. ݁+ ⊹ general physics 2 ; "exam 3"
1/53
Earn XP
Description and Tags
last hurrah!!!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
☆ Blackbody Radiation
Blackbody radiation = electromagnetic radiation emitted by hot objects.
Blackbody
Ideal object that absorbs all radiation falling on it.
Also a perfect emitter.
Its spectrum depends only on its temperature.
Color vs Temperature
At about 1000 K: objects glow red.
Lower frequency, longer wavelength.
Above about 2000 K: objects glow yellow/white.
Higher frequency, shorter wavelength.
Big idea: as temperature increases, the frequency of emitted radiation increases.
☆ Planck Hypothesis & Photons
Planck’s hypothesis
Energy in electromagnetic radiation is not continuous.
It comes in discrete chunks called quanta.
Energy quantum / photon
Energy of radiation:
E = nhf
n = integer number of quanta
h = Planck’s constant
f = frequency
Single photon energy:
E = hf = hc/λ
Photon
A photon is a packet of electromagnetic energy.
It is not a classical particle with rest mass.
Higher frequency = higher photon energy.
☆ Photoelectric Effect — Basic Idea
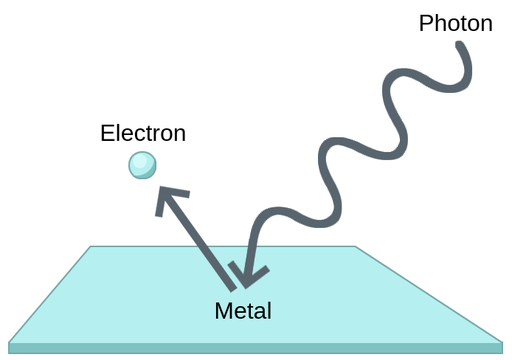
Photoelectric effect
When light shines on a metal surface, electrons are emitted from the surface.
Why it matters
It gives strong evidence that light energy is carried in photons.
Electrons are only emitted if each photon has enough energy to overcome the metal’s work function.
☆ Photoelectric Effect — Experimental Setup
Apparatus
Two metal plates/electrodes in a vacuum.
A galvanometer measures current.
A battery creates a potential difference.
Basic process
Light shines on the emitting plate.
Electrons are ejected.
Electrons travel to the opposite plate.
A current is detected.
☆ Ionization Energy
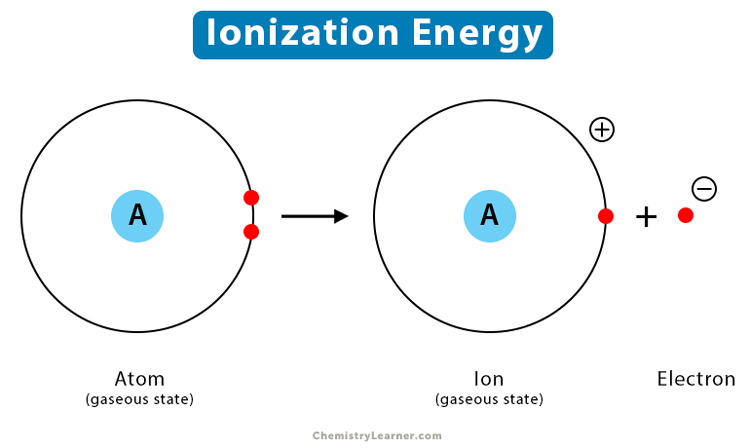
Ionization energy = energy needed to remove an electron completely from the atom.
Removing an electron means taking it to n = infinity.
At n = infinity, energy is 0 eV.
Ionization energy from level n:
Eion = 0 − En
☆ Hydrogen-Like Ions
Hydrogen-like ions have only one electron.
Examples:
H has Z = 1
He+ has Z = 2
Li2+ has Z = 3
Be3+ has Z = 4
They use the Bohr formulas with Z² included.
Higher Z means stronger attraction and larger energy gaps.
☆ Energy Transitions
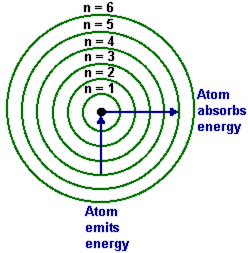
An electron moving from high n to low n emits a photon.
An electron moving from low n to high n absorbs energy.
Photon energy equals the energy difference:
Ephoton = Ei − Ef for emission
Ephoton = Ef − Ei for absorption
Since photon energy is positive, use the magnitude of the energy difference.
☆ Emission vs Absorption
Emission
Electron drops to a lower energy level.
Photon is released.
Absorption
Electron jumps to a higher energy level.
Photon energy is absorbed.
The photon energy must exactly match the energy gap.
☆ Rydberg Formula
The Rydberg formula finds wavelengths of spectral lines.
Formula for hydrogen-like atoms:
1/λ = RZ²(1/nf² − 1/ni²)
ni = initial level.
nf = final level.
R = 1.097 × 10⁷ m⁻¹
For emission, ni > nf.
For absorption, ni < nf, but use the energy gap.
☆ Spectral Series
A spectral series is a group of transitions ending at the same final level.
Series names for hydrogen:
Lyman series: transitions ending at n = 1.
Balmer series: transitions ending at n = 2.
Paschen series: transitions ending at n = 3.
The final level determines the series.
☆ Lyman Series
Lyman series = transitions ending at n = 1.
These photons are high energy.
They are usually in the ultraviolet region.
Formula setup:
nf = 1
☆ Balmer Series
Balmer series = transitions ending at n = 2.
Some Balmer lines are visible.
Formula setup:
nf = 2
The shortest wavelength in the Balmer series happens when:
ni = infinity
☆ Paschen Series
Paschen series = transitions ending at n = 3.
These photons have lower energy than Lyman and Balmer transitions.
They are usually infrared.
Formula setup:
nf = 3
☆ Shortest Wavelength in a Series
Shortest wavelength means highest energy photon.
Highest energy transition in a series comes from:
ni = infinity
Use Rydberg formula with:
1/ni² = 0
Higher energy = shorter wavelength.
☆ Longest Wavelength in a Set of Transitions
Longest wavelength means lowest energy photon.
Lowest energy photon comes from the smallest energy gap.
For an electron starting at n = 4, the smallest drop is usually:
4 → 3
Smaller energy gap = lower frequency = longer wavelength.
☆ Maximum Number of Photons from Level n
If an electron starts at level n, the maximum possible number of different photon energies is:
n(n − 1)/2
Example: from n = 4:
4(3)/2 = 6 possible photons
This counts all possible downward transitions.
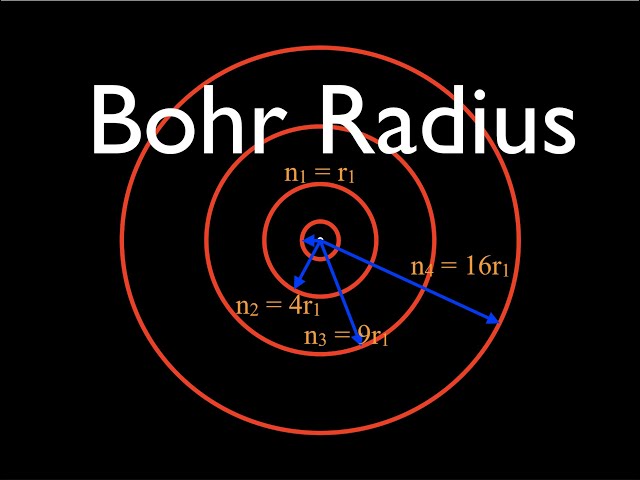
☆ Bohr Radius
Allowed orbit radius for hydrogen-like atoms:
rn = a₀(n²/Z)
a₀ = 5.29 × 10⁻¹¹ m
For hydrogen, Z = 1, so:
rn = a₀n²
Radius increases with n².
Higher energy levels are farther from the nucleus.
☆ Electron Speed in Bohr Orbit
Electron speed can be found using angular momentum quantization:
mvr = nh/(2π)
Solve for speed:
v = nh/(2πmr)
Higher Z generally means stronger attraction and faster electron speeds for the same n.
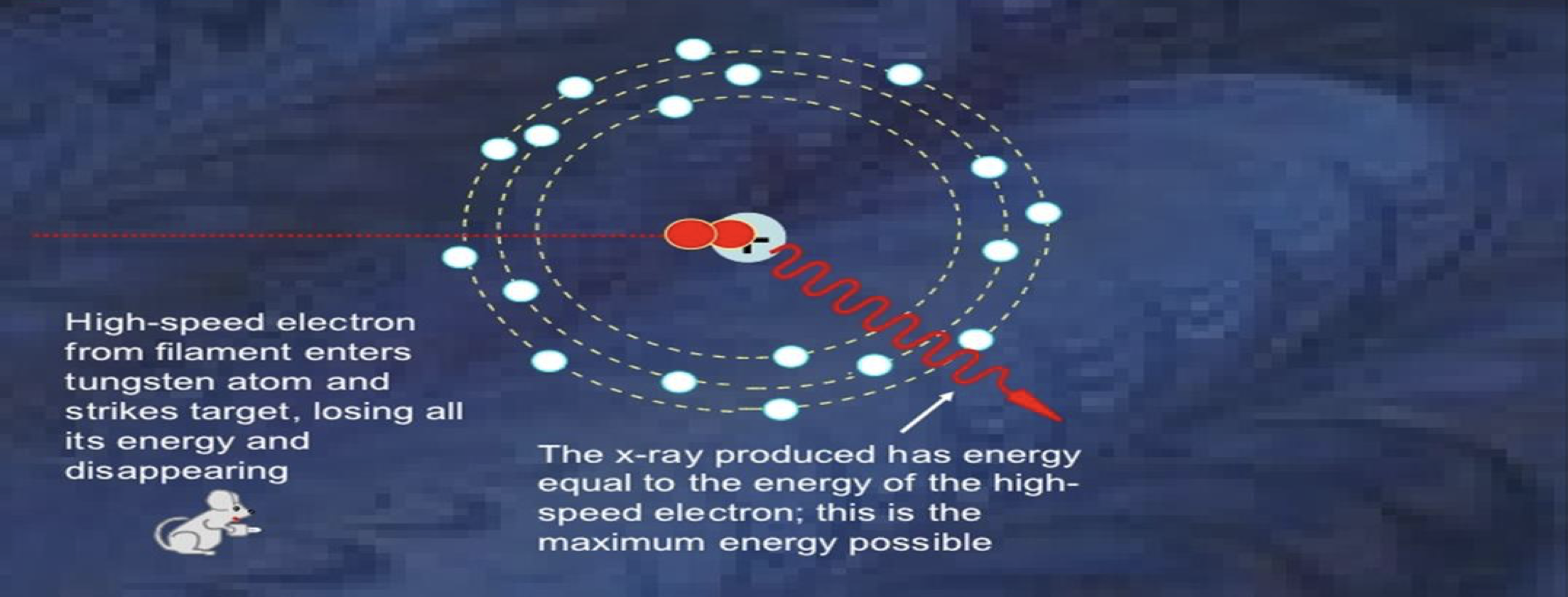
☆ X-Ray Production
X-rays can be produced when fast electrons hit a metal target and slow down.
Lost kinetic energy becomes photon energy.
If all kinetic energy becomes one photon:
qV = hc/λ
Minimum accelerating voltage:
V = hc/(qλ)
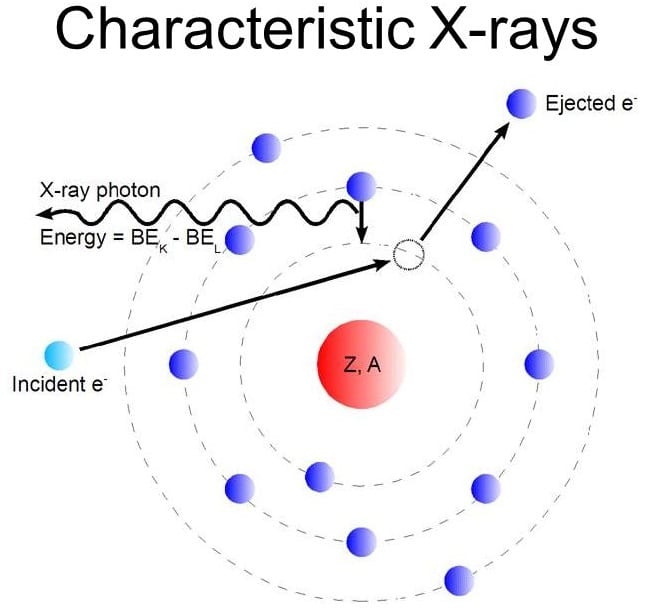
☆ Characteristic X-Rays
Characteristic X-rays happen when inner-shell electrons are removed and outer electrons fall into the vacancy.
The emitted photon has energy equal to the energy difference between atomic levels.
Each element produces characteristic X-ray wavelengths.
☆ X-Ray Tube Voltage
An electron accelerated through voltage V gains kinetic energy:
KE = qV
If this energy becomes an X-ray photon:
qV = hc/λ
Higher voltage produces higher energy X-rays and shorter wavelengths.
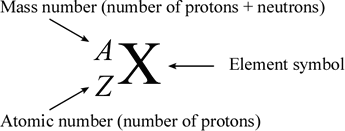
☆ Nucleus Basics
The nucleus contains protons and neutrons.
Protons have positive charge.
Neutrons have no charge.
The nucleus is tiny, dense, and contains most of the atom’s mass.
Electrons occupy the space outside the nucleus.
☆ Atomic Number, Z
Atomic number, Z = number of protons.
Determines the element.
Example:
Carbon has Z = 6.
Uranium has Z = 92.
Changing Z changes the element.
☆ Mass Number, A
Mass number, A = protons + neutrons.
Formula:
A = Z + N
N = number of neutrons.
To find neutrons:
N = A − Z

☆ Isotopes
Isotopes are atoms of the same element with different numbers of neutrons.
Same Z, different A.
Example:
Carbon-12 and carbon-14 both have 6 protons.
Carbon-14 has more neutrons.
☆ Nuclide Notation
Nuclear notation shows mass number and atomic number:
A over Z X
X = element symbol.
A = mass number.
Z = atomic number.
Example: carbon-14 has:
A = 14
Z = 6
☆ Strong Nuclear Force
The strong nuclear force holds protons and neutrons together in the nucleus.
It is very strong but acts only over very short distances.
It overcomes repulsion between protons.
Without it, the nucleus would fly apart.
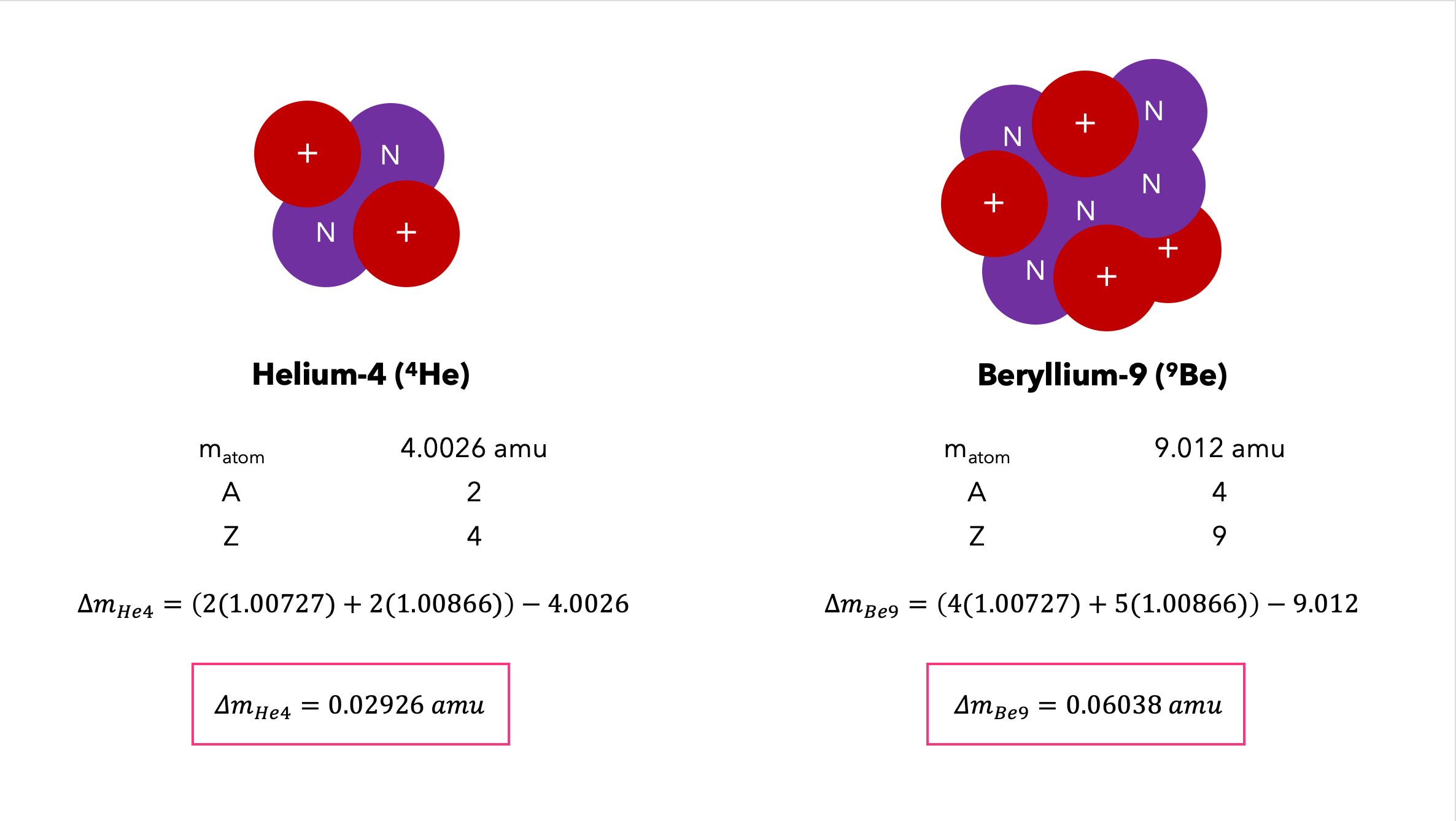
☆ Mass Defect
Mass defect = missing mass when nucleons bind together.
A nucleus has less mass than the total mass of its separated protons and neutrons.
Formula:
Δm = mass of separated nucleons − mass of nucleus
Missing mass becomes binding energy.
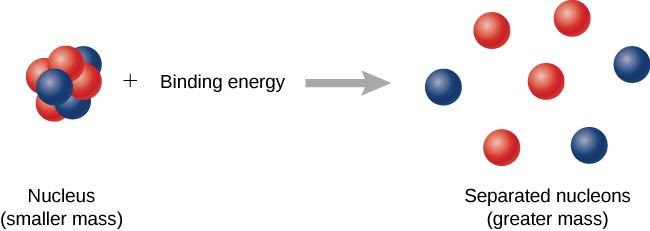
☆ Binding Energy
Binding energy = energy needed to separate a nucleus into individual protons and neutrons.
It is also the energy released when the nucleus forms.
Formula:
BE = Δmc²
Using atomic mass units:
BE = Δm(931.5 MeV/u)
☆ Binding Energy per Nucleon
Binding energy per nucleon measures nuclear stability.
Formula:
BE per nucleon = total binding energy / A
Higher binding energy per nucleon usually means a more stable nucleus.
Iron/nickel region has very high stability.
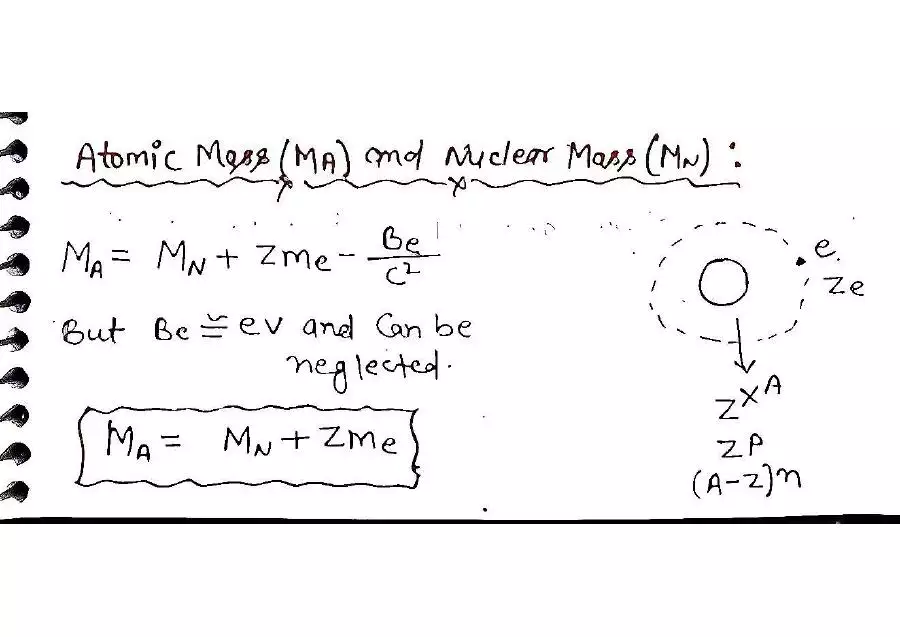
☆ Atomic Mass vs Nuclear Mass
Atomic mass includes electrons.
Nuclear mass does not include electrons.
To find nuclear mass:
nuclear mass = atomic mass − electron masses
Electron mass in u:
me = 0.000548 u
☆ Using Atomic Masses in Nuclear Reactions
Atomic masses often work directly if electrons cancel on both sides.
For beta-minus decay, atomic electron masses usually cancel.
For beta-plus decay, be careful because positron/electron counting matters.
If unsure, use nuclear masses.
☆ Radioactivity
Radioactivity = spontaneous decay of unstable nuclei.
Unstable nuclei emit particles or energy to become more stable.
Decay is random for individual nuclei but predictable statistically for large samples.

☆ Alpha Decay
Alpha decay emits an alpha particle.
Alpha particle = helium nucleus:
4 over 2 He
Changes:
A decreases by 4
Z decreases by 2
General form:
Parent → daughter + alpha particle
☆ Beta-Minus Decay
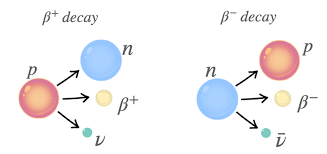
Beta-minus decay emits an electron.
A neutron turns into a proton.
Changes:
A stays the same
Z increases by 1
General form:
neutron → proton + electron + antineutrino
In nuclear notation:
Parent → daughter + beta-minus particle
☆ Beta-Plus Decay
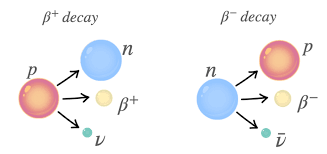
Beta-plus decay emits a positron.
A proton turns into a neutron.
Changes:
A stays the same
Z decreases by 1
General form:
proton → neutron + positron + neutrino
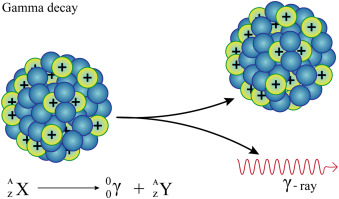
☆ Gamma Decay
Gamma decay emits high-energy electromagnetic radiation.
It changes the energy state of the nucleus.
Changes:
A does not change
Z does not change
Gamma rays often occur after alpha or beta decay.
☆ Balancing Nuclear Equations
Nuclear equations must conserve:
Mass number, A
Atomic number, Z
Charge
Add A values on both sides.
Add Z values on both sides.
Both totals must match.
☆ Half-Life
Half-life = time required for half of the radioactive nuclei to decay.
After 1 half-life: 1/2 remains.
After 2 half-lives: 1/4 remains.
After 3 half-lives: 1/8 remains.
Half-life is constant for a given isotope.
☆ Decay Constant
Decay constant, λ measures probability of decay per unit time.
Formula:
λ = 0.693/t1/2
Larger λ means faster decay.
Shorter half-life means larger λ.
☆ Radioactive Decay Law
Number of nuclei remaining:
N = N₀e^(−λt)
Activity also decays the same way:
A = A₀e^(−λt)
If using half-lives:
N = N₀(1/2)^(t/t1/2)
A = A₀(1/2)^(t/t1/2)
☆ Activity
Activity = number of decays per second.
Formula:
A = λN
SI unit:
Becquerel, Bq
1 Bq = 1 decay/s
☆ Curie
Curie, Ci is another unit of activity.
Conversion:
1 Ci = 3.70 × 10¹⁰ Bq
To convert Bq to Ci:
Ci = Bq / (3.70 × 10¹⁰)
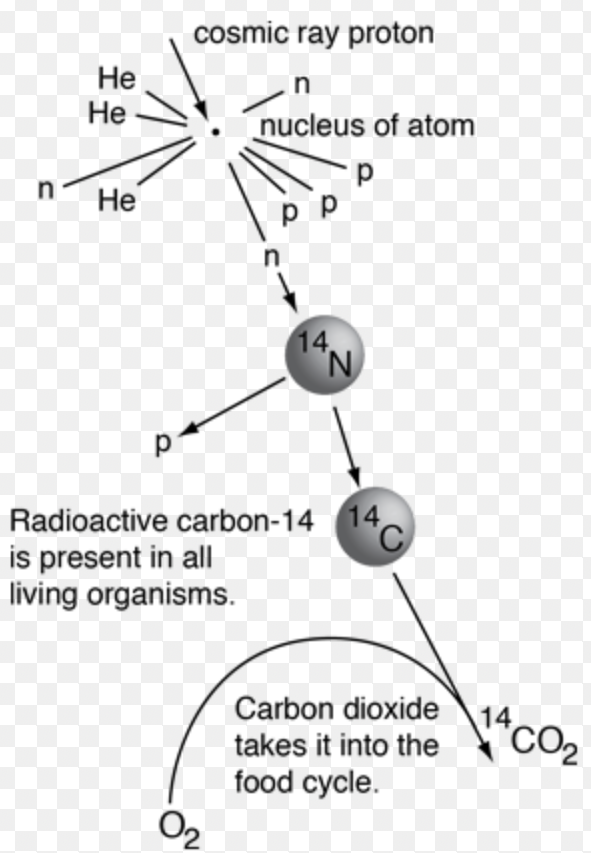
☆ Carbon Dating
Carbon dating uses carbon-14 decay to estimate the age of once-living material.
Living organisms maintain a roughly constant carbon-14 ratio.
After death, carbon-14 decays and is not replaced.
Use activity decay:
A = A₀e^(−λt)
Solve age:
t = −ln(A/A₀)/λ
☆ Number of Decays Between Two Times
Activity changes with time:
A = A₀e^(−λt)
Number of decays between t1 and t2 can be found from:
ΔN = (A1 − A2)/λ
This works because A = λN.
☆ Energy Released in Nuclear Reactions
Nuclear reaction energy comes from mass difference.
Formula:
Δm = mass reactants − mass products
Q = Δm(931.5 MeV/u)
If Δm > 0, energy is released.
If Δm < 0, energy must be supplied.
☆ Q-Value
Q-value = energy released or absorbed in a nuclear reaction.
Formula:
Q = (mass reactants − mass products)c²
Positive Q means exothermic/released energy.
Negative Q means endothermic/energy required.
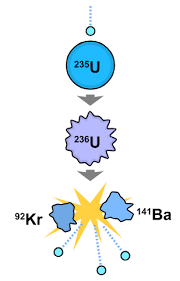
☆ Fission
Fission = splitting a heavy nucleus into smaller nuclei.
Usually releases energy because products have greater binding energy per nucleon.
Often also releases neutrons.
Neutrons can cause a chain reaction.
☆ Chain Reaction
A chain reaction happens when neutrons from one fission event cause more fission events.
If controlled, it can power a nuclear reactor.
If uncontrolled, it can lead to explosive release of energy.
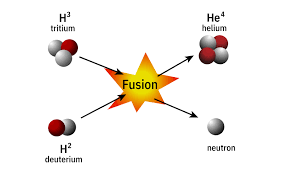
☆ Fusion
Fusion = combining light nuclei to form a heavier nucleus.
Fusion releases energy for light elements because products are more tightly bound.
Fusion powers stars.
Requires very high temperature/pressure because nuclei repel each other electrically.
☆ Alpha Particle Energy
In alpha decay, released energy becomes kinetic energy of products.
If recoil is ignored, alpha particle gets approximately all released energy.
Use:
Q = KE = 1/2mv²
Solve speed:
v = √(2KE/m)
☆ Beta Decay Energy with Atomic Masses
For beta-minus decay, using atomic masses:
Q = [Mparent − Mdaughter](931.5 MeV/u)
Electron masses cancel automatically.
For beta-plus decay, electron/positron mass issues must be handled carefully.
Safer method: use nuclear masses if the problem gives electron mass.
☆ Nuclear Stability
Stability depends on the neutron-to-proton ratio.
Light nuclei are stable near N = Z.
Heavy nuclei need more neutrons than protons.
Very heavy nuclei are often unstable and radioactive.
☆ Why Heavy Nuclei Decay
Heavy nuclei have many protons.
Proton-proton repulsion is large.
Extra neutrons help, but beyond a point the nucleus becomes unstable.
Decay helps move the nucleus toward a more stable