2026 Equilibrium Chemistry Exam
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
Collision Theory
For a chemical reaction to occur- the particles must collide with the correct orientation and with sufficient energy to break existing bonds/forces of attraction (called activitation energy)
What are the five factors that can change the speed of chemical reactions?
Concentration (Solutions only), pressure (gases only), particle size (solids), catalysts and temperature
Explain why a concentrated solution will increase the rate of reaction
more H+ ions in the same space, incr frequency of coll b/w _ and _, incr frequency of successful coll and incr rate of reaction
Partial pressure
The pressure of one gas in a mixture of gases
Explain why high pressure increases the rate of reaction
Gas particles are closer together, incr frequency of coll b/w, incr frequency of successful coll and incr rate of reaction
The effect of inert (unreactive) gases on the rate of reaction
Gases like argon although will incr pressure do not alter the rate of reaction as partial pressure of the reactants do not change
The effect of larger surface area on incr rate of reaction
Smaller pieces of __ have larger surface area and therefore more particles available for collisions, frequent coll b/w, frequency of successful coll and incr rate of reaction. *increase in particle size decreases rate, but increase in state of subdivision increases rate.
The effect of a catalyst on incr rate of reaction
Catalyst provides a reaction pathway of lower activitation energy, greater PROPORTION of coll will collide with energy greater than activation, greater PROPORTION of successful coll incr rate of reaction
Enzymes
Biological catalysts (proteins) that are used to change the speed of chemical reactions in plants and animals
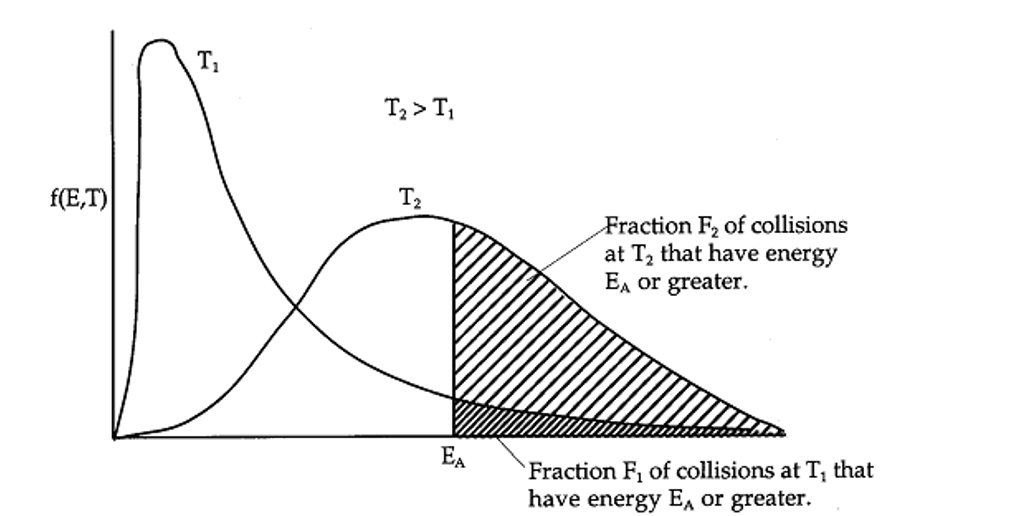
The effect off temperature on incr rate of reaction
Move faster and have a higher average kinetic energy, collide more frequently AND a ‘greater proportion of collisions occur with energy greater than activation energy’ (most important reason), frequency and proportion of successful collisions, incr rate of reaction.
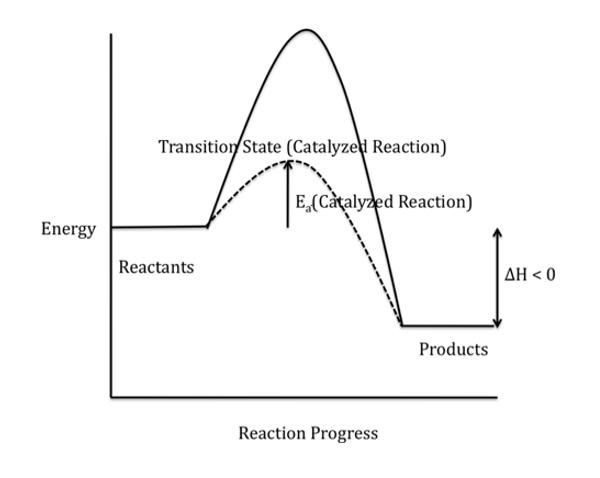
Draw and interpret potential energy diagrams for exothermic reactions and state characteristics of exothermic reactions
Energy is released to the surroundings, causing the surroundings to increase in temperature. The reactants have more energy than the products, ΔH is negative, and bond forming releases energy.
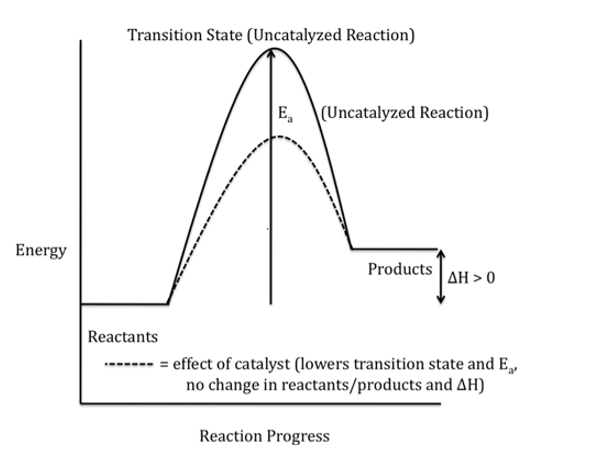
Draw and interpret potential energy diagrams for endothermic reactions and state characteristics of endothermic reaction
Energy is absorbed from the surroundings, causing the surroundings to decrease in temperature. The products have more energy than the reactants, ΔH is positive, and bond breaking absorbs energy.
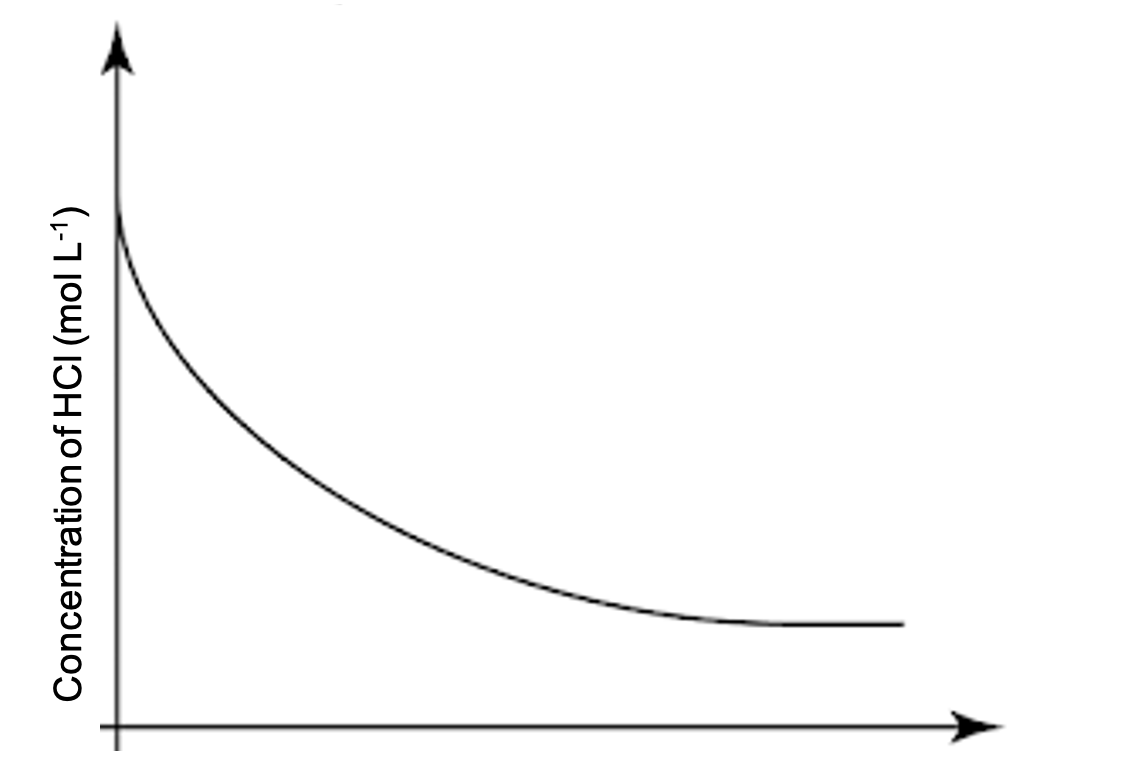
What are completed reactions
Reactions that continue until one of the reactants runs out (limiting reagent) and the reaction stops e.g. explosions (combustion), acid-metal reaction. The slope is exponential as the rate of consumption begins to decrease as concentration decreases.
What are reversible reactions
Reactions can go forward and backwards
Define an open chemical system
Chemical system that allows matter and energy to be exchanged with the surroundings.
Define a closed chemical system
Chemical system that allows energy (heating/cooling), but not matter, to be exchanged with the surroundings to establish equilibrium. Chemicals can’t enter or escape.
Define equilibrium
Equilibrium is the constancy of macroscopic properties in a closed system. It is dynamic because reactions are still occurring, but they equal and opposite to each other.
Define constancy of macroscopic properties
The concentration of reactants and products stay the same at equilibrium. No change is seen or can be measured.
What are three macroscopic properties of chemicals that can be used to identify from a qualitative description whether a particular chemical system is at equilibrium.
Colour, volume/pressure, temperature that remain constant when a system reaches equilibrium
Define the activation energy of a reaction and explain its significance to the reaction rate.
Activation energy (Ea) is the minimum energy that reacting particles must have for a collision to be successful and result in a chemical reaction. Only particles with energy equal to or greater than the activation energy can react. Most particle collisions do not lead to reaction because they do not have sufficient energy. Increasing temperature or using a catalyst increases the number (or proportion) of particles with enough energy to overcome the activation energy. This results in more successful collisions per second, increasing the rate of reaction.
Explain the reversibility of chemical reactions in terms of the activation energies of the forward and reverse reactions.
In a reversible reaction, both the forward and reverse reactions have activation energies. Reacting particles can undergo the forward reaction if they have sufficient energy to overcome the forward activation energy. Product particles can undergo the reverse reaction if they have sufficient energy to overcome the reverse activation energy. Because particles in the system can overcome the activation energy barriers in both directions, the reaction can proceed forwards and backwards. At equilibrium, particles overcome both activation energies at the same rate, so the rates of the forward and reverse reactions are equal.
Predict the effect of a decrease in temperature on a chemical system at equilibrium (given ΔH)
Endothermic reactions require energy and will be favoured by an increase in energy (temperature). Favours the endothermic reaction. Cooling favours the exothermic reaction, therefore the forward reaction is favoured
Predict the effect of a increase in temperature on a chemical system at equilibrium (given ΔH)
All reactions (both forward and reverse) will increase in speed with an increase in temperature. The forward reaction is exothermic. Heating favours the endothermic reaction, therefore the reverse reaction is favoured
Explain the effect of an increase in temperature changes in terms of forward and reverse reaction rates
Increases the rate of both forward and reverse reactions, the reverse reaction is favoured
Explain the effect of a decrease in temperature changes in terms of forward and reverse reaction rates
Decreases the rate of both forward and reverse reactions, the reverse reaction is favoured
Predict the effects of changes in adding concentrated HCl on systems at equilibrium
Change to system: increase in [H+]
Partial counteraction: decrease the [H+]
Reaction favoured: forward (consumes H+)
Equilibrium position: shifts to the right
Result: more products formed
Predict the effects of changes in adding concentrated Cr₂O₇² on systems at equilibrium
Change to system: increase in [Cr2O72−]
Partial counteraction to change: decrease the [Cr2O72−]
Reaction favoured: reverse (consumes Cr2O72−)
Equilibrium position: shifts to the left
Result: higher proportion of reactants (more CrO42−)
Predict the effects of changes in adding NaOH(aq) on systems at equilibrium
Change to system: decrease in [H+]
Partial counteraction: increase the [H+]
Reaction favoured: reverse (produces more H+)
Equilibrium position: shifts to the left
Result: more reactants present
Predict the effects of changes in adding AgNO₃(aq)on systems at equilibrium
(Ag⁺ forms a precipitate with CrO₄²⁻)
Change to system: decrease in [CrO42−
Partial counteraction: increase the [CrO42−]
Reaction favoured: reverse (produces more CrO42−)
Equilibrium position: shifts to the left
Predict the effects of changes in partial pressure on systems at equilibrium
Increase pressure (decrease volume):
Favours the reaction with fewer moles of gas
Decrease pressure (increase volume):
Favours the reaction with more moles of gas
If moles of gas are equal:
No reaction is favoured
No shift in equilibrium
Explain the effect of adding more concentrated acid to the equilibrium using collision theory
Adding concentrated acid increases the hydrogen ion concentration
This increases the frequency of successful collisions between H+H^+H+ and reactant ions
The rate of the forward reaction increases with no immediate change to the reverse reaction
The forward reaction is favoured
Explain the effect of adding more precipitate to the equilibrium using collision theory
Adding AgNO₃ removes CrO42−CrO_4^{2-}CrO42− from solution as a precipitate
This decreases the frequency of successful collisions between reactant ions
The rate of the forward reaction decreases with no immediate change to the reverse reaction
The reverse reaction is favoured
Explain the effect of increasing pressure on the equilibrium using collision theory
Increasing pressure forces gas particles closer together
This increases the frequency of successful collisions
The rate of both forward and reverse reactions increases
The reaction with more moles of gas is affected more
The reaction with the greater rate increase is favoured
Explain the effect of dilutions on equilibrium using collision theory
Adding water decreases the concentration of all aqueous species…
This decreases the frequency of successful collisions…
This decreases the rate of both the forward and reverse reactions… As there are 4 moles of aqueous reactant and 1 mole of aqueous product, the rate of the forward reaction decreases by more than the rate of the reverse reaction.
This favours the reverse reaction. The reaction with the most moles of aqueous species is most affected.
State Le Châtelier’s Principle
If a chemical system at equilibrium is subjected to a change in conditions, the system will adjust to establish equilibrium in such a way as to partially counteract the imposed change.
Use Le Châtelier’s Principle to predict temperature changes in equilibrium proportions
Increase temperature: favours the endothermic reaction
Decrease temperature: favours the exothermic reaction
Both forward and reverse reaction rates change, but equilibrium shifts based on ΔH
Use Le Châtelier’s Principle to predict concentration changes in equilibrium proportions
Increasing concentration of a species favours the reaction that consumes it
Decreasing concentration of a species favours the reaction that produces it
Equilibrium shifts to partially counteract the change
Use Le Châtelier’s Principle to predict partial pressure / total gas volume changes in equilibrium proportions
Increase pressure → favours side with fewer moles of gas
Decrease pressure → favours side with more moles of gas
Equal moles of gas → no change in equilibrium (neither reaction is favoured)
Use Le Châtelier’s Principle to predict addition of a catalyst in equilibrium proportions
Catalyst increases the rate of both forward and reverse reactions equally
Equilibrium is reached faster
No change in equilibrium position or concentrations
Neither reaction is favoured
Use Le Châtelier’s Principle to predict dilution (aqueous systems) in equilibrium proportions
Adding water decreases the concentration of all aqueous species
The reaction with more aqueous particles is affected more
The reaction that produces more aqueous species is favoured
Equilibrium shifts to partially counteract the dilution
Effect of solids, liquids, and catalysts on equilibrium proportions
Changes in the mass of a solid, the volume of a liquid, or the presence of a catalyst have no effect on the relative proportions of products and reactants at equilibrium. A catalyst increases the rate of both forward and reverse reactions equally but does not change equilibrium concentrations
What factors change the equilibrium constant (K)?
The only factor that can change the value of the equilibrium constant K is a change in temperature, depending on whether the reaction is endothermic or exothermic.
Effect of temperature on K for an endothermic reaction
For an endothermic reaction, increasing temperature favours the forward reaction, resulting in a greater concentration of products, therefore K increases with increasing temperature.
Effect of temperature on K for an exothermic reaction
For an exothermic reaction, increasing temperature favours the reverse reaction, resulting in a lower concentration of products, therefore K decreases with increasing temperature.
Ionisation of water and temperature
Water remains neutral despite temperature changes because the [H⁺] is equal to [OH⁻], even though the values of both change.
Write the equilibrium law expression for systems at equilibrium
K= [products]/ [reactants]. Gases use: Concentrations in heterogeneous systems and Partial pressures in homogeneous gaseous systems. Pure solids and pure liquids are not included in equilibrium expressions.
Why is water not included in equilibrium expressions?
Water is a pure liquid and so does not have a concentration. Therefore, water is not included in the equilibrium law expression
Meaning of the equilibrium constant (K)
The equilibrium constant K:
Indicates the relative proportions of products to reactants at equilibrium
Has a fixed value for a given reaction at a given temperature
Provides no information about the rate of the reaction
Using K to predict equilibrium position (qualitative)
K > 100 → considerably more products than reactants. K < 0.01 → considerably more reactants than products. 0.01 ≤ K ≤ 100 → similar concentrations of reactants and products (only equal if K=1).
Define chemical synthesis
Chemical synthesis involves forming products with specific properties and may require sequences of reactions and careful selection of reagents and reaction conditions to optimise rate and yield.
What is a limiting reagent?
The limiting reagent is the reactant that is completely used up first and therefore limits the amount of product that can be formed in a chemical synthesis reaction.
What are enzymes in chemical synthesis?
Enzymes are protein molecules that act as biological catalysts, allowing reactions to occur at lower temperatures and pressures, making them economically viable (e.g. fermentation and lipase‑catalysed biodiesel).