chemistry - elements, compounds and molecules
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
solubility
what is meant by the term solute
the substance being dissolved in the solvent
what is meant by the term solvent
the liquid the solute is dissolving in
what is meant by the term solution
the substance produced when the solute is dissolved in the solvent
what is meant by the term saturated solution
when the maximum amount of solute is dissolved in a certain amount of solvent at a certain temperature
what does solubility in the units per 100g of solvent mean?
the measurement of the maximum amount of solute that can dissolve in exactly 100g of solvent at a certain temperature
how to plot and interpret solubility curves
plot:
measure the solubility (maximum amount) of solute that can dissolve in the solvent at a certain temperature
do this multiple times at different temperatures, and then plot and draw the curve
interpret:
if the curve is going up as the temperature increases, the solute was a solid
if the curve is going down as the temperature increases, the solute was a gas
explain the dilution of coloured solutions and the diffusion of gases
practical: investigate the solubility of a solid in water at a certain temperature
method:
set the water bath to a certain temperature, measure a certain amount and add the solid
continue to add more solid until it stops dissolving and remains solid at the bottom of the beaker (ensures it is saturated)
weigh the evaporating basin and then pour a portion of the solution (with no remaining solid) in it
weigh the evaporating bassin and the solid together to get the total mass
use a bunsen burner to evaporate the solvent until only crystals of the solute are left
weigh the evaporating basin + solute crystals
use these masses to calculate solubility
repeat the experiment at different temperatures
what should happen:
as the temperature increases, the time taken for the solid to dissolve should decrease
this is because as the particles re heated up, they gain kinetic energy, which reduces the sxtrength of the intermolecular forces and causes the particles to break apart, making them more soluble
how to classify substances as an element, compound and mixture
element - substance made of one type of atom
compound - a substance where 2 or more different atoms chemically bonded together
mixture - a substance where 2 or more different atoms NOT chemically bonded together
melting/boiling points of pure substances vs mixtures
a pure substance will have a fixed melting and boiling point
a mixture will have a range for their melting/boiling point
separation of mixtures
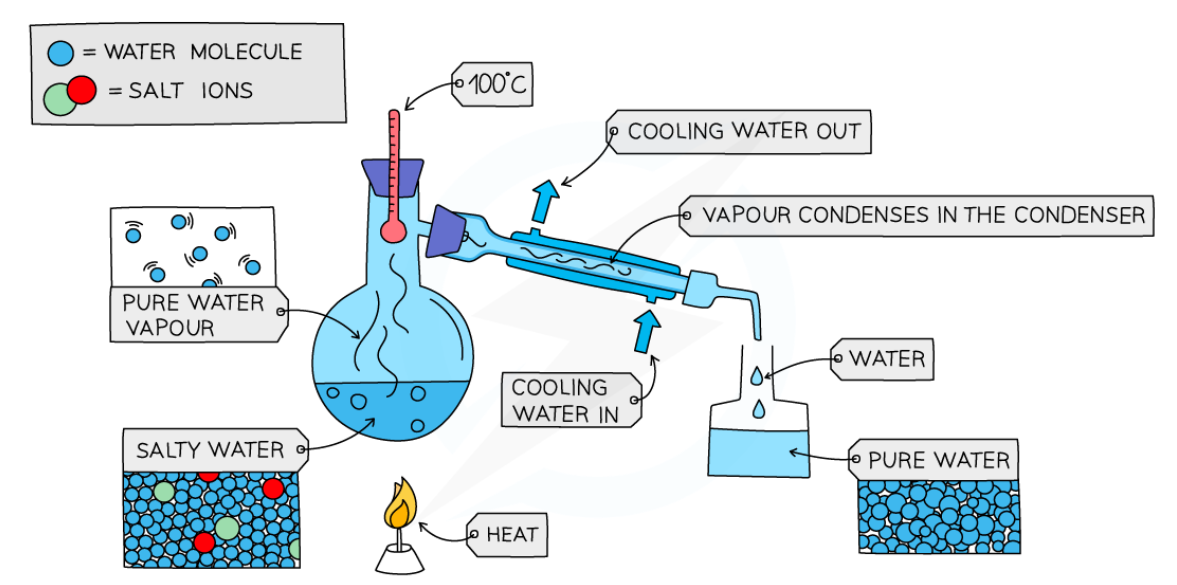
simple distillation
a mixture of a soluble solid and a liquid
as the substances have different boiling points, the solution is heated until one of the substances reaches their boiling point and evaporates
the evaporated liquid rises, adn the gas is eventually condensed in the condenser back into a liquid, and pours into a separate flask
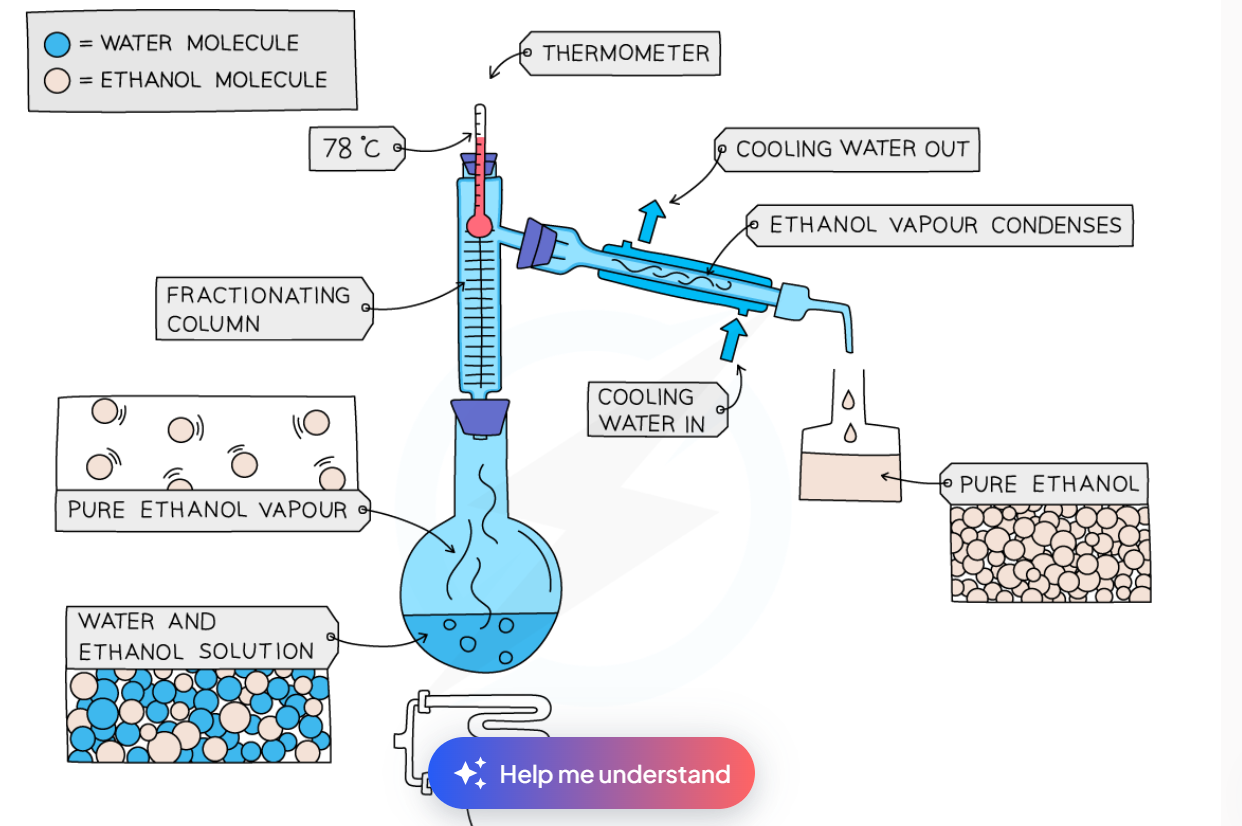
fractional distillation
used to separate 2 or more miscible liquids
the solution is heated and the substance with the lowest boiling point will evaporate and rise
it will travel through a fractionating column, whcih will cause the liquid to heat and condense many times, ensuring the purest separation
the gas will pass through a condenser, and pour into another flask
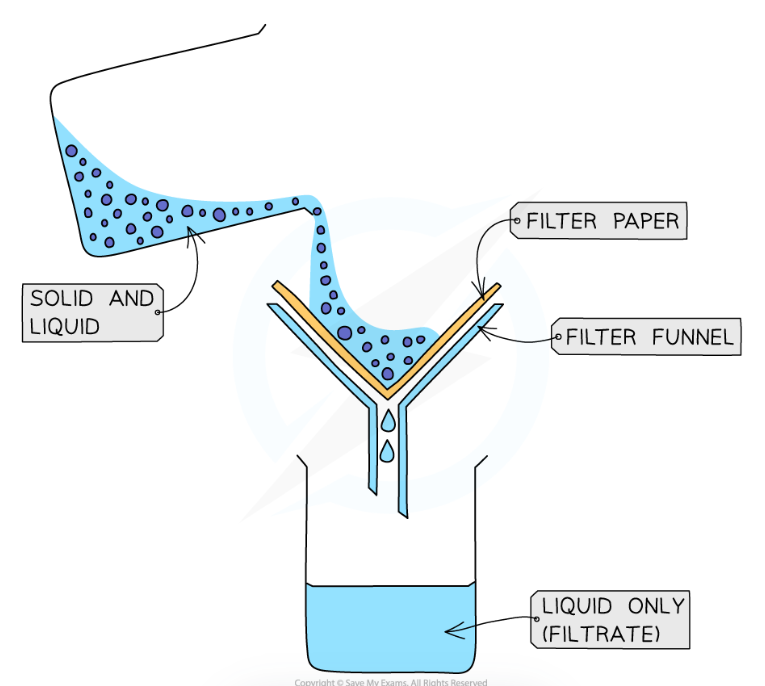
filtration
used to separate an insoluble solid from a solution
a piece of filter paper is placed in a funnel, and the solution is poured into the funnel
the liquid will travel through the filter paper into a beaker, while the undissolved solid will be caught in the paper as residue
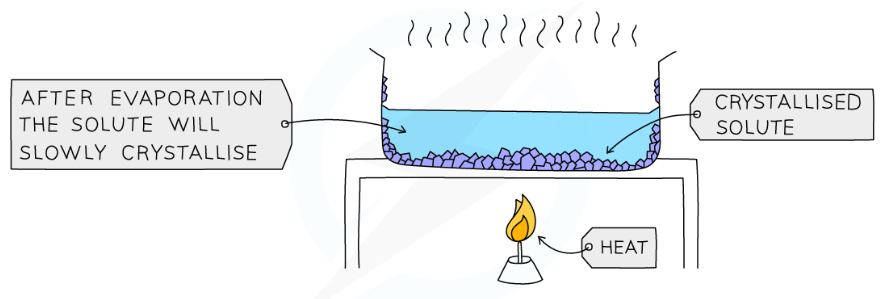
crystallisation
used to separate a soluble solid from a solution
the solution is heated, allowing the liquid to evaporate so that you are left with a saturated solution
the saturated solution will be left to cool, so that crystals willf orm due to decreasing solubility
the mixture will be filtered and you will be left with the solid
paper chromatography
used to separate substances with different solubilities in a certain solvent
draw a pencil line about 1cm from the edge of the filter paper, and put sample sof the different dyes on it
fill a beaker with water and place the paper in it, ensuring that the water level is below the level of the solvent
the solvent travels up the paper, taking the different dyes with it (in different amounts based on their solubility in the solvent), allowing you to see the different dyes
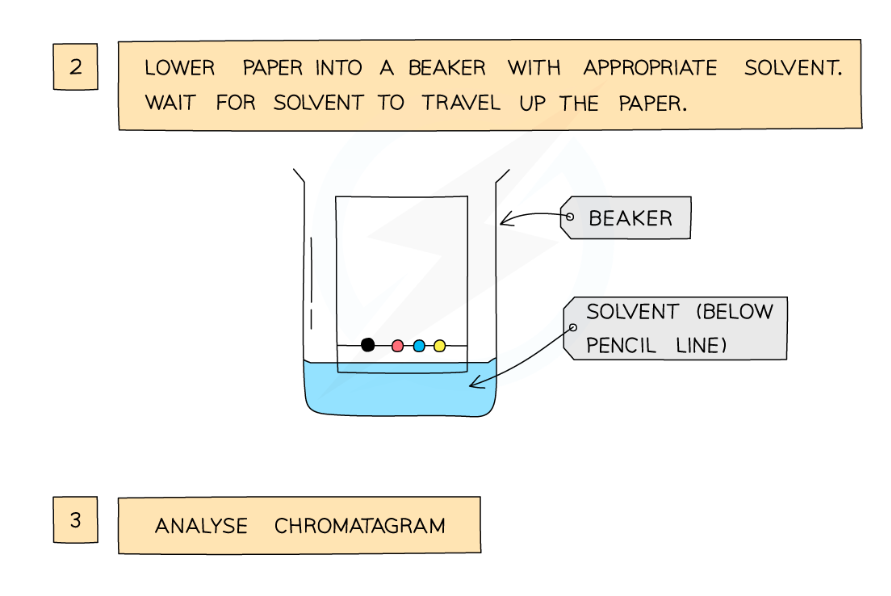
how does a chromatogram provide information about the composition of a mixture
because the different dyes have different solubilities in water, they will travel at different rates, therefore travellign different distances up the paper, identifying the different substances
caluclating the Rf value
distance travelled by dye/ distance travelled by solvent