Ethers and Epoxides; Thiols and Sulfides
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
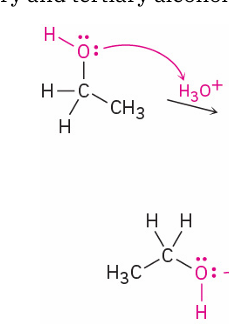
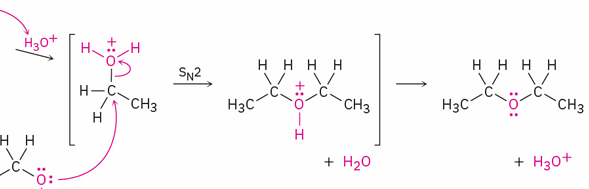
ethanol → ether ( sulfuric acid catalyzed dehudration)
mechanism
reagent
SN2
acid
alcohol → alkoxide
reagent
mechanism
strong base (NaH) and THF
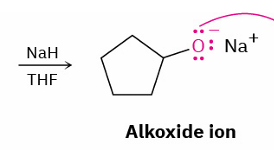
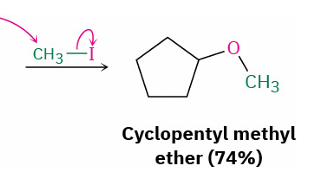
alkoxide → ether
reagent
mechanism
primary alkyl halide eg. CH3-I
SN2
what competive E2 elimination means for making unsymmetrical ethers
more hindered alkoxide rather than more hindered alkyl halide and
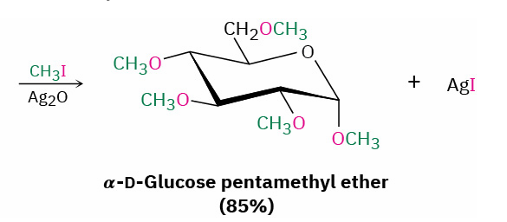
direct williamson ether synthesis
reagent
Ag2O (silver oxide) - milder base
no alkoxide intermediate
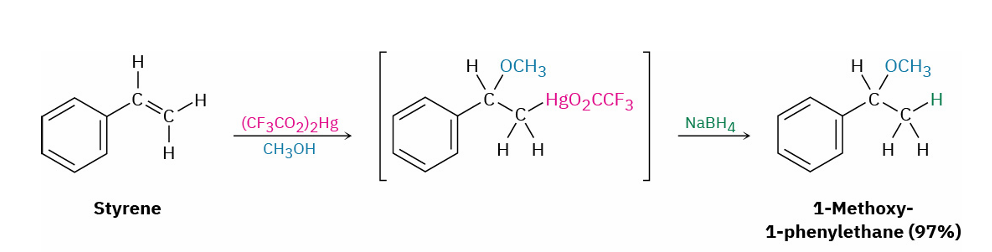
alkene → ether ( alkoxymercuration)
reagents
stereochem
mercuric trifluoroacetate (CF3CO2)2Hg and alkyl alcohol (e.g. CH3OH)
NaBH4
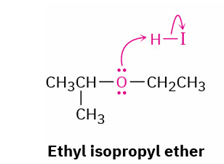
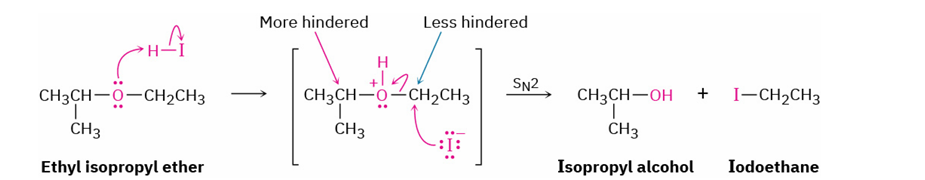
acid cleavage of primary or secondary groups substituted ethers
reagent
product
mechanism
HBr / HI and water
HCl does not cleave ethers
alcohol and alkyl halide
SN2 (reacts with protonated ether at less hindered site)
less hindered becomes halide
More hindered becomes alcohol
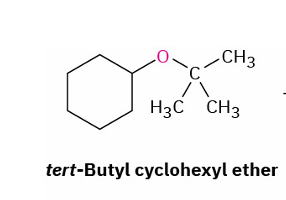
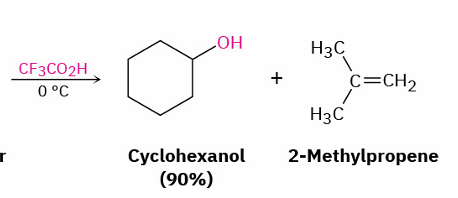
acid cleavage of tertiary/ benzylic/ allylic group substituted ethers
reagents
mechanism
trifluoracetic acid (CF3CO2H) / HBr/ HI
SN1 or E1 mechanism
(OH on less substituent part)
alkene to epoxide
reagents
peroxyacid (RCO3H) mostly m-chloroperoxybenzoic acid
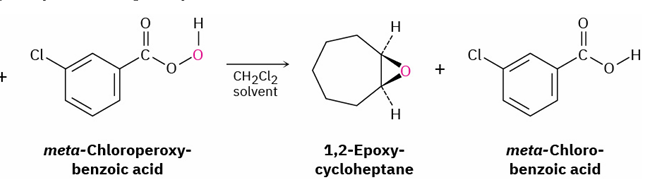
alkene → epoxides
reagents?
Cl2, H2O
NaOH, H2O
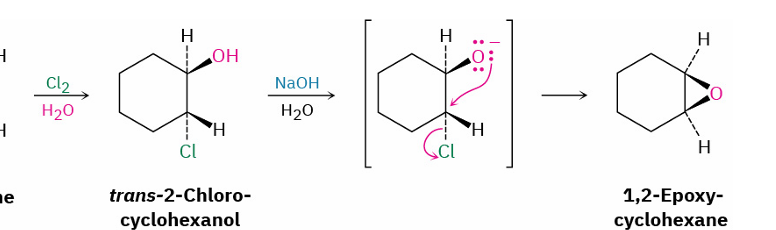
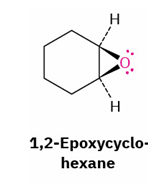
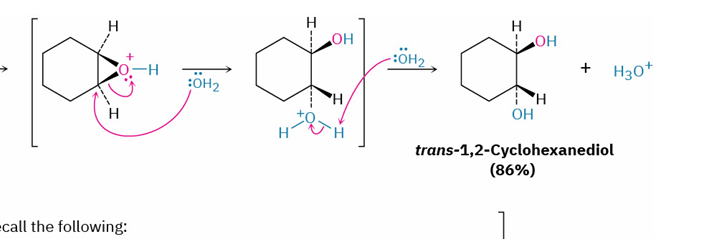
epoxide to trans 1,2 diol/ trans halohydrin by acid
mechanism?
reagent
stereochem
SN2 backside attack
H3O+ or HX
primary or secondary subs → X on less substituted
tertiary subs → X on more substituted
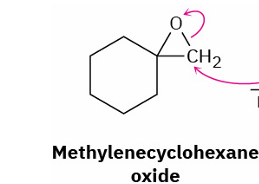
base catalyzed epoxide opening
reagents
base e.g. NaOH and H2O
also be done by RMgX , (RNH2 or R2NH)
adds 2 OH unless with nucleophile
nucleophile attacks less hindered site
OH on more substituted C
crown ethers nomenclature
x - crown - y
x- number of atoms in ring
y- number of O atoms
sulfur iupac
same principles as alcohol
but -ol is replaced with -thiol
SH - group ( as a subs on benzene)
mercapto group
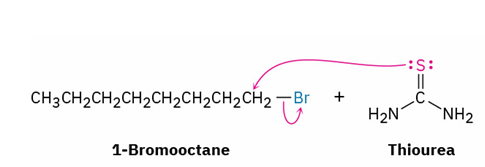
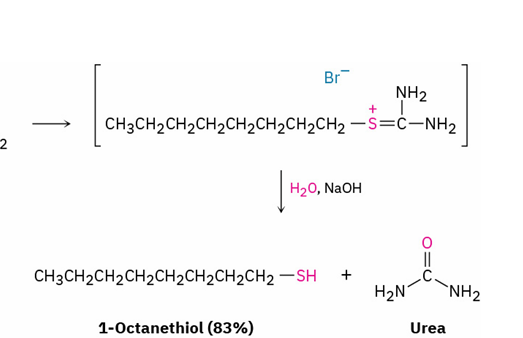
alkyl halide → thiol
reagents
mechanism
sulfur nucleophile like (NH2)2C=S
aqueous base ( NaOH , H2O)
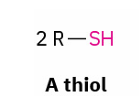
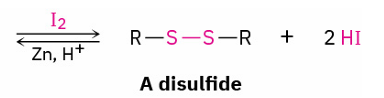
thiols → disulfide and reverse rxn
Br2 or I2
Zinc + acid
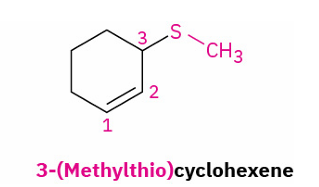
sulfide iupac
same as ether
sulfide replaces ether
alkylthio replaces methoxy
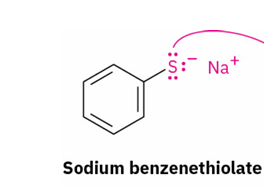
thiol → sulfide ( Williamson ether synthesis)
reagent
primary alkyl halide
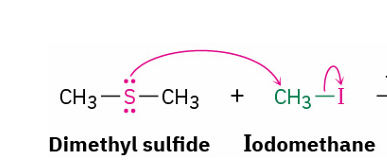
dialkyl sulfides react with primary alkyl halude to give..
by which mechanism
sulfonium ion
SN2
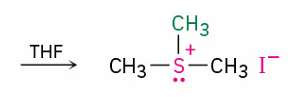
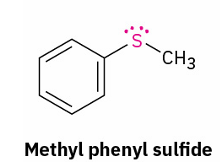
sulfide to sulfoxide
reagent
hydrogen peroxide (H2O2)
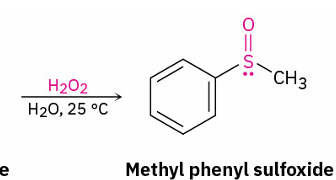
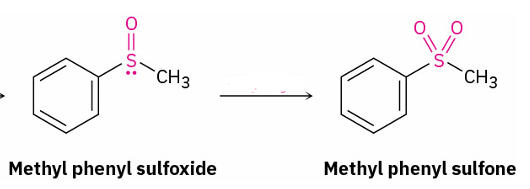
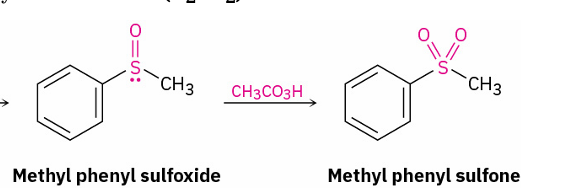
sulfoxide to sulfone
peroxyacid eg. CH3CO3H