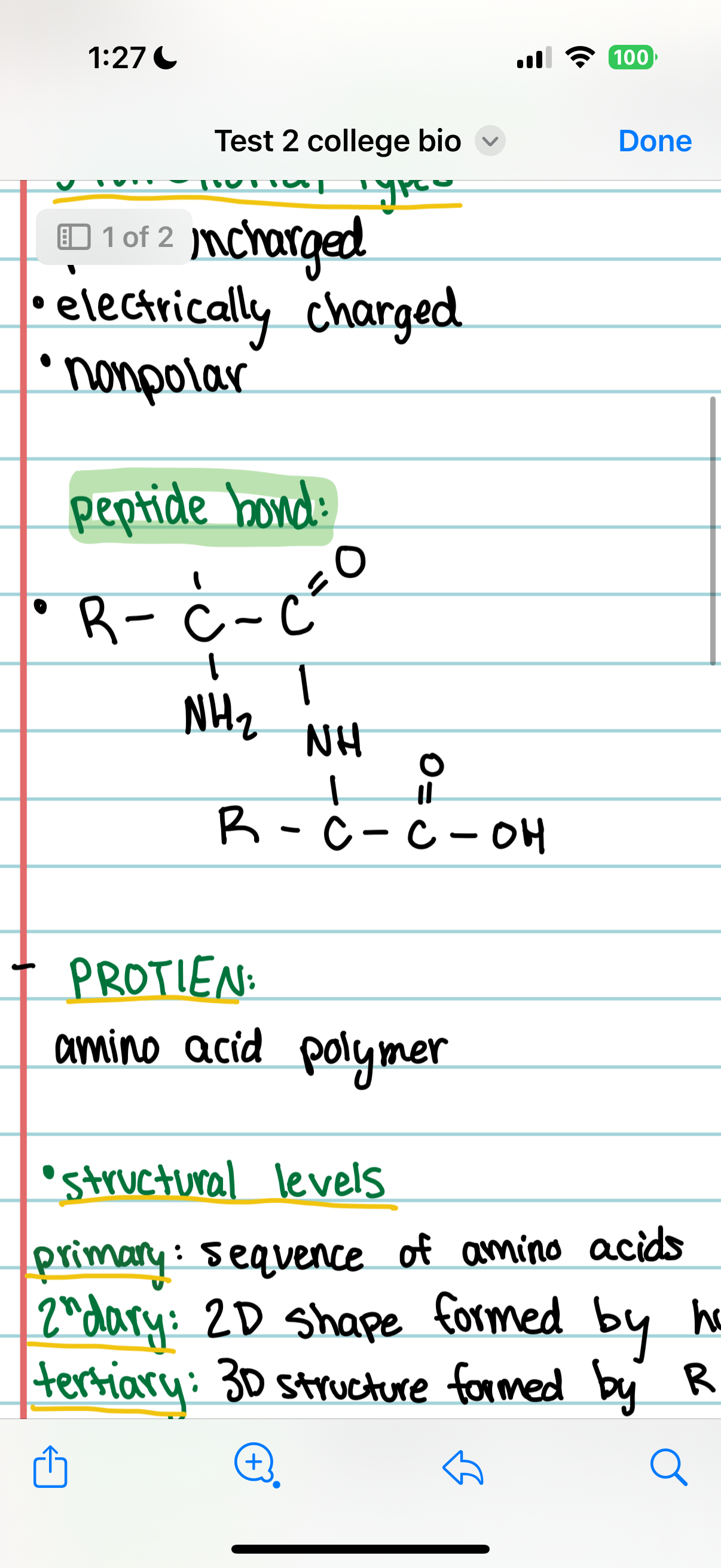
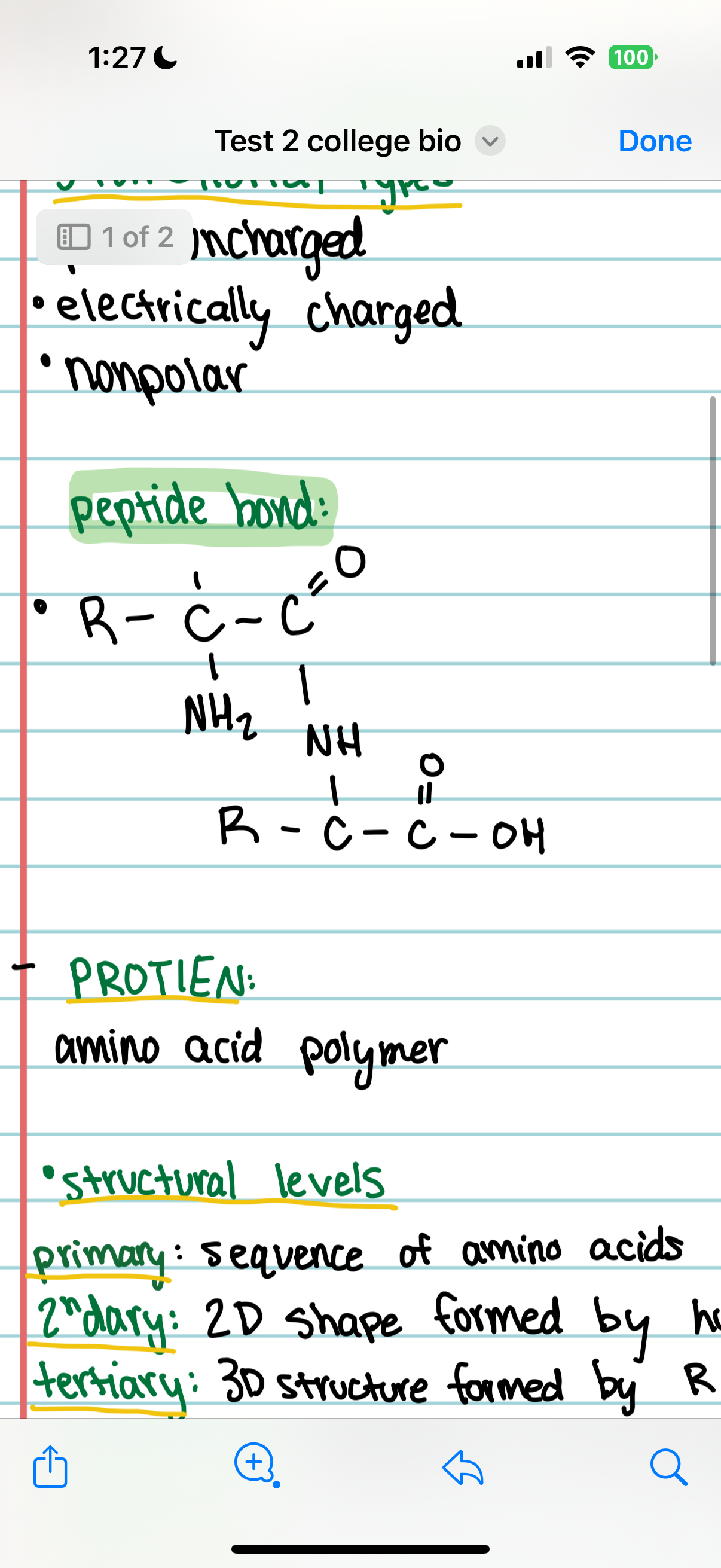
Primary Structure
The order in which amino acids are joined together in a protein.
Secondary Structure
Regular coils and folds formed by hydrogen bonding between amino acids, creating a 2D structure.
Tertiary Structure
The 3D structure of a protein resulting from interactions between R-groups.
quaternary structure
the joining of two or more proteins
Denaturation
The process that changes a protein's preferred conformation, inhibiting or inactivating it(can be permanent or temporary).
Causes of Denaturation
High temperatures, excessive salinity, and changes in pH.
Enzymes
Protein catalysts that facilitate a wide range of biological processes.
Catalyst Properties
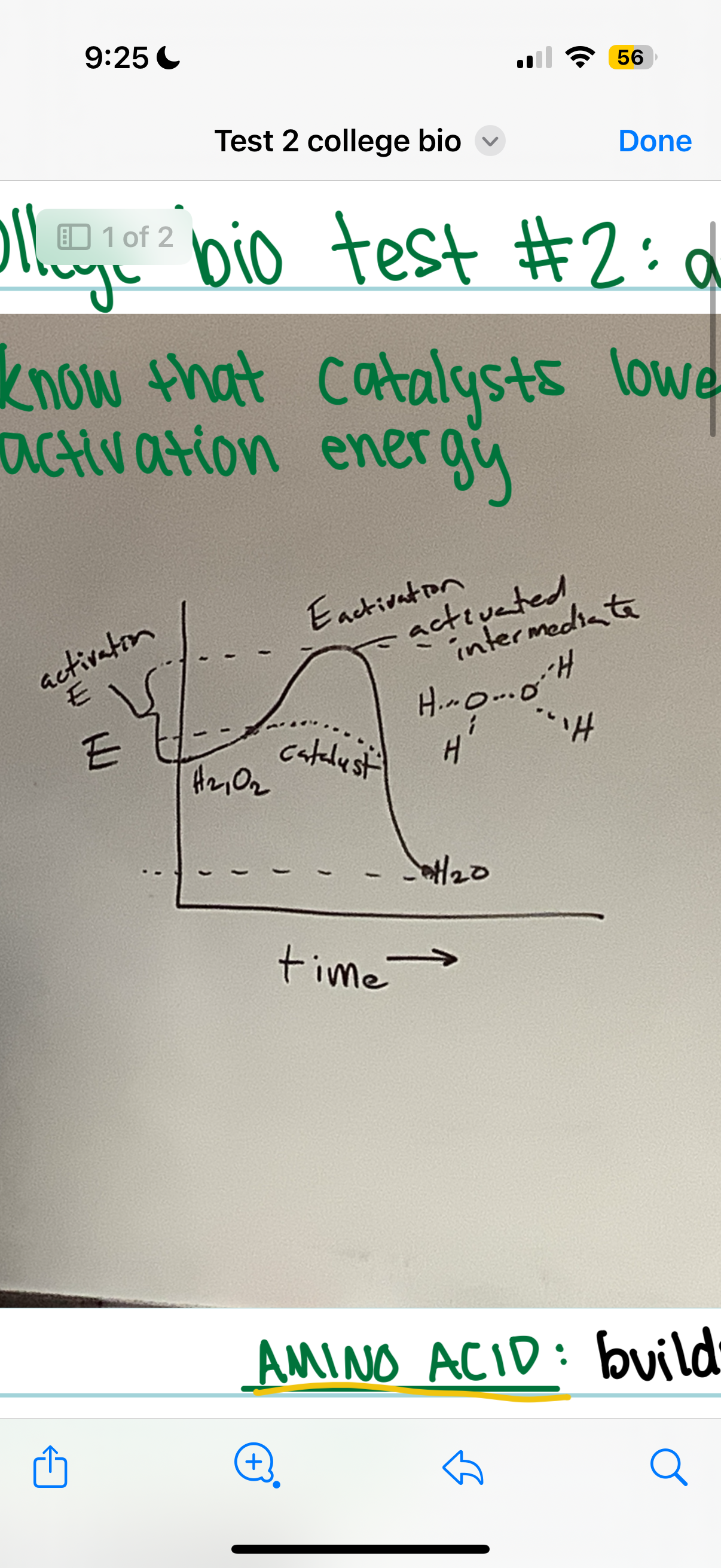
Catalysts alter the rate of chemical reactions(usually speeds up) without being consumed and lower the activation energy
Enzyme Function
Depends on an exact conformation.
Enzyme Structure
Consists of a structural region(the enzyme), a binding site, and the substrate
Lock and Key Model
Describes how enzymes function by fitting substrates into their active sites.
Nucleotides
Composed of phosphate, a pentose sugar, and the nitrogenous base(know the order)
Types of Nucleotides
adenine, thymine, uracil, guanine, and cytosine.
Complementary Base Pairing
A binds to T and U; C binds to G.
Nucleotide Polymers
Form DNA and RNA.
ATP
The ultimate energy source for all cell activities.
ATP Structure
Composed of adenosine triphosphate (three phosphate) groups (A-P-P-P).
Energy Release
ATP decays to ADP and releases energy when the unstable P-P bond breaks.
Conformation
the 3 dimensional shape for any given protein, and allows it to function most effectively/efficiently
Ribose Structure
5 Carbons, four of which are bonded to an OH, and the last Carbon is double bonded to an O
Deoxyribose
5 Carbons, one is empty, 3 are single bonded to an OH, and the last is double bonded to an O
Nitrogenous Base
Purines and pyrimidines
Adenine(A)
Ribose OR deoxyribose
Thymine(T)
ONLY deoxyribose
Uracil(U)
ONLY ribose
Guanine(G)
ribose AND deoxyribose
Cytosine(C)
ribose AND deoxyribose
GTP(Guanosine Triphosphate)
Equivalent to ATP, but not used without ATP or even used much at all
Catalyst properties
do not favor a reaction direction, not use up in reaction, and chemically selective
substrate imitators
AGonist: partial enzyme response
ANTagonist: bind, but no response
substrate binding
substrates, agonists, and sometimes antagonists bind weakly and repeatedly attach and reattach
what do catalysts do
lower the activation energy
peptide bond
peptide bond
dipeptides
two amino acids joined by a peptide bond