Nuclear
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
Nuclides with common properties
Isotopes have the same atomic number Z, and so have the same chemistry.
Isobars have the same atomic mass A=N+Z.
Isotones have the same number of neutrons N.
Early experiments to determine atomic structure
The Rutherford experiment established that there is a positively charged nucleus at the centre of the atom via scattering an α-particle on larger atoms.
Assumed scattering of point-like, spinless objects
Scattering experiments with electrons probed the charge distribution of the nucleus. First calculated by Mott.
Takes spin of electrons into account.
Mass using Avogadro’s constant
mass [in grams]=Avogadro’s constant(NA)number of nuclei (N)×atomic number (A)
where number of nuclei N can be found via the activity.
Reaction rate for fission
The reaction rate is given by: Wfission=JNσfission, where J is the neutron flux, N is the number of nuclei (which can undergo fission), and σfission is the neutron cross section for fission.
Recall that power is related to this by P=WfissionEfission, where E is the energy released per fission.
Similarly, the capture rate is given by Wcapture=JNσcapture, where N is the nuclei which capture neutrons (may be different to N for fission).
To find a rate, use the ratio of the fission and capture rates to cancel out J.
Units of rate is atoms per second.
How to find mass if given a number of atoms?
The mass is given by the product of the number of atoms, their atomic number, and the unified atomic mass unit.
Estimate of radius from atomic number
R=R0A1/3=1.2fmA1/3
Mass deficit
Nuclei are lighter than the sum of the mass of the constituting nucleons. This “missing mass” is the mass deficit, and is due to the binding energy.
Binding energy
Is the energy released when nucleons form a nucleus, and is equivalent to the mass deficit, (so can be computed by E=Δmc2).
Generally, binding energy is the minimum energy required to disassemble a system of particles into its constituent parts.
Binding energy per nucleon
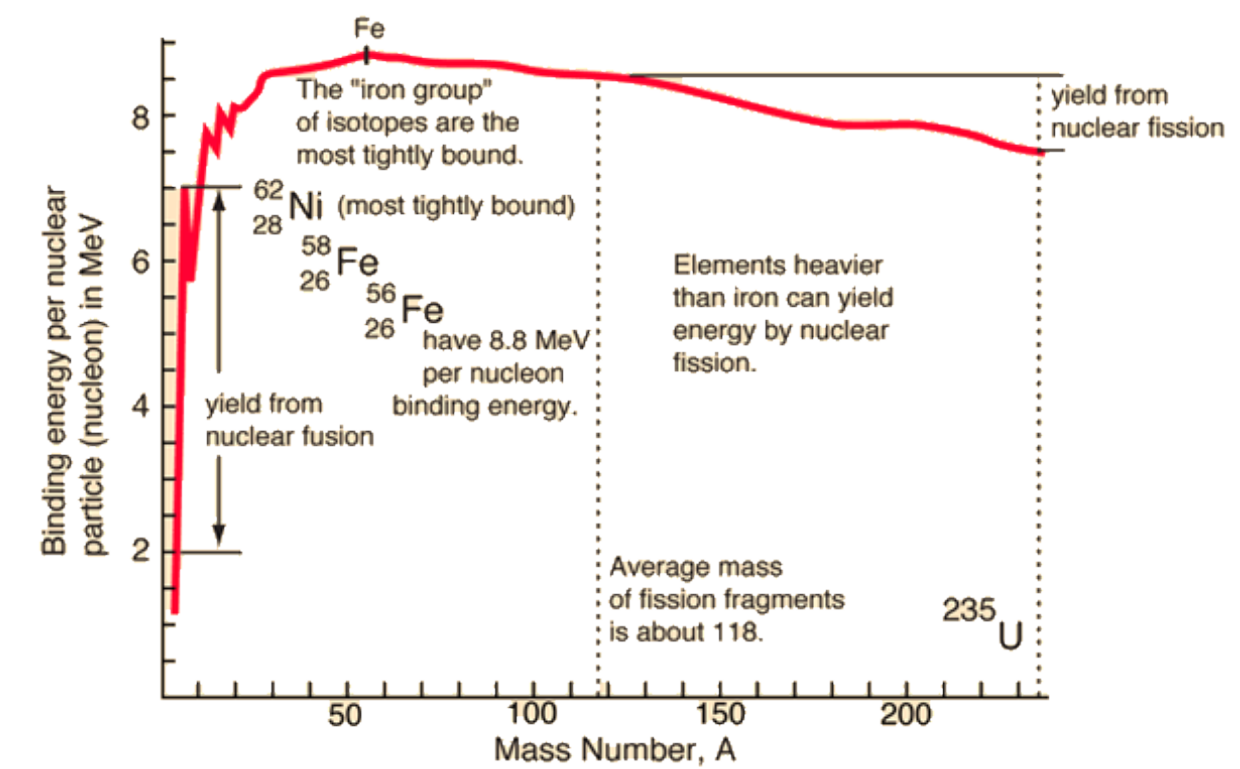
Iron has the highest binding energy per nucleon, and is the most tightly bound (most stable).
Semi empirical mass formula (SEMF)
Attempts to describe the mass M(Z,A) of nuclei depending on A and Z.
Assumed the liquid drop model, where interior density are approximately constant.
The binding energy B(Z, A) can be calculated from the SEMF via B(Z,A)=−ΔM(Z,A).
Which terms increase/decrease binding energy in the SEMF?
Defined binding energy B(Z, A) = −∆M(Z, A)
Volume term, increases binding energy as nucleons are bound to each other
Surface term, decreases binding energy as nucleons on the surface lack binding partners
Coulomb term, decreases binding energy as same charge protons repel each other
Asymmetry term, decreases binding energy as an excess of either protons or neutrons carries an energy penalty (as excess nucleons occupy higher energy levels)
Pairing term, even-even nuclei are more tightly bound and hence have an increased binding, odd-odd nuclei are less tightly bound and hence have a decreased binding, (in even-odd effects cancel and term becomes zero)
Up to which element is fusion possible?
Element 56Fe or 62Ni. The binding energy reaches a maximum around that value, so energy is released when lighter nuclei fuse together up to that point.
Fusion in stars occurs up to iron 56Fe, but it turns out that the most tightly bound nucleus is 62Ni.
How to find value of A that maximises binding energy?
Assuming that Z=N=A/2:
Substitute all Z for A/2 in the SEMF, where M(Z,A)=−B(Z,A)
Calculate B/A
Differentiate B/A with respect to A and set this to zero, i.e. ∂A∂(B/A)=0
Rearrange for A and substitute in values of constants
Should give A~48
Constituent mass term of the SEMF
The 0th term adds the masses of all constituents:
f0(Z,A)=Z(Mp+me)+(A−Z)Mn
Volume term of the SEMF
The 1st term assumes that nucleons bind to each neighbour and the binding energy is proportional to the volume of the nucleus.
In the simple drop model we can assume a sphere of radius R∼A1/3.
Binding energy reduces the mass of the nucleus and hence is negative:
f1(Z,a)=−a1A
Surface term of the SEMF
2nd term. Nucleons on the surface lack binding partners, hence the binding energy is reduced by an amount proportional to the surface area:
f2(Z,A)=+a2A2/3
Coulomb term of the SEMF
3rd term. Protons carry positive electric charge and hence repel each other according to the Coulomb law, reducing the binding energy.
f3(Z,A)=+a3A1/3Z2
Asymmetry term of the SEMF
4th term, and cannot be derived from the liquid drop model, unlike all other terms. Protons and neutrons are fermions hence the Pauli exclusion principle applies. For unequal numbers of p and n, higher energy levels are filled, so there is an energy penalty for an excess of p or n.
f4(Z,A)=+a4A(Z−A/2)2
Pairing term of the SEMF
5th term. Nucleons in the same spatial state, but opposite spin directions can combine to give a wave-function of spin-0.
This bosonic state follows Bose–Einstein statistics and hence the wavefunctions overlap heavily.
This means the nucleons are tighter bound if they can be paired and the binding energy is maximised when both Z and N are even f5(Z,A)=−f(A)=−a5A−1/2
In an odd–even or even–odd nucleus the effects cancel and f5(Z,A)=0.
Finally for an odd–odd nucleus with both an unpaired proton and an unpaired neutron the binding energy is reduced by f5(Z,A)=f(A)=a5A−1/2
Activity
Activity A is the number of decays in a given time,
A=−dtdN=τN
where τ is the lifetime, and N is the number of nuclei.
Integration gives the number of nuclei at a time t as N(t)=N0e−t/τ.
Half-life
The time after which half of the nuclei have decayed. It is related to the lifetime:
0.5N0=N0e−T1/2/τ⇒T1/2=τln2
Units used to measure activity
Becquerel, where 1 Bq=1 decays−1
Curie, where 1Ci=3.7×1010 decays−1
Units used to measure expose and dose
Roentgen is used to measure exposure, 1 R = 2.58 C/Kg.
Rad or Gray are used to measure absorbed dose, 1 rad = 0.01 J/Kg, or Gray, 1 Gy = 1 J/Kg.
Rem or Sievert Sv is used to measure the biologically equivalent dose, were varying biological effects of α, β and γ radiation into account.
How does mass depend on Z for a fixed A?
From the SEMF, for a fixed mass number A, the mass depends quadratically on the number of protons Z.
Hence a parabola is formed for every odd mass number A. The most stable nucleus is found at the bottom of this parabola.
Effects of an odd mass number
An odd mass number leads to either an odd–even or even–odd nucleus for which the pairing effects cancel and hence only a single parabola is formed.
Nuclei with an even number of protons Z and even number of neutrons are more tightly bound than odd–odd nuclei, due to the pairing term.
Hence the SEMF shows two parabolas separated by 2ap/A. The more stable nucleus is usually found on the lower parabola with even–even nuclei.
Favourable decay options for nuclei
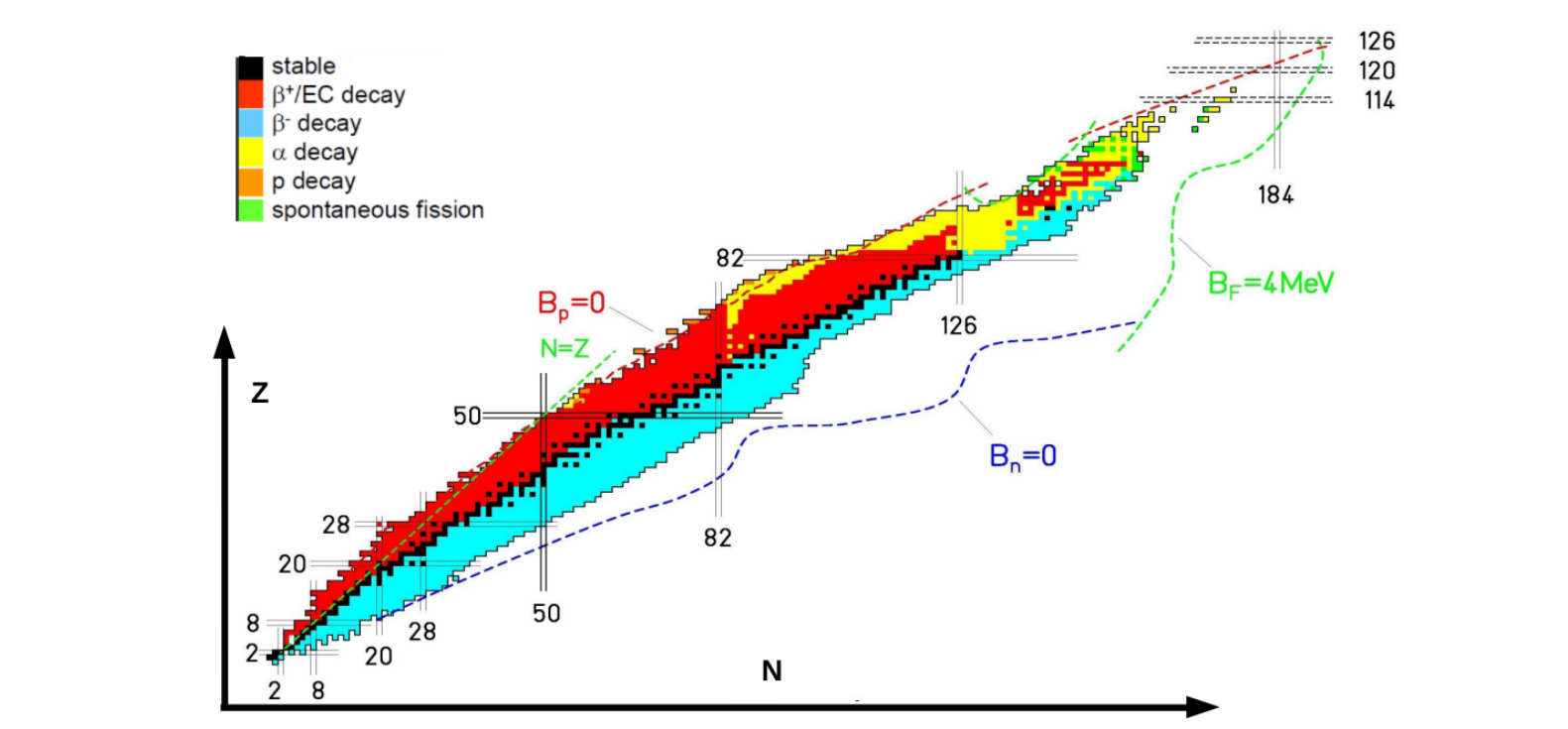
Elements on the left of the parabola minimum are neutron unstable, i.e. it is energetically favourable to turn a neutron into a proton. This is β− decay,
n→p+e−+νˉe
The nucleus moves closer to the minimum of the SEMF parabola, so this decay is allowed if M(Z, A) > M(Z + 1, A).
Elements on the right of the parabola minimum are proton unstable, i.e. it is energetically favourable to turn a proton into a neutron. This is β+ decay,
p→n+e++νe
The nucleus moves closer to the minimum of the SEMF parabola, with the most stable isobar. This decay is allowed if M(Z, A) > M(Z − 1, A) + 2m_e.
Also, p → n via electron capture, where p+e−→n+νe, which is allowed if M(Z, A) > M(Z − 1, A) + \epsilon, where ϵ is the excitation energy of the atomic shell of the captured electron.
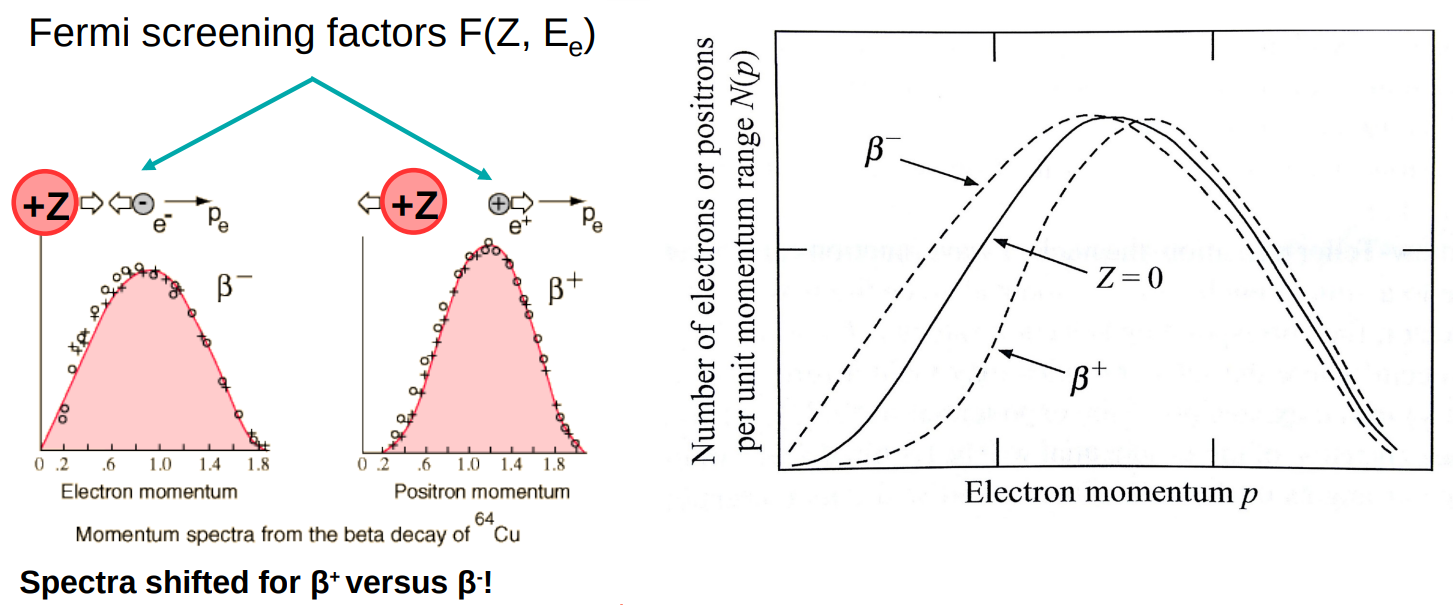
Beta decay Fermi theory vs reality
Fermi theory treats beta decay as a 4-point interaction, but we now know that it goes via a W boson.
Screening potential
Electrons and positrons are deflected differently in the Coulomb potential of the nucleus, giving rise to a screening potential F(Z,pe).
This leads to a sharper perk in momentum for positrons than electrons.
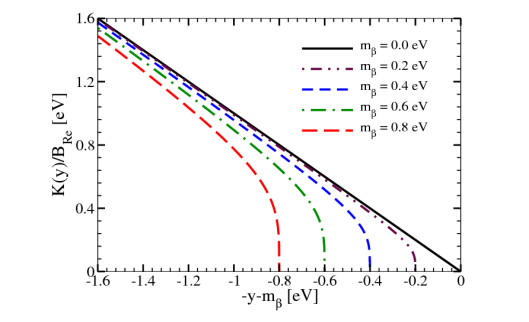
How can neutrino mass be measured using beta decay?
Using the transition rate W and the momentum of electrons/positrons via dW/dpe. A Kurie plot can be used to see the neutrino mass, as massless and massive neutrinos will produce different results (x-axis is electron KE).
When is alpha decay favourable?
Emitting an α-particle is energetically favoured if B(2, 4) > B(Z, A) − B(Z − 2, A − 4).
An estimate of the binding energy difference can be found by using the SEMF and the slope of the B/A plot.
What kind of process is alpha decay?
A quantum tunnelling process. Though the large binding energy of the α-particle increases the energy available for it to escape the parent nucleus, we still have E_α< V_C.
Transmission probability through the Coulomb barrier can be calculated using the Gamov factor.
Gamma decay
Nuclei often de-excite by emitting a photon via γ-decay, often immediately after a preceding beta or alpha decay.
Meta-stable states
Nuclei which have relatively long gamma decay half-lives are called meta-stable states.
Spontaneous fission
Occurs for heavier nuclei, where spontaneous fission with approximately equal mass daughters is energetically allowed for A>100.
Fermi gas model assumptions
Assumes a simple 3D well potential (different for protons and neutrons),
Assumes nucleons can move freely inside the nucleus. This agrees with the idea that they experience an overall effective potential created by the sum of the other nucleons.
This model can explain terms in SEMF that were not derived from the liquid drop model.
When does J=0?
J=0 in the shell model if:
all shells are filled and there are no unpaired nucleons
or there are an even number of protons and neutrons in each sub-shell including the last one.
According to the pairing hypothesis for ground state nuclei, pairs of neutrons and protons in a given sub-shell always couple to give a combined angular momentum of zero, even when the sub-shell is not filled.
How many nucleons can a state with a certain value of angular momentum j have?
A state with total angular momentum j=l±1/2 can contain a maximum number Nj=(2j+1) nucleons. The value of l can be determined by considering the parity of the state.
Collective model
Is an attempt to bring together shell and liquid drop models, where:
Interactions between nucleons lead to permanent deformation of the potential.
Deformation represents collective motion of nucleons and are related to liquid drop model.
Two major types of collective motion: vibrations, which are surface oscillations, and rotations, which are the rotation of a deformed shape.
Potential well for protons vs neutrons in the Fermi gas model
The potential well is deeper for neutrons than it is for protons, as heavy nuclei have a surplus of neutrons due to Coulomb repulsion of protons reducing the binding energy.
The nucleons fill energy levels in the well up to the Fermi energy.
Fermi momentum
In the nuclear ground state, all states up to a maximum momentum, the Fermi momentum pF, will be occupied.
The number density is found by integrating the number of possible states available to a nucleon inside a volume V and a momentum region dp, up to pF.
Stability line if neutron and proton wells are equal
If the neutron and proton potential wells are the same, then the stability line can be approximated with Z=N=A/2, giving pFn=pFp.
In reality, these momenta differ due to the difference in their potentials caused by the Coulomb repulsion felt by protons.
Depth of the potential well
The potential well is the sum of the Fermi energy (energy of the highest occupied state) and the binding energy B/A.
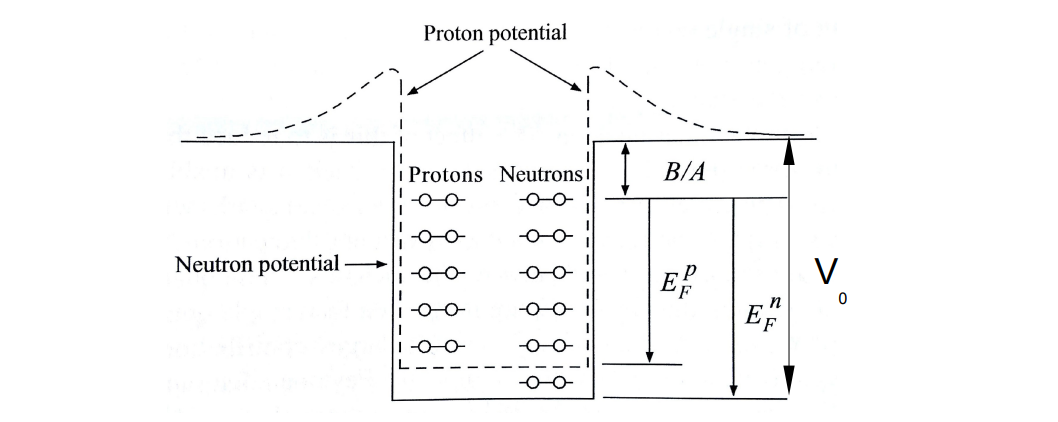
V0=EF+B/A∽40MeV
Origin of the asymmetry term
Cannot be derived from the liquid drop model, but can be derived from the Fermi gas model.
Found by considering the average KE per nucleon, integrating up to the Fermi momenta for p and n.
For a fixed A, a Taylor expansion can be done about the minimum at Z=N=A/2 to give:
Ek(N,Z)∼10M3(Rℏ0)2(89π)2/3(A+95A(N−Z)2+...)
where the first term gets absorbed by the volume term, and the second corresponds to the asymmetry term.
Why do heavy nuclei have an excess of neutrons.
Coulomb repulsion reduces binding energy for protons and dominates for heavier nuclei. So even though the asymmetry term prefers nuclei with N=Z, the Coulomb term gives the result of an excess of neutrons over protons.
Shell model notation
Describes the structure of the nucleus in terms of energy levels or shells, similar to the atomic model.
A level filled with N nucleons is written as (nlJ)N, where n is the main quantum number, l is the orbital angular momentum and J is the total angular momentum of the level.
So shell configurations are written down like N:(1s1/2)2(1p3/2)4(1p1/2)2(1d5/2)6(2s1/2)2... Z:(1s1/2)2(1p3/2)4(1p1/2)2(1d5/2)6(2s1/2)2...
Note that the symmetries and degeneracy from nuclear potentials are different from Coulomb potentials used to describe atomic levels.
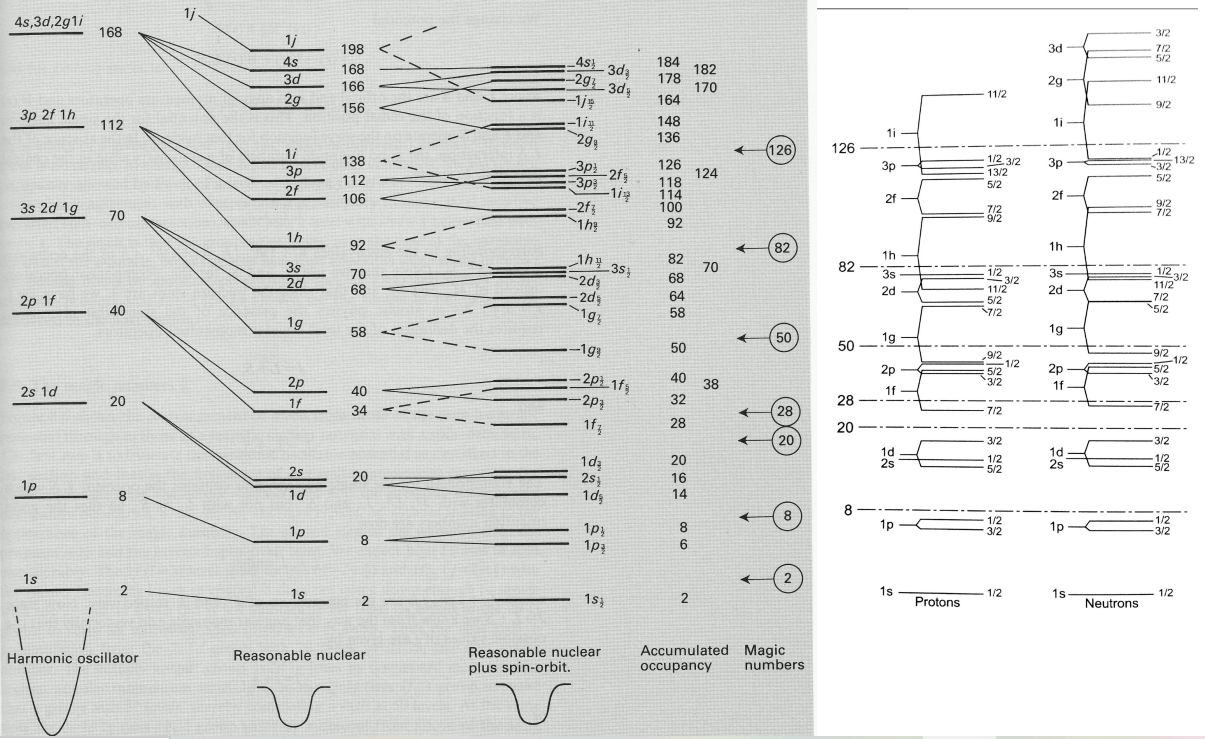
Magic numbers
Nuclides with a magic number of protons or neutrons are particularly tightly bound, making them very stable.
Magic nuclei have more stable isotopes, a larger nuclear excitation energy, and for magic neutron numbers, a smaller capture cross section.
Doubly magic nuclei are those where both the number of protons and neutrons is magic.
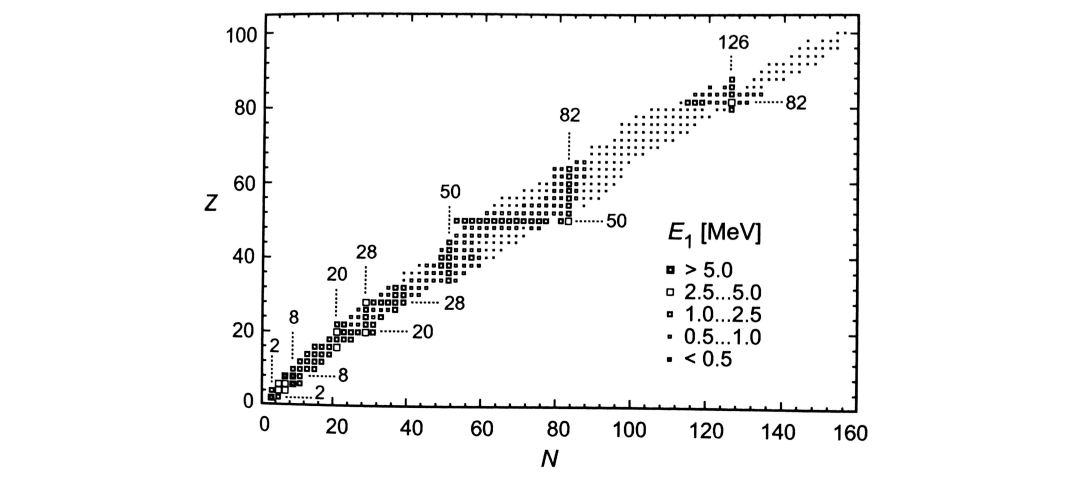
The existence of magic numbers suggests a shell structure of energy levels.
Potentials to explain magic numbers
Calculations using simple square potential well or the harmonic oscillator potential do not lead to energy levels consistent with the observed magic numbers.
The Woods-Saxon potential is an intermediate form between a simple square potential well and the harmonic oscillator potential, but still does not describe observations fully.
Inclusion of spin-orbit coupling explains the energies, as this gives an energy level splitting which increases linearly with the angular momentum.
Pairing hypothesis
States that: In a fully filled shell, for every nucleon with a projection of J along a chosen axis mJ, a nucleon with the opposite projection −mJ exists and the two nucleons pair up to combine to zero total angular momentum.
This is true for all even pairings of nucleons.
How is the spin of a nucleus determined?
By the angular momentum contributions of the constituent nucleons, for which odd/even nucleon contributions are different.
Angular momentum contributions to nuclei
According to the pairing hypothesis all nuclei with fully filled shells and all even–even nuclei will have spin 0.
For odd–even and even–odd nuclei the spin will be given by the total angular momentum of the unpaired nucleon.
In odd–odd nuclei with both an unpaired proton and neutron, the angular momenta have to be added. This means an exact prediction is not possible, but the spin sN of the nucleon will be in the range ∣Jp−Jn∣≤sN≤Jp+Jn, where Jp,n are the total angular momenta of the unpaired proton/neutron.
Parity
Intrinsic parities of the proton and neutron are +1, and parity due to orbital angular momentum is given by P=(−1)l.
Pairing hypothesis for parity
Even–even nuclei have even parity.
Even–odd and odd–even nuclei, the last unpaired nucleon determines parity.
In an odd–odd nucleus with both an unpaired proton and neutron, use the fact that parity is a multiplicative quantum number: Pnucleus=Plast proton×Plast neutron.
Predictions of the shell model
Determining nucleus spin by the angular momentum contributions of the constituent nucleons.
Determining the parities of nuclei.
Magnetic moment of a nucleus, but these predictions are not very reliable.
Excited nuclei states, but it is difficult to predict the correct order of these.
Predicting the magnetic moment of a nucleus
Are done via μ=gjℏ⟨j⟩μN for the shell model, but these are not very reliable, likely due to deformations that have to be taking into account.
Excited nuclei states
Excitations from the ground state are possible by promoting nucleons from their ground state level into higher levels.
However, predicting the correct order of these excited states proves to be difficult, especially the calculation of higher excited states.
Fission vs fusion
Fission: Splitting a heavy nucleus results in two lighter nuclei, each with larger negative binding energy. This results with less total energy in the nuclei, and the remaining energy is released.
Fusion: Fusing two light nuclei into a heavier nucleus with a larger negative binding energy. This results with less total energy in the nucleus, and the remaining energy is released.
Spontaneous vs induced fission
Spontaneous fission refers to when a fission process occurs without external action, and is only favourable for very heavy elements with Z > 116 and A ≥ 270.
Induced fission and is achieved by supplying a flow of neutrons to supply the energy needed to overcome the barrier.
Activation energy (fission barrier)
The energy required in order to induce fission.
Consider the fission process by starting with the deformation of a spherical nucleus into a prolate shape, then the binding energy changes as the nucleus deforms.
As the nucleus stretches into a prolate shape, the surface term will increase and the Coulomb term will decrease (assuming constant volume).
If the change in Coulomb term is larger than the change in the surface term then the deformed shape will be energetically favourable and the nucleus is unstable, meaning that spontaneous fission can occur.
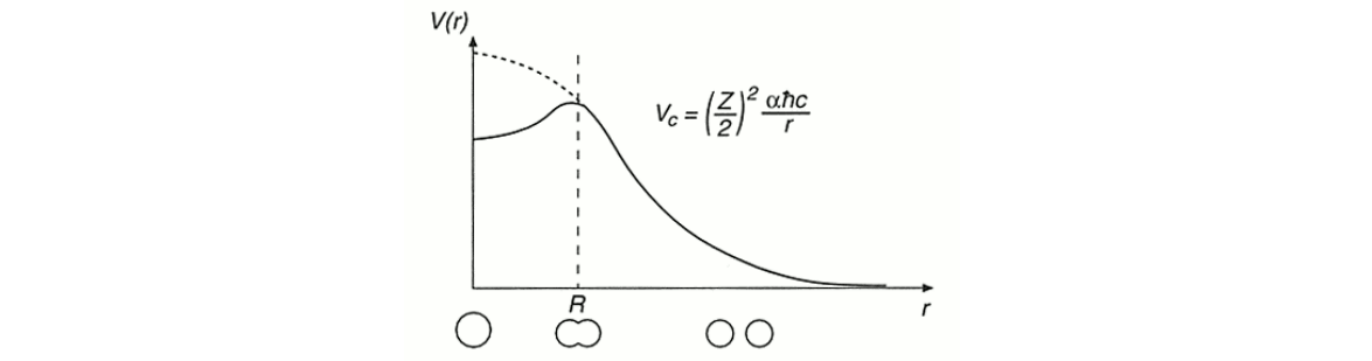
Why are neutrons used to induce fission?
Since neutrons are electrically neutral, they can approach the nuclei and be attracted by the strong nuclear force.
A flow of low energy neutrons can induce neutron capture reactions, pushing the nucleus into an excited state above the fission barrier.
When a nucleus absorbs a neutron some energy is released due to the binding energy of that neutron. If this energy is as large as the activation energy then fission can be induced.
Fissile materials
Materials which only require slow neutrons to induce fission.
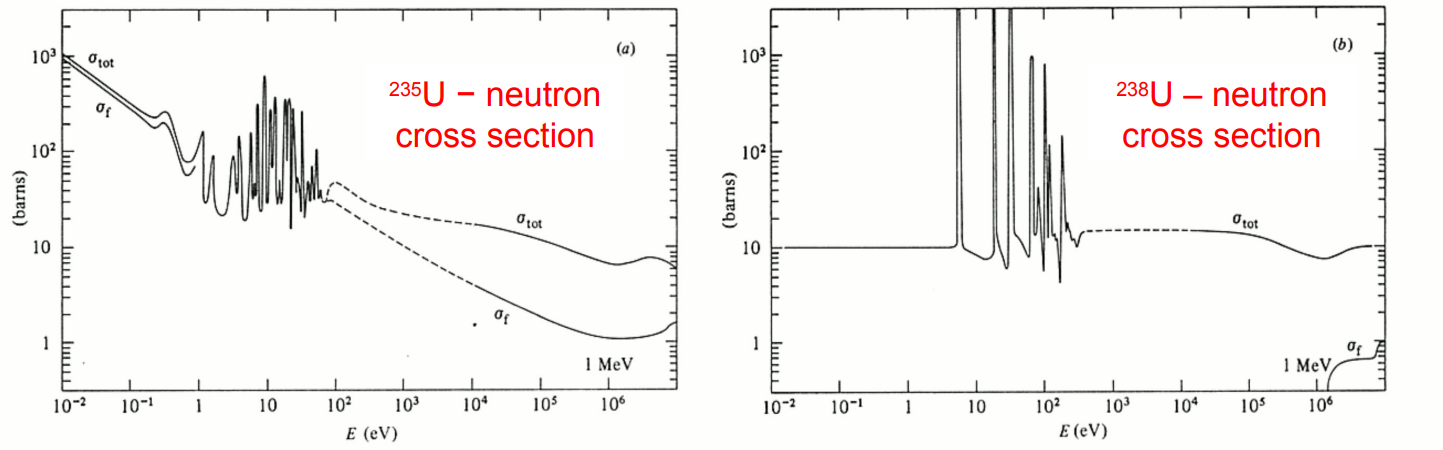
Nucleus-neutron cross sections
The total cross section includes contributions from elastic and inelastic scattering, and from neutron absorption.
There are resonant regions where the compound nucleus produced after the neutron absorption is in a region of excitation, where its energy levels are well separated.
Chain reaction
If the probability that a given neutron induces fission is q and each fission reaction produces an average of n neutrons, then each neutron will lead to (nq−1) additional neutrons in a time tp, where tp is the average time before absorption of the neutron occurs.
This gives a number of neutrons of:
N(t)=N(0)e(nq−1)t/tp
Reaction types via number of neutron
UsingN(t)=N(0)e(nq−1)t/tp:
For nq < 1, N(t) decreases exponentially, the process is said to be subcritical and the reactions will soon die out.
For nq=1, N(t) remains constant, the process is said to be critical and the conditions are right for a sustained, controlled reaction as is required in a nuclear power plant.
For nq > 1, N(t) increases exponentially and the process is said to be supercritical. The energy will grow very rapidly, leading to an explosion, which is needed in a nuclear fission bomb.
Conditions for a nuclear reactor plant
To produce power from nuclear fission, a critical reaction is required, with nq = 1.
To ensure a critical reaction in uranium:
Enrich the uranium so that it contains 2–3% of U-235, making it more likely that a neutron will induce fission in a U-235 nucleus.
Surround the natural uranium fuel in a large volume of moderator material, which slows down the fast neutrons produced in fission via elastic collisions. The slow neutrons are not energetic enough to be absorbed into a U-238 resonance, and so are more likely to meet and induce fission in a U-235 nucleus, the cross section for which is high at low energies.
How are the conditions for a controlled nuclear reaction sustained?
Operation with precisely nq = 1 is achieved with control rods that are mechanically inserted into the reactor whenever the reaction needs to be reduced.
Made out of materials with a very high absorption cross section for neutrons, usually Cadmium.
Can take delayed neutrons into account to require (nprompt+ndelayed)q=1, which keeps npromptq enough below zero that small variants in q cannot lead to an explosive reaction. Without this, fluctuating temperatures would lead to explosive reactions on small enough timescales that mechanical insertion of control rods could not prevent.
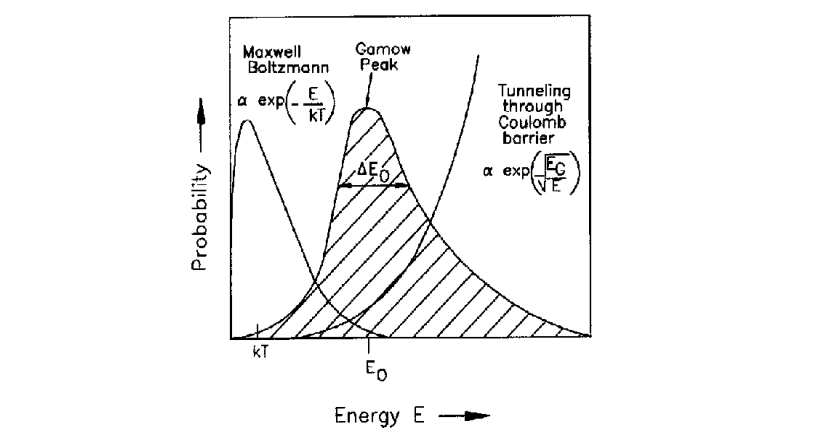
How can fusion occur at lower temperatures than expected?
Quantum tunnelling: similar to α-decay, the probability to penetrate an energy barrier via quantum tunnelling is e−G, where G is the Gamow factor: G=EG/E, where EG increases as the barrier increases and E is the energy of the particles.
A collection of nuclei with a given mean energy will have a Maxwellian distribution of energies about the mean and the distribution of the energies will have the form e−E/kBT.
The effect from quantum mechanical tunnelling increases with energy and that from the energy distribution of the particles decreases with energies, leading to a small range of energies which fusion can take place at a non-negligible rate.
Proton-proton fusion chain
Occurs in the sun via:
Two hydrogen nuclei (protons) fuse to form a deuterium nucleus: 11H+11H→12H+e++νe+0.42 MeV.
The deuterium fuses with more hydrogen to produce helium and a photon: 11H+12H→23He+γ+5.49 MeV.
Two helium nuclei (from the above two steps occurring twice) fuse to form an α particle plus some extra hydrogen nuclei: 23He+23He→24He+11H+11H+12.86 MeV.
The energy released in each step is the difference in binding energies between the initial and final states.
For the 1st step, the differences in the masses of the particles in the initial and final states must also be taken into account.
Overall PPI cycle equation
4(1H)→4He+2e++2νe+2γ
Which is the slowest step in the p-p chain?
The first step is slow as it involves the weak interaction and it therefore sets the scale for the long lifetime of the sun.
Extra energies and losses in the p-p chain
(For a complete chain the first two steps occur twice.) The positrons produced will annihilate with electrons in the plasma providing extra energy.
The neutrinos produced will carry of some energy which will be lost in space.
The rest of the energy is transported to the surface of the sun and emitted as photons or ejected high-energy particles.
Which reactions are candidates for fusion reactors?
Deuterium–deuterium fusion: via 12H+12H→23He+n+3.27 MeV or 12H+12H→13H+p+4.03 MeV
and tritium–deuterium fusion: 12H+13H→24He+n+17.62 MeV
(Proton-proton interactions in the sun are very slow and cannot be used).
How can plasma in fusion reactors be contained?
magnetic confinement, where the charged particles in the plasma follow a helical path as they curve round a magnetic field with a direction that points in a circle around a doughnut shape
inertial confinement, where pulsed lasers bombard small pellets of a tritium–deuterium mixture in many directions at the same time at very high energies.
Lawson criteria
Where the ratio of energy output to energy input (required to reach such high temperatures) is greater than one, meaning that fusion is feasible for a source of power.