3.9- Acid-Base Equilibria
1/81
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
Define acid
A substance that releases or provides H+ ions, i.e. A proton donor
What definition is this?
The Lowry-Bronsted definition
Define base
A substance that removes or accepts H+ ions, i.e. A proton acceptor
Define salt
When the H+ ion in an acid is replaced by a metal ion or the ammonium ion
pH
-log10[H+]
Define weak acid
An acid that partially dissociates in solution
Define strong acid
Acids that dissociate completely
Calculate the pH of these strong acids:
0.2moldm-3
2.00 moldm-3
0.125 moldm-3
0.7
-0.3
0.9
Calculate the pH of these strong alkalis:
0.2moldm-3 KOH
2.00 moldm-3 aqueous NaOH
0.00562 moldm-3 aq caesium hydroxide
13.3
14.3
11.7
What is the acid dissociation constant?
Ka = [H+][A-]/[HA]
Where is this derived from?
The equilibrium constant: HA <=> H+ + A-
When is Ka larger?
When there is greater dissociation of the acid, as there will be more hydrogen ions and anions
So a strong acid will have?
A high Ka
How do you calculate pKa?
-log10(Ka)
What is the pKa of a strong acid?
Small
Weak acid?
Larger
What is the equilibria for water?
H2O <=> H+ + OH-
What is the equilibrium constant for water?
Kc= [H+][OH-]/[H2O]
What is Kw?
The ionic product of water
Kw=
[H+][OH-]
What is the value of Kw at 298K?
1x10^-14
What must be the same in pure water?
The concentration of H+ and OH-
So what is the pH of pure water? (calculate)
Kw= 1 x 10^-14= [H+]^2
[H+]= Root 1 x 10^-14
[H+]= 1 x 10^-7
pH= -log10 ( 1 x 10^-7)
pH= 7
Why can you use the conc of H+ ions in calculations for strong acids but not weak?
Because all of the H+ ions are dissociated in strong acids but they're only partially dissociated in weak
Equation for dissociation of ethanoic acid:
CH3COOH <=> CH3COO- + H+
If the Ka value is 1.7 x 10-5, what is the conc of H+ ions of a 0.5moldm-3 solution?
Ka= [CH3COO-][H+]/[CH3COOH]
Ka= [H+]^-2/[CH3COOH]
[H+]= Root Ka x [CH3COOH]
[H+]= Root 1.7 x 10^-5 x 0.5
[H+]= 0.00292 moldm-3
pH= 2.53
Work out the Ka of a weak acid if a 2.0moldm-3 solution has a pH of 4.5?
pH= -log10[H+]
[H+]=10^-4.5
=3.2 x 10^-5
Ka= [H+][A-]/[HA]
Ka= (3.2 x 10^-5)^2/2
Ka= 5.12 x 10^-10 moldm^-3
What happens when strong bases dissolve in water?
All of their OH- ions are produced
What does this mean?
The conc of the base gives the conc of the OH-
e.g. in 1.0moldm-3, [OH-]= 1.0
How can you then work out the conc of H+ ions in the base?
Kw= [H+][OH-]
Eg. what is the pH of a solution of NaOH of concentration 0.1 moldm-3?
[H+]= Kw/[OH-]
[H+]= 10^-14/0.1
[H+]= 10^-13
pH= -log10 [10^-13]
= 13
What is a buffer?
Resists changes in pH as small amounts of acid and alkali are added
What is a buffer solution made from?
A weak acid e.g. CH3COOH
A salt of the same acid with a strong base e.g. CH3COO-Na+
How does this work?
There is a high concentration of the anion as the sodium salt dissociates completely:
CH3COONa -> CH3COO- + Na+
The weak acid dissociates partially:
CH3COOH <=> CH3COO- + H+
What happens when an acid is added to a buffer?
The conc of H+ is increased, and this causes the equilibrium to shift to the left, removing the H+ by reaction with CH3COO-
What happens when an alkali is added to a buffer?
Removes some of the H+ ions present, so equilibrium shifts to the right, producing H+ ions
What are buffers used for in biological systems?
Using or storing enzymes, to ensure pH remains at the optimum value
Storage of biological molecules e.g. pharmaceuticals, which will be denatured at the incorrect pH
What buffer is used from making alkaline pH?
NH4Cl -> NH4+ + Cl-
What is the key equilibrium for this?
NH4+ <=> NH3 + H+
What does the addition of a base cause?
Removal of H+ ions and this causes equilibrium to shift to the right to produce more H+ ions
What do we need to know to calculate the pH of buffers?
The Ka value of the weak acid
The ratio of the concentration of the acid and salt
What expression is used?
[H+][A-]/[HA]
What can be assumed?
[HA] is equal to the conc of acid used
[A-] is equal to the conc of the salt
Work out the pH of a buffer that contains equal concentrations of ethanoic acid and sodium ethanoate (Ka= 1.7 x 10^-5)
Ka= [H+] x [CH3COO-]/[CH3COOH]
1.7 x 10^-5= [H+] x 1
[H+]= 1.7 x 10^-5
pH= -log10^ 1.7 x 10^-5
pH= 4.77
What is the Henderson-Hasselbalch equation?
pH (buffer)= pKa + log ([salt]/[acid])
When the conc of salt= conc of the acid, pH=
-log10Ka = pKa
What will a salt produced from a strong acid and a weak alkali be?
An acidic salt
Weak acid and strong base?
Basic salt
What do the ions released from some salts react with water molecules to do?
To release or remove H+ ions
What is this called?
Salt hydrolysis
Talk about dissolving NH4Cl
Produces free NH4+ and Cl- ions. The equilibrium NH4+ <=> NH3 + H+ will occur in solution, releasing free H+ ions in solution, making it slightly acidic.
What happens when CH3COONa dissolves in water?
Produces CH3COO- and Na+.
CH3COO- + H+ <=> CH3COOH occurs, removing H+ from the water. As conc of H+ decreases, the pH increases slightly.
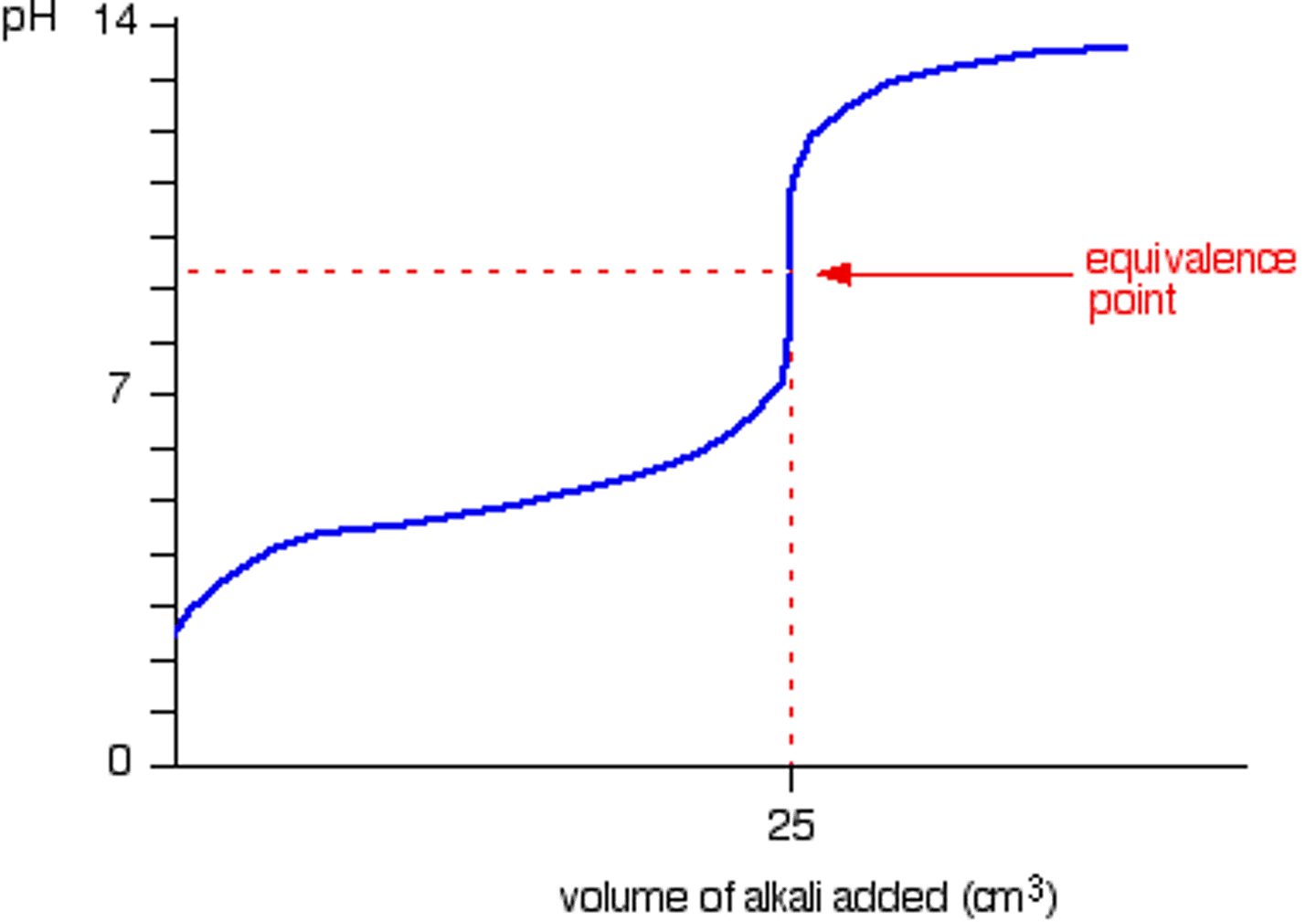
What does a titration curve for strong acid-strong base look like?
Starts very low
Ends very high
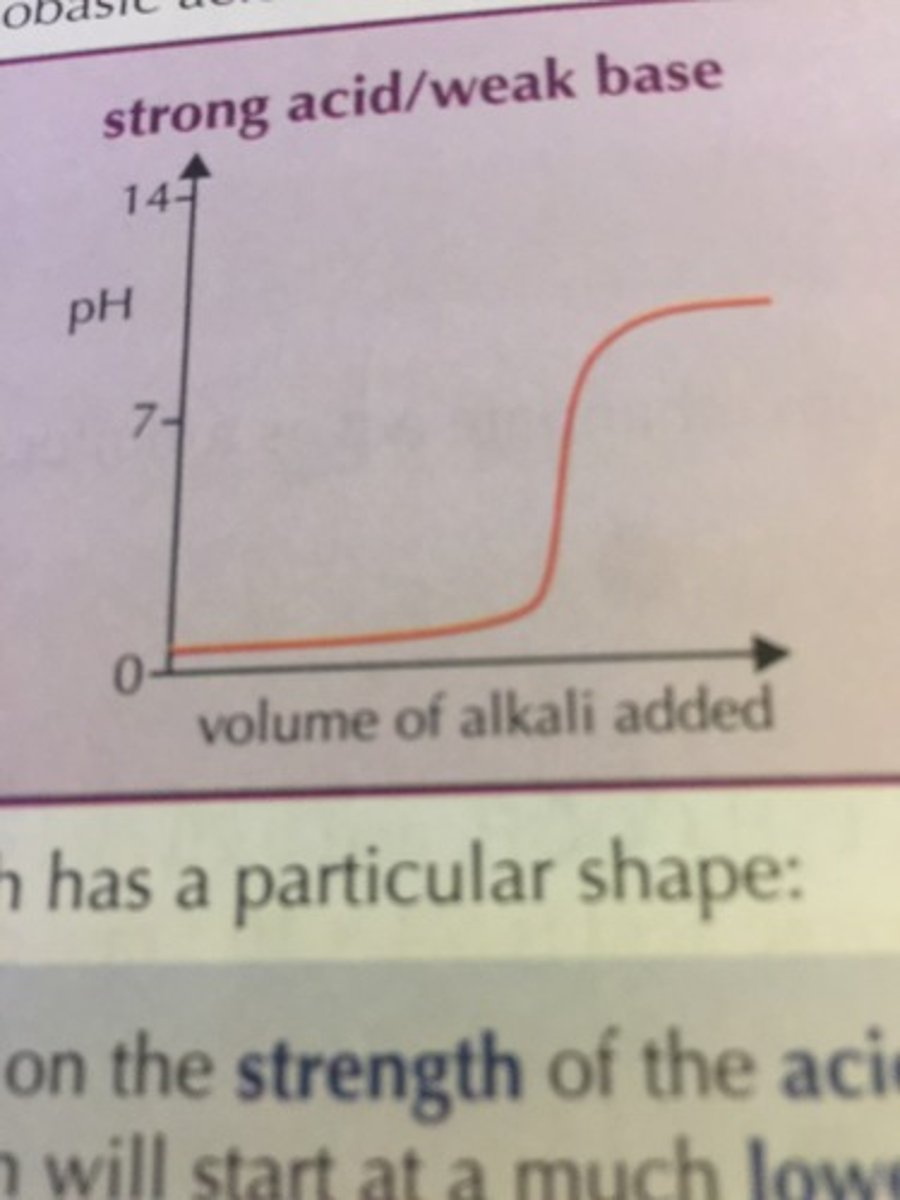
Strong acid-weak base?
Starts very low
Ends not so high
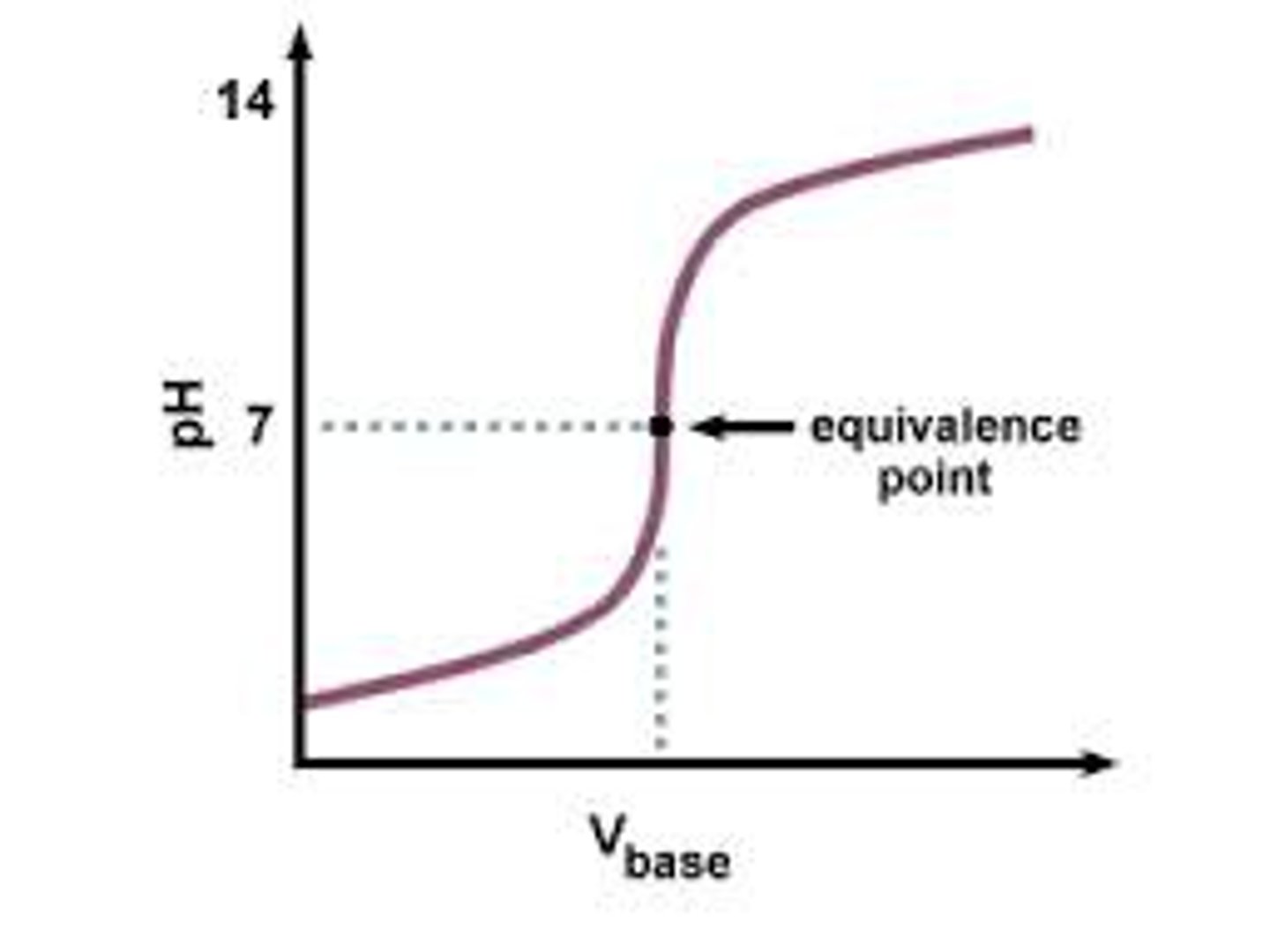
Weak acid-strong base
Starts higher
Ends very high
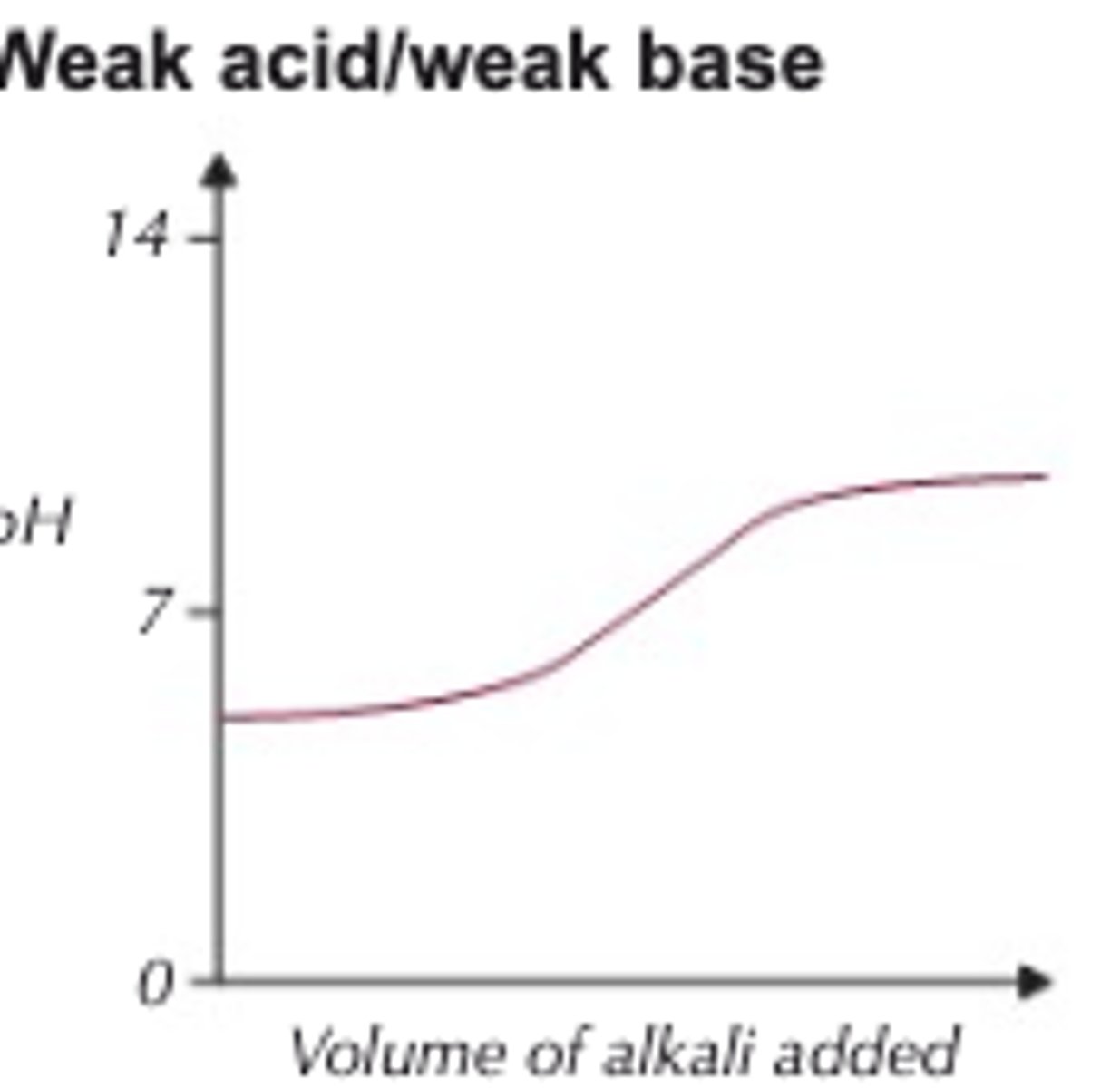
Weak acid-weak base
Starts higher
Ends lower
Why do all of the curves start lower?
Because the bases are added to the acids, not the other way around
For strong acid-strong base, describe the graph.
There is a slow gradual increase in pH as the first 20cm3 is added
Sudden increase from 2-12 pH
Slow gradual increase after
When does the vertical region of the curve occur?
When the number of moles of alkali added equals the number of moles of acid in the original solution
What is this called?
The equivalence point
In weak acid-strong base titration curve, why does the pH level off before the jump?
Because a mixture is formed containing the unreacted acid and the salt formed by neutralisation of the acid, which acts as a buffer.
What can be said for strong acid-weak base?
Graph is the same as strong acid-strong base at beginning, after jump the buffer effect happens and levels off
Why is it harder to study weak acid-weak base using an indicator?
There is no clear vertical region/equivalence point
What can be used instead?
A pH probe
What does this do?
Measures the pH throughout the titration.
What can the pH at the equivalence point be found from?
The midpoint of the vertical region
What will this be for strong acid-strong base?
7
What will happen when a weak acid or weak base is used?
It will shift away from neutral
Why?
At the equivalence point, the solution only includes a salt, and the salts formed from weak acids are basic and those formed from weak alkalis are acidic
What is an indicator?
A substance that changes colour as the pH changes.
What is the equilibrium for indicators?
HInd <=> Ind- + H+
What does most of the indicator exist as in acid?
The neutral form (HInd)
In alkali?
The anion (ind-)
What does indicator do?
Change colour over a range of pHs.
Why can you see a quick colour change in a titration?
Because the pH of the solution changes sharply at the end point
What does the range of the indicator have to lie in?
The vertical section
For a strong acid-strong base the pH increases sharply from 2-12.
Indicators:
A- 8.3-10.0
B- 6.0-7.5
C-4.0-6.5
D-3.2-4.4
Which could be used?
All of them as they all change fully in this range
For weak acid-strong base, pH increases sharply from 5 to 12. Which could be used?
A
B
For strong acid-weak base, pH increases sharply from 2 to 8. Which used?
D
C
B
What about for weak acid-weak base?
There is no significant vertical area showing a significant change in pH so no indicator would therefore work
What is a dilute acid?
An acid that contains a low ratio of acid to volume of solution.
What is a concentrated acid?
An acid that contains a high ratio of acid to volume of solution.
In the equation:
HCl + H2O <=> H3O+ + Cl-
Label conjugate acid-base pairs 1 and 2
HCl + H2O <=> H3O+ + Cl-
Acid-Base pair 1: HCl (acid) Cl- (base)
Acid-base pair 2: H2O (base) H3O+ (acid)