electrolysis (summarised)
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
electrolytic cell definition
consists of two electrodes dipped into an electrolyte (one which conducts electricity in molten or aqueous state) and is chemically decomposed when an electric current passes through it
for electrolyte (must be molten or aqueous) but what if solid?
no electricity produced
does not contain mobile ions unlike molten or aqueous state so no reaction occurs
electrolytic cell process/reaction
non spontaneous (needs a battery )
electrical —> chemical energy
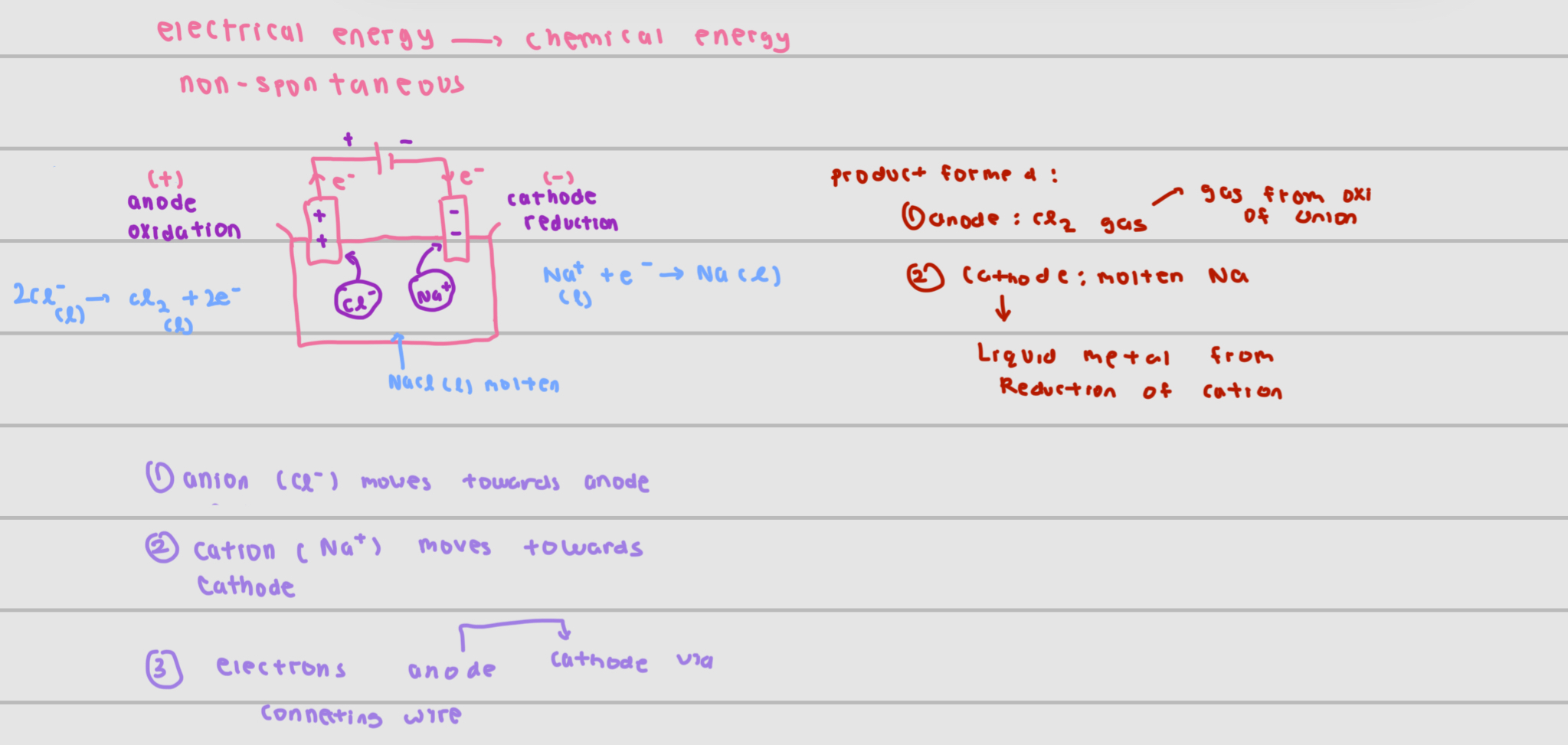
draw + explain set up
positive terminal of battery—> positive ions for that electrode (vice versa)
electrons move from anode to cathode via connecting wire
at anode , oxidation occurs / at cathode ,reduction occurs
anion in electrolyte moves to anode to undergo oxidation
cation in electrolyte moves to cathode to undergo reduction
polarity at anode and cathode for electrolysis
anode —> positive
cathode —> negative
(because of the terminal of the battery it Is connected to)
movement of ions in electrolyte
anion move towards anode
cation moves towards cathode
movement of electrons in the circuit
from anode to cathode through connecting wire ONLY
(because at anode oxidation occurs = lose electrons —> Zn—>Zn2++2e-) and cathode is where reduction occurs =gain electron from the ones anode has released —> Cu2+ + 2e- —>Cu
)
electrolysis of molten salt
presence of only liquid cation and anion in electrolyte
product at anode
gas from oxidation of anion
product at cathode
Liquid Metal from reduction of cation
inert (chemically inactive)electrodes for electrolysis
graphite (high melting point, react with oxygen)
platinum (low melting point , dont react with oxygen)
aluminium is extracted from
bauxite which contains aluminium oxide mixed with iron oxide (Fe2O3)and silica SiO2
electrolysis of molten aluminium oxide will yield aluminium
pure aluminimum oxide characteristics
expensive
dangerous to carry out
high melting point
pure aluminimum oxide is mixed with
cryolite (Na3AlF6), melts at lower temp
carbon anodes
frequently attacked by oxygen gas at high temperature to form gaseous products, mainly carbon dioxide
this causes carbon anodes to burn away so must be replaced regularly
more reactive metal
stronger reducing agent/gets oxidised
voltaic cell
serves like a battery + convert chemical energy to electrical energy
electrical energy produced from spontaneous redox reaction in which electrons are transferred through an external circuit
simple voltaic cell consists of
2 electrodes made of metals with different reactivities
both electrodes immersed in aqueous electrolyte solution
electrons flow form to __ (voltaic cell)
through the external circuit (wire) to the less reactive metal which acts as the positive terminal (cathode)
at the cathode , positive ions in the electrolyte gain electrons and are reduced to form neutral atoms Eg : Cu2++2e- —> Cu
the greater the distance between two elements
greater cell voltage
voltaic cell polarity of electrodes
cathode : postive
another : negative
(because of the flow of electrons)
observations in reactions
at the anode : electrode “dissolves” into solution / becomes smaller/eventually gone/mass decrease (ion discharged into electrolyte )
at the cathode : electrode gain mass / become larger in size (gain ion from electrolyte )
solution: talk about colour change
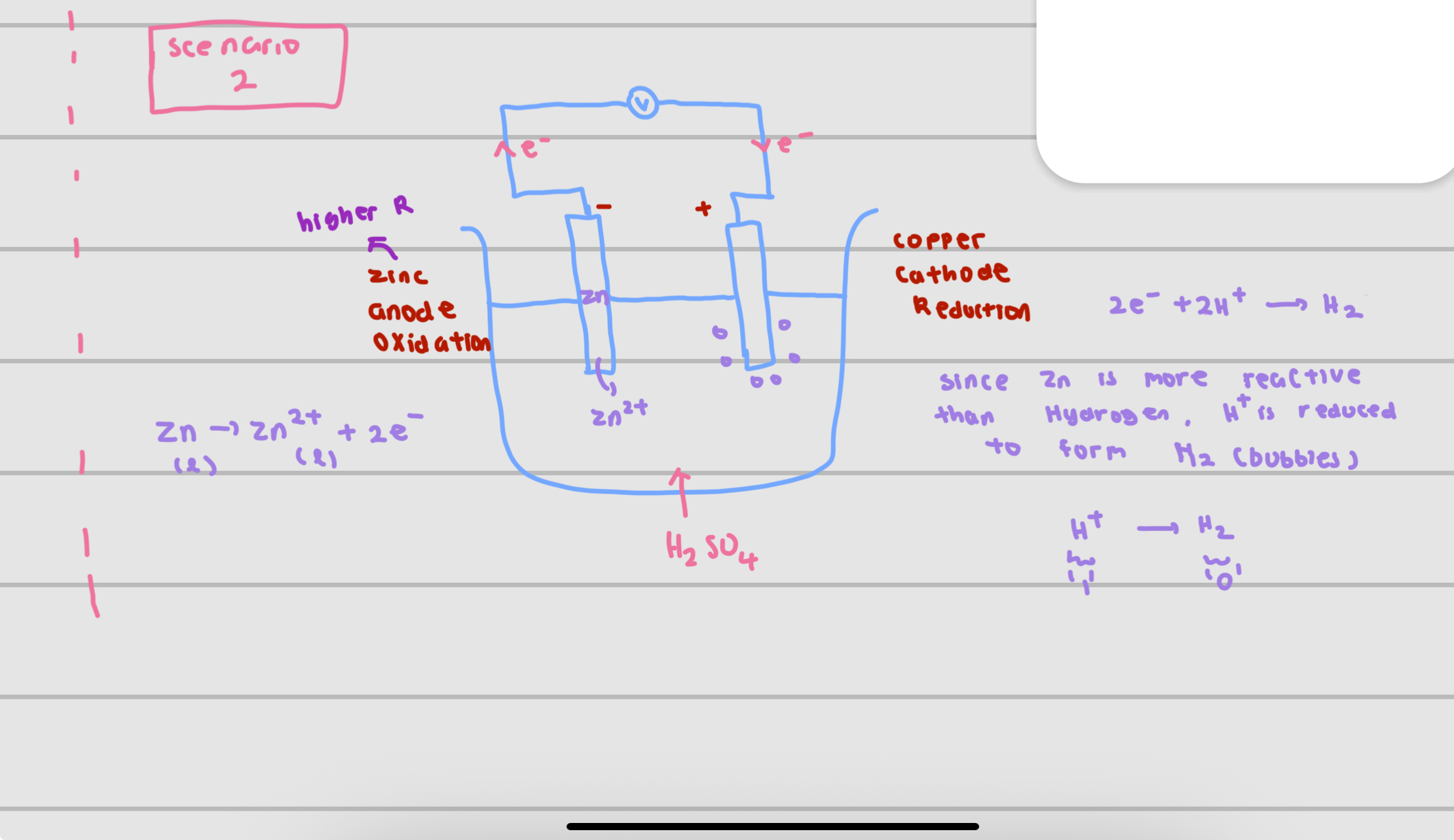
explanation for the diagram/why use hydrogen
at cathode : attract positive cations (hydrogen is positive )
then hydrogen is more reactive than copper , copper will be reduced until used up so left with hydrogen and forming bubbles/efferversence