CH26 Cancer Cell Biology
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
how are cancers named
based on cell type that became cancerous
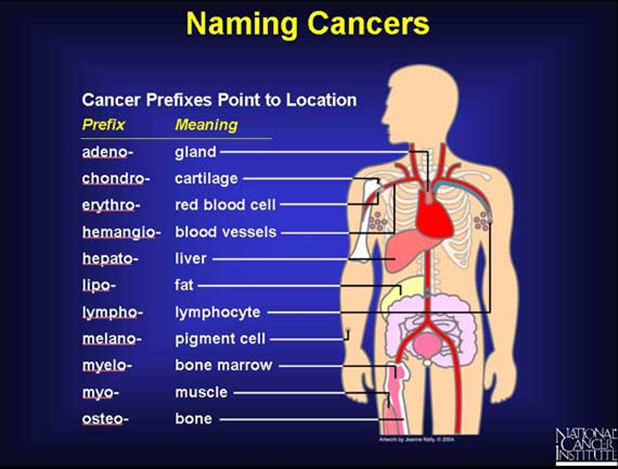
carcinomas
90% from epithelial cells
sarcomas
come from supporting tissues
leukemias
from blood or lymphatic cells
common causes of cancer mutations
base substitution or chromosome abberation
usually not inherited
2 phenotypes of cancer
loss of ability to regulate growth - proliferation
no longer restricted to certain locations - metastasis
benign vs malignant tumor
benign: localized, only proliferation control lost (growth)
malignant: proliferation and metastasis control lost (cancer)
conditions met for normal cell growth
contact with ECM through integrins
no contact with other cells
anchorage independent growth
cancerous cells do not require contact with the ECM
density independent growth
cancerous cells are not inhibited by contact with other cells
mechanisms where proliferation control is lost
DNA damage repair
Cell cycle regulation
chromosomal telomere integrity
apoptosis pathway
fundamental defect leading to cancer
inability to maintain a cell’s DNA
does not cause cancer itself but increases likelihood
mutator phenotype
cells with a high level of genomic instability
example of myeloid leukemia
cancer type arises from a specific DNA alteration
chromosome translocation in this case
types of DNA modification that can cause deleterious changes
not nucleotide sequence alteration
methylation
modification of histone proteins
speed of cell cycle throughout life
embryo: rapid
maturity: cells in G0
neurons remain in G0, RBCs constantly divide
2 commonly disrupted signaling pathways
pathway that links growth hormone receptors to cell cycle
inhibitory pathways that restrict proliferation
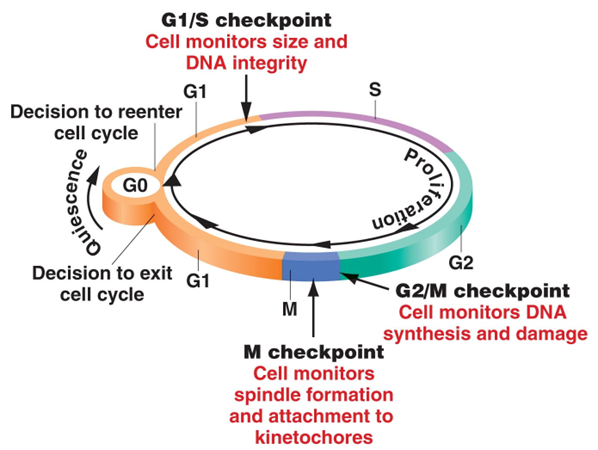
checkpoints of cell cycle are where
G1/S
G2/M
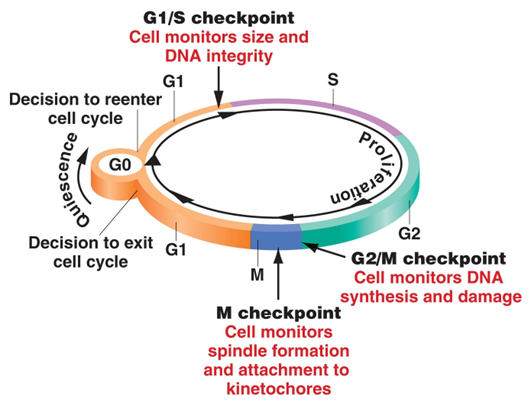
main cell cycle regulation mechanisms
series of kinase
cyclin proteins → interact with cyclin dependent kinases
vascularization of tumors
body tumors are extensively vascularized
tumors can activate blood vessel formation
outside of the body, tumor cells only form small tumors due to lack of diffusion
VEGF and FGF
angiogenesis activating molecules
activate local blood vessel growth
important to tumors
inhibitors of angiotensin
context of tumor growth
angiostatin, thrombospondin, endostatin
tumor has to inactivate inhibitors or overproduce growth factors
most common cancers
breast, lung, prostate, colorectal
fist mentions of cancer in history
Egypt 3000 years ago
Hippocrates
Oncos 180 AD
most effective mechanism to remove skin cancer
surgery
radiation if metastasized
photodynamic therapy, biological therapy (immune system)
most common cancers present at birth
most commonly in gonadal refion
not common
first step of metastasis
invasion: leaving original location to surrounding tissues
how is motility for invasion possible
microfilament based amoeboid movement
must be activated
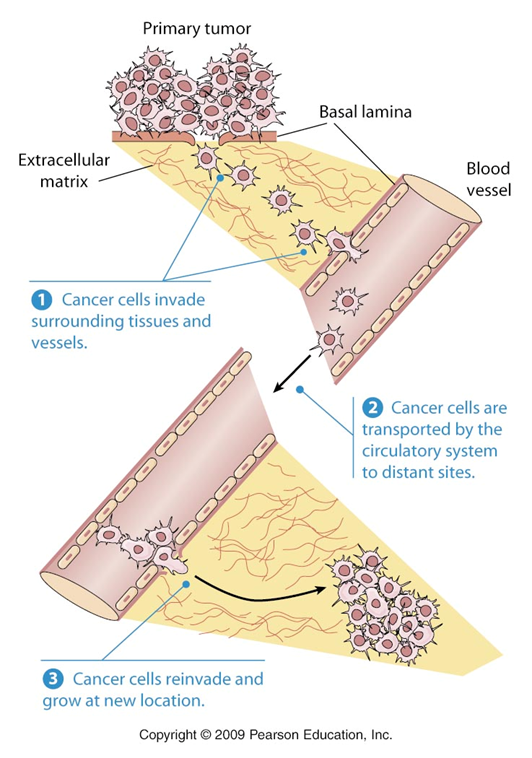
what do cancer cells do after invasion
penetrate wall of blood vessel and move into new tissue; true metastasis
how do cadherins play into metastasis
cadherins normally bind cells to prevent migration
in metastasis they are reduced
what do cancer cells produce to migrate through basal lamina
proteases
steps of cancer cell spread
1) invasion: leave OG tissue to the surroundings
2) travel blood stream
3) metastasis: invade new tissue
4) move through ECM
plasaminogen (plasmin)
common protease in the ECM which can also activate other proteases to amplify effect
effects of proteases on blood vessel walls
loosen connections between blood endothelial cells to allow access through circulatory and lympahtic systems
where do cancer cells translocate to through blood stream
most die
system they arrive at first
most supportive tissue for growth
immune surveillance theory about cancer
debate
most cancers are destroyed by the immune system
debated about if immune system can recognize cancer cells
effect of immunosurpressed transplant and HIV in regards to cancer rate
IT patients: higher cancer rate
HIV: same cancer rate
amount of genes involved in cancer
average is 90 with 10 contributing to cancerous phenotype
two general types of gene mutation shown in cancer analysis
mutation in genes that promote cell growth and development (active in embryogenesis but not adults)
mutation in proto-oncogenes which drive proliferation when activated
common protooncogenes
involved in growth signaling pathways
growth factors and their receptors, G-protein signaling components, pathway signal protein kinases, transcription factors
Ras protein normal vs cancer
normal: transduces signals from growth hormones to activate proliferation
cancer: active even without hormone signal
cyclins normal vs cancer
normal: regulate progression through cell cycle
cancer: high level of cyclins allow cell cycle to continue when it shouldn’t
tumor supressor genes
shut off proto-oncogenes
can be mutated to loss-of-function and allow proliferation
p53 tumor surpressor
normally low in cells so as not to alarm
increased in cells that have sustained DNA damage
activates transcription of cell cycle arrest genes and slowing DNA replication
mutated in over 50% of cancers so it cannot repress
p53 relation to apoptosis
if DNA damage is severe, p53 will activate apoptosis
pRB protein
normally sequesters transcription factors to keep cell cycle in arrest
block at G1/S stage
when mutated, S phase continues and there is proliferation
carcinogen
compound or condition that can damage DNA
natural carcinogen example
aflatoxin is made by mold
ames test
analyzes a compound for ability to create mutation based on rate of mutated bacteria
combined with liver extract which has enzymes to activate potential carcinogens
risk factor of viral gene transfer
retroviruses integrate into a host and can convert a proto-oncogene to an oncogene based on proximity of integration
capture of genes by retroviruses
pick up cellular DNA and copy oncogene form then transfer oncogene to new cell
significant cancers from viral infection
HPV - cervical cancer
Hepatitis B - liver cancer
inherited mutations often need more mutations to cause cancer
inherited mutations often need more mutations to cause cancer
early detection
cancer treatment is highly successful if caught early
routine physicals
cancer treatment
surgery
radiation and chemotherapy to target dividing cells
novel cancer treatments
boosting immune system with antigens, interferons, and antibodies
molecular targeting to inactivate cancer — possible because of genetic testing to have better cancer characterization
marijuana
may reduce cancer symptoms
anti-angiogenesis therapy
increase angiogenesis inhibitors so tumor can’t vascularize
limits ability to move and survive
stabilizer, doesn’t cure