TEAS CHEMISTRY
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What does PAN MAN stand for?
Protons, Atomic numbers, Number of Protons, Mass Number, for Add, for Neutrons.
What is sublimation?
Where a solid turns into a gas without first becoming a liquid.
What is deposition?
Where a gas turns directly into a solid.
Explain Catalyst.
A substance that increases the weight of a chemical without becoming part of the reaction.
H 2 0
Formula for water
Proton
Single positive charge.
Neutron
A particle with no electrical charge.
Electrons
Circle/Orbit around the nucleus. They have a negative charge. 2,000 times smaller then a proton.
Isotopes
Atoms of the same element that have a different number of neutrons.
Atomic Mass
Total number of protons and neutrons.
Atomic Number
Number of protons in an atom.
Ions
Ions are atoms that have gained or lost electrons, as a result has a positive or negative charge.
Cations
Are atoms that have lost on or more electrons and have a resulting positive charge.
Anions
are atoms that have gained one or more electrons and have resulting a negative charge.
What are the basic components of an atom and their charges?
Protons (+) and Neutrons (0) are located in the nucleus.
Electrons (-) orbit the nucleus in electron shells.
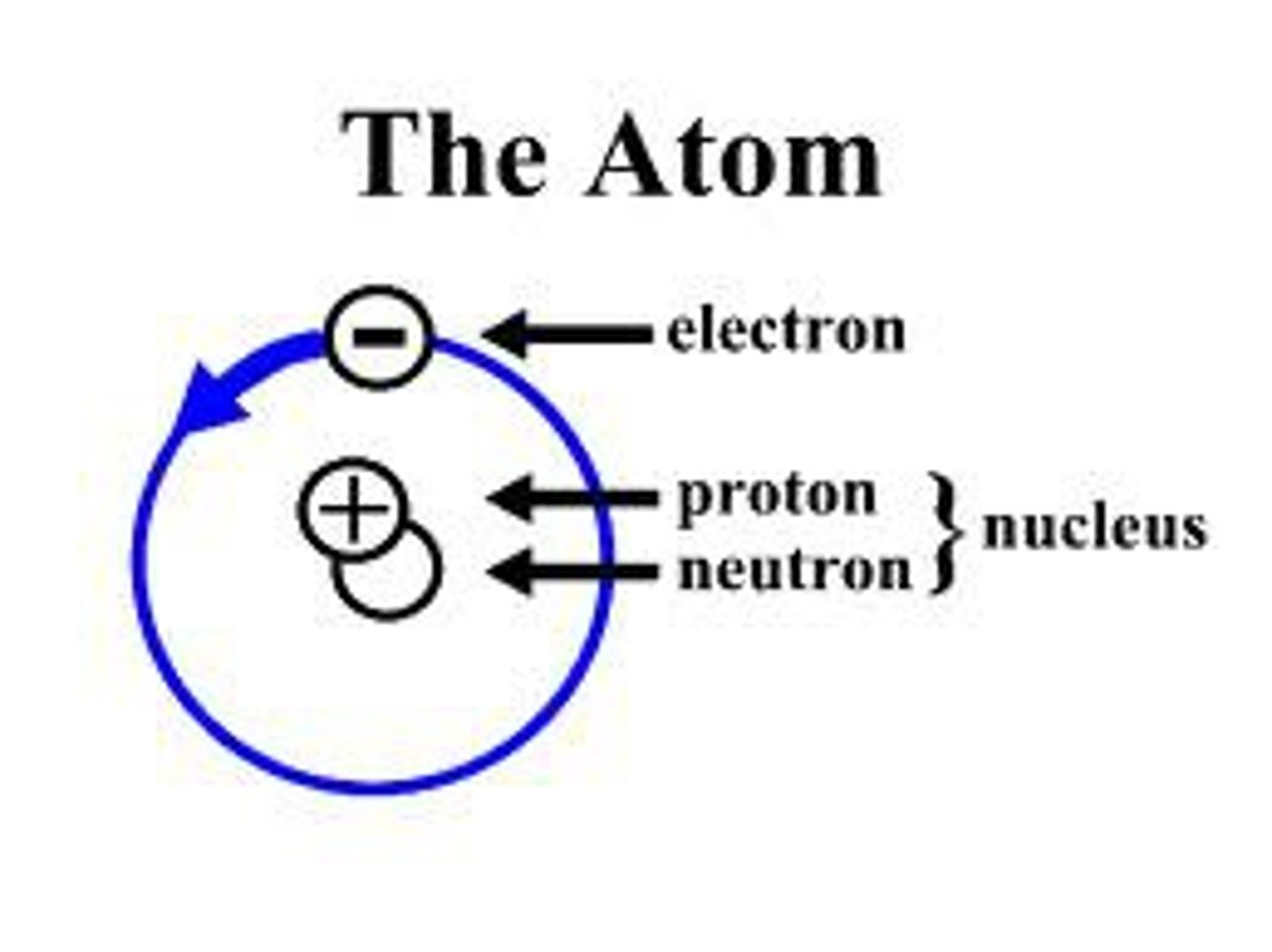
How do you identify the number of protons, neutrons, and electrons in an atom?
Atomic Number = Number of Protons (defines the element)
Mass Number = Protons + Neutrons
Electrons = Protons (in a neutral atom)
Ions: Gain or loss of electrons creates a cation (positive charge) or an anion (negative charge).
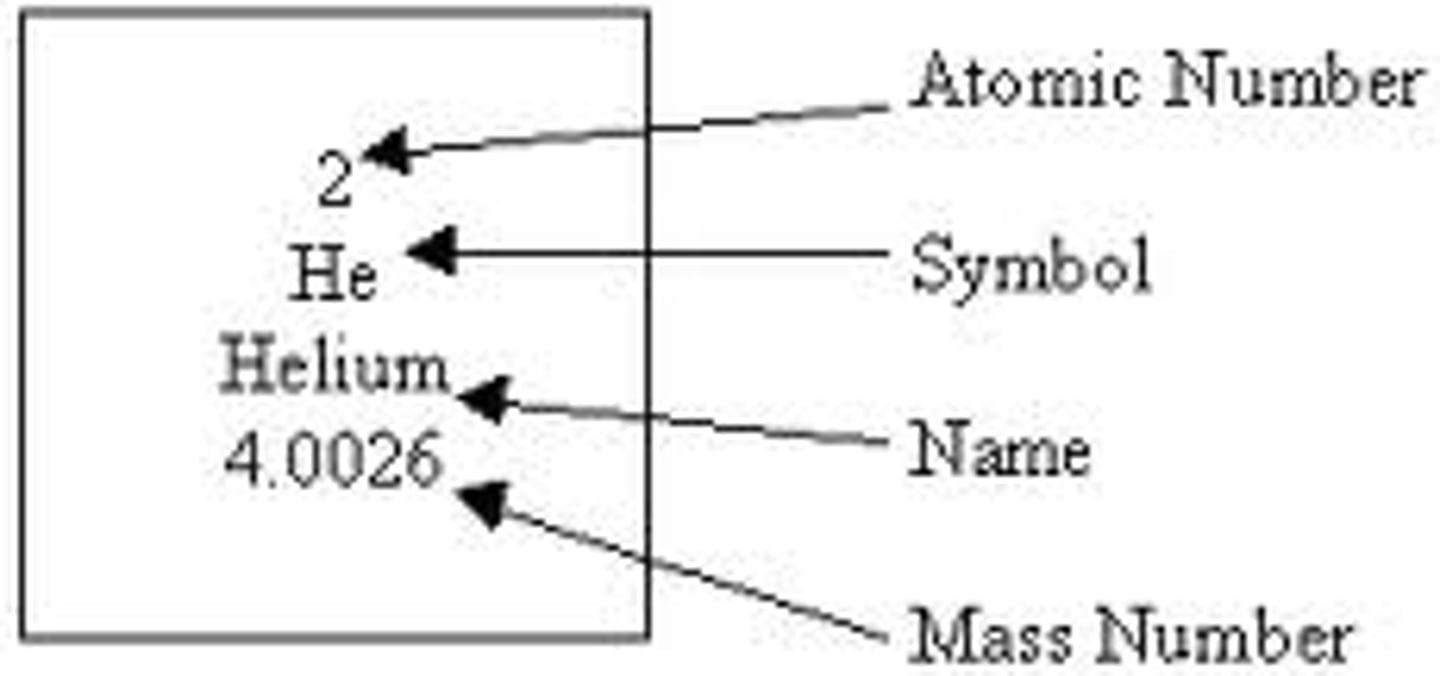
How can the periodic table be used to predict the properties of elements?
Groups (columns): Elements in the same group share similar properties.
Periods (rows): Indicate the number of energy levels in electrons.
Trends: Electronegativity: Increases across a period, decreases down a group. Atomic Radius: Increases down a group, decreases across a period. Ionization Energy: Increases across a period, decreases down a group.
What is the relationship between mass, volume, and density?
Mass: Amount of matter (g, kg)
Volume: Space occupied (mL, L, cm³)
Density Formula: Density = Mass/Volume (g/mL or g/cm³)
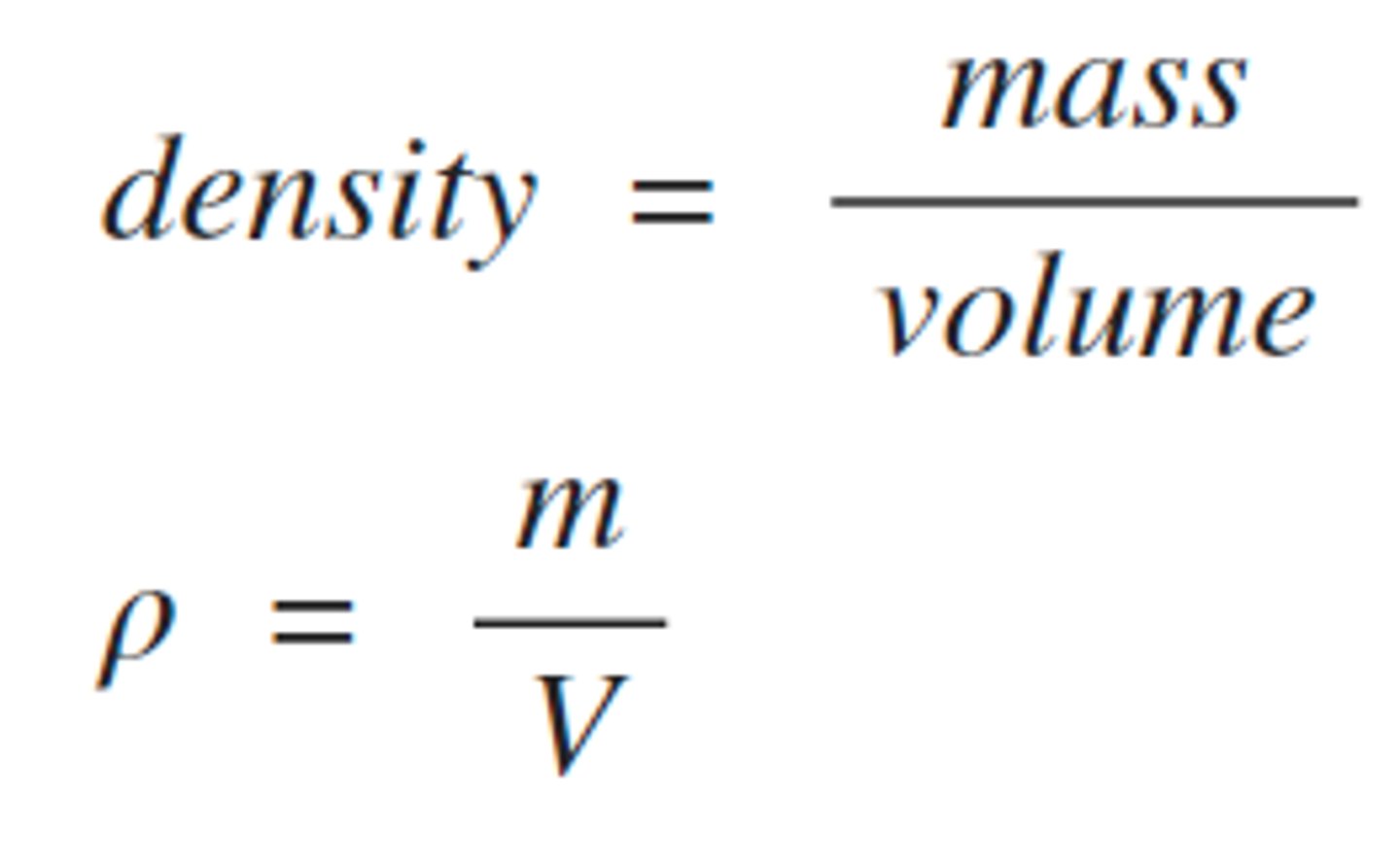
How do the states of matter differ in terms of molecular motion?
Solid: Fixed shape, slow molecular motion.
Liquid: Takes shape of the container, moderate molecular motion.
Gas: Expands to fill the container, fast molecular motion.
Plasma: Ionized gas with high energy and rapid molecular motion.
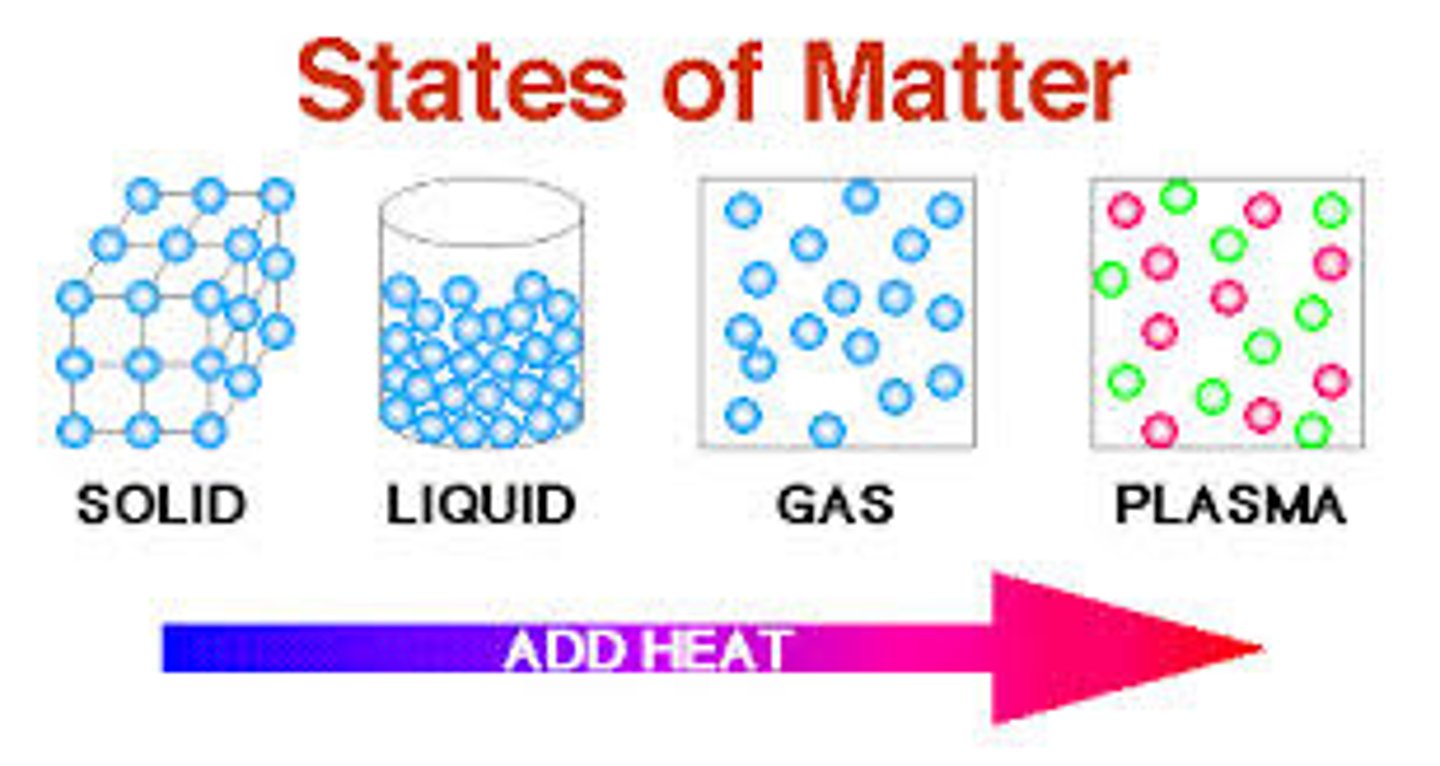
What are the common physical changes between states of matter?
Melting: Solid → Liquid
Freezing: Liquid → Solid
Evaporation: Liquid → Gas
Condensation: Gas → Liquid
Sublimation: Solid → Gas
Deposition: Gas → Solid
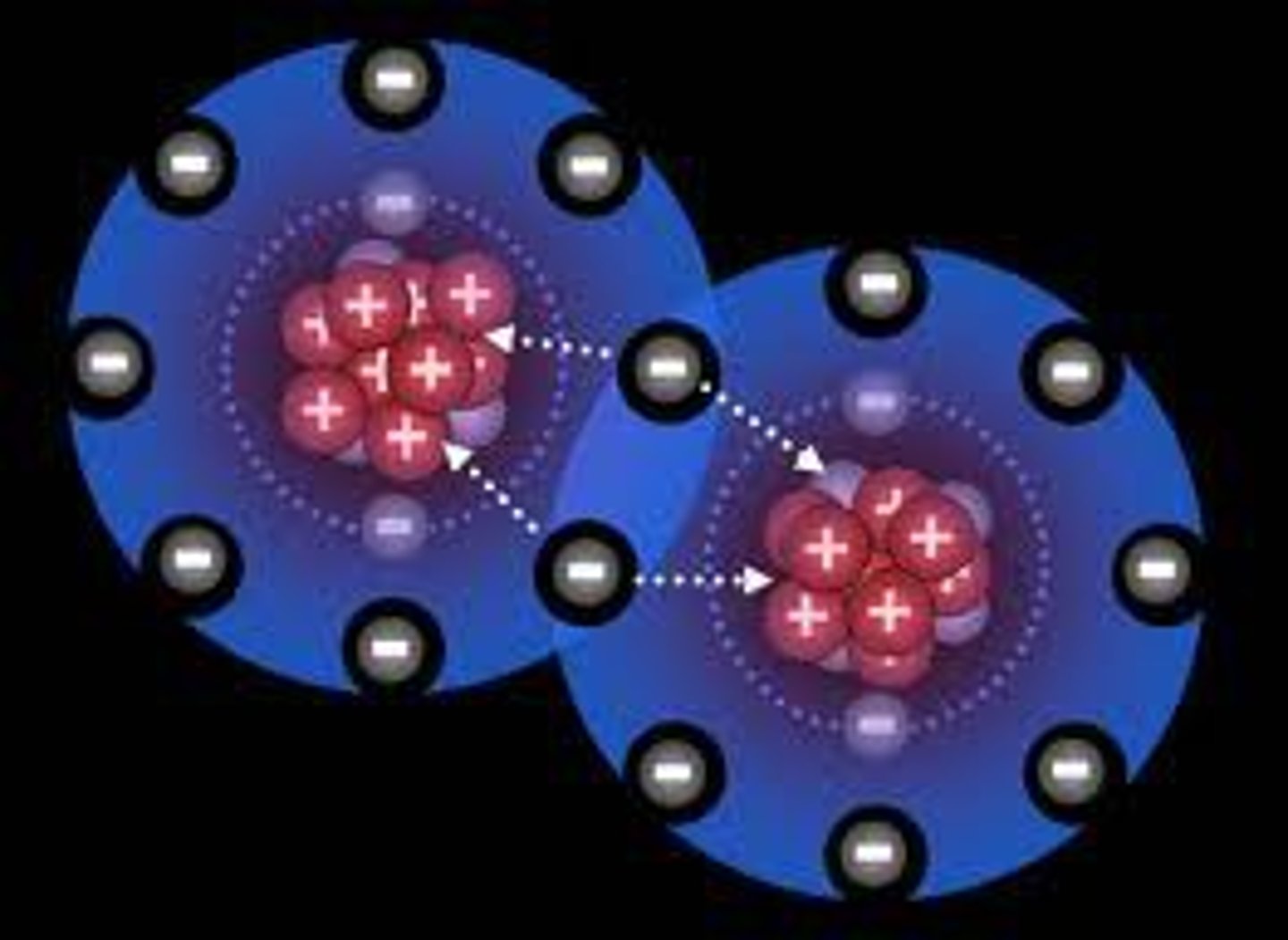
What are the two main types of chemical bonds and how do they differ?
Ionic Bonds: Transfer of electrons from a metal to a nonmetal.
Covalent Bonds: Sharing of electrons between two nonmetals.
What are reactants and products in a chemical reaction?
Reactants are the starting substances that undergo chemical change.
Products are the new substances formed after the reaction.
Law of Conservation of Mass: Mass remains constant during a chemical reaction.
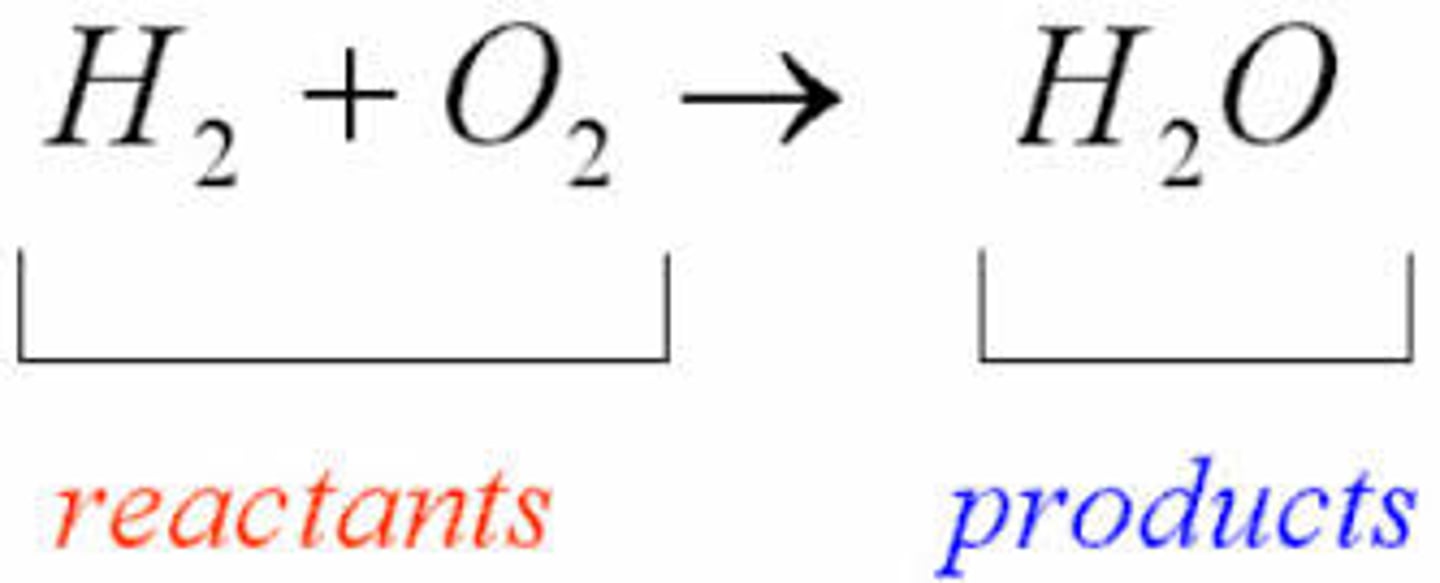
How do you balance a chemical equation?
Adjust the coefficients to ensure the same number of atoms on both sides of the equation.
The goal is to conserve atoms and maintain mass balance.
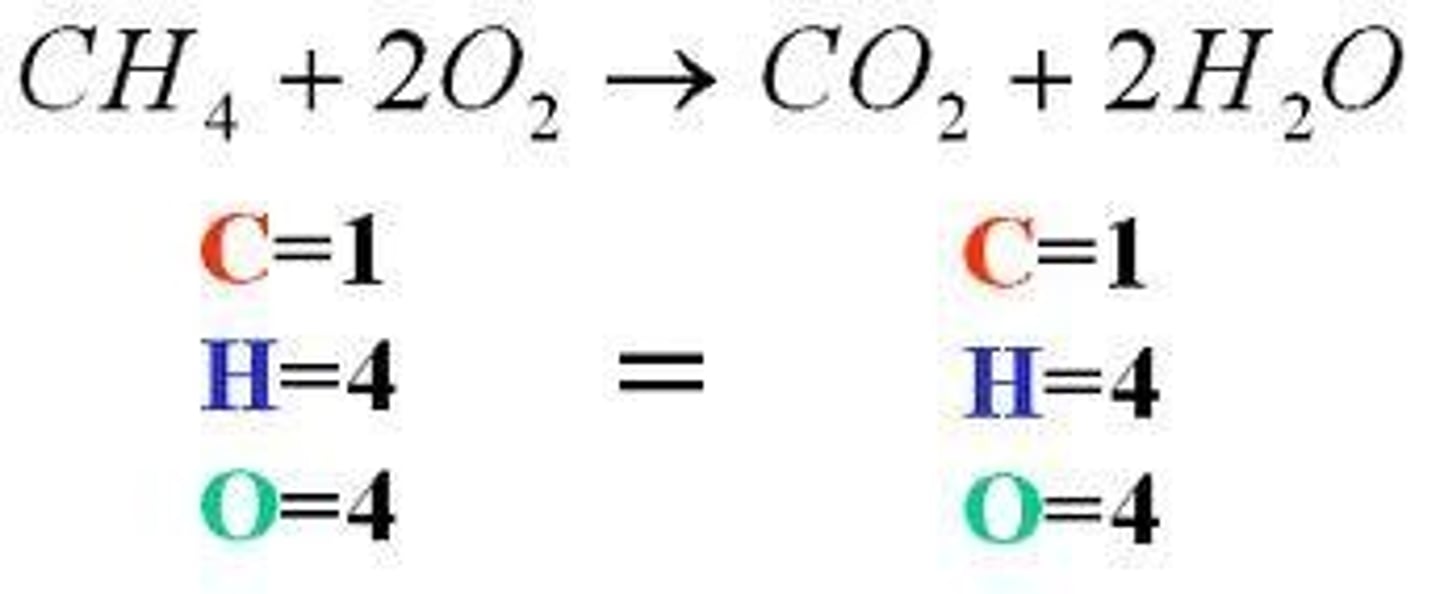
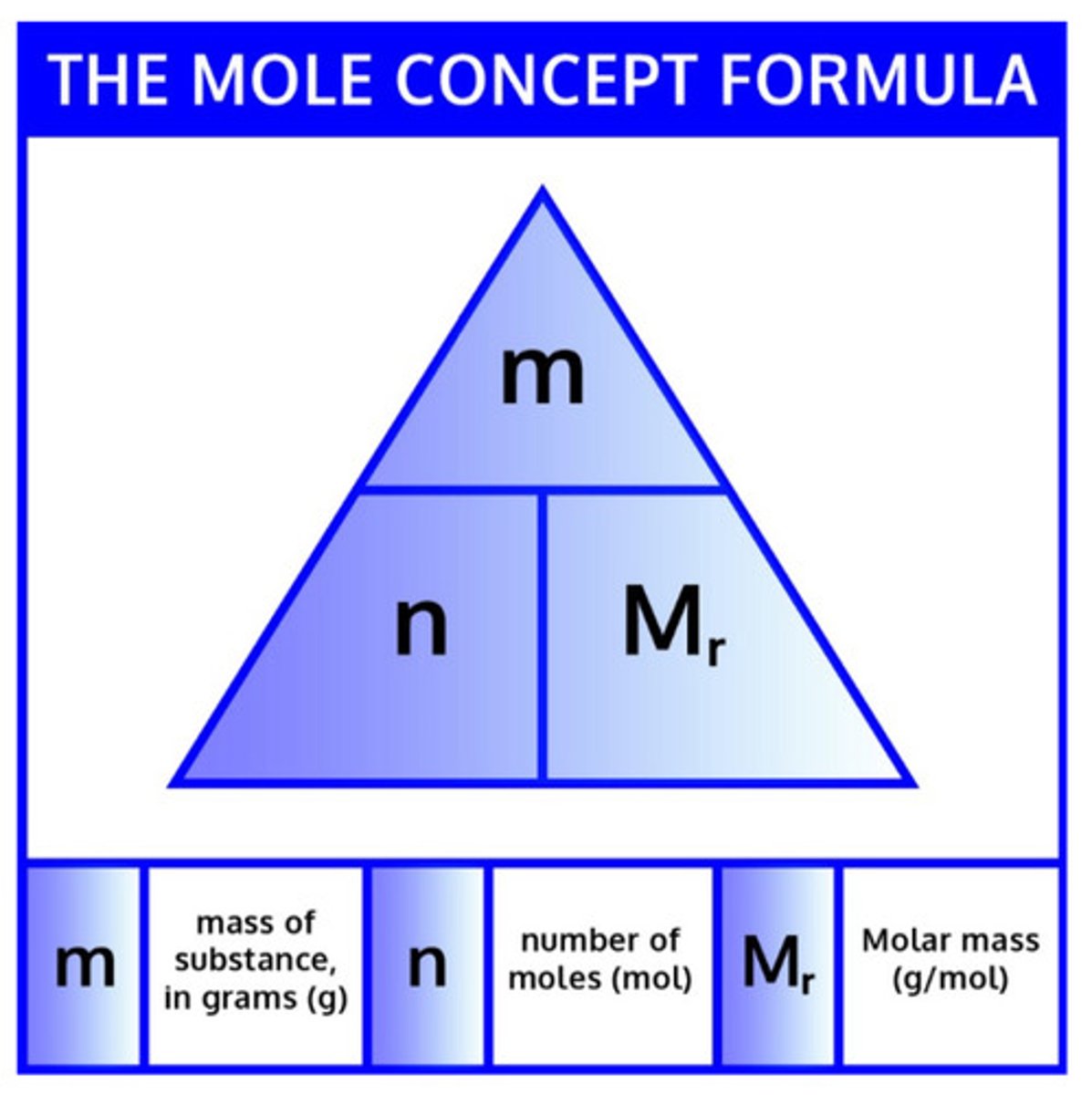
What is the mole concept and how is it used in chemical reactions?
1 mole = 6.022 × 10²³ molecules (Avogadro’s Number)
The mole concept is used to calculate the amounts of reactants and products in chemical reactions.
What factors influence the rate of chemical reactions?
Temperature: Higher temperature = faster reaction
Concentration: Higher concentration = more collisions
Surface Area: More surface area = faster reaction
Pressure: Higher pressure (for gases) = faster reaction

What does Le Chatelier's Principle state about chemical equilibrium?
When a system at equilibrium is disturbed, it shifts to counteract the change and restore equilibrium.
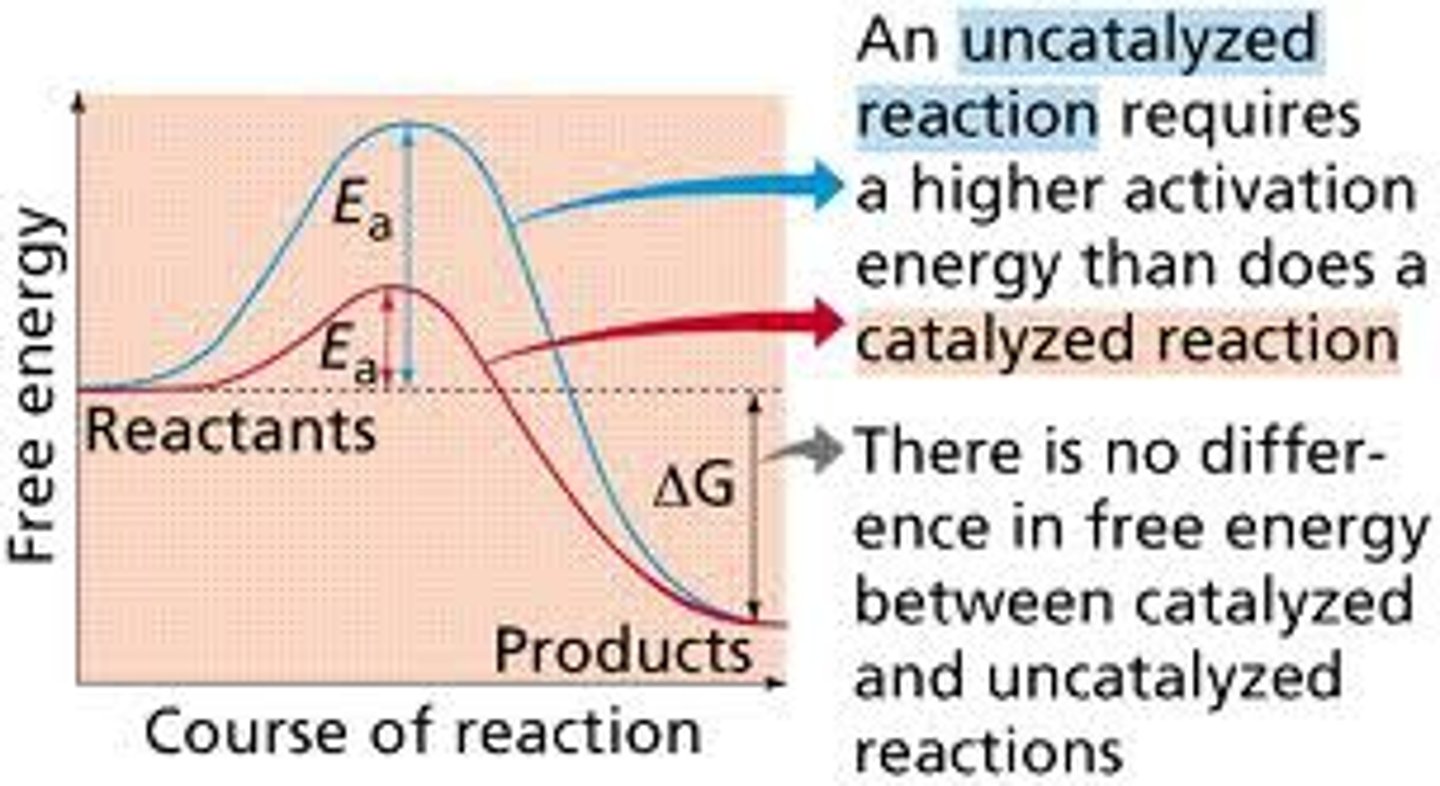
How do catalysts and enzymes affect chemical reactions?
Catalysts: Speed up reactions by lowering activation energy.
Enzymes: Biological catalysts that speed up biochemical reactions.
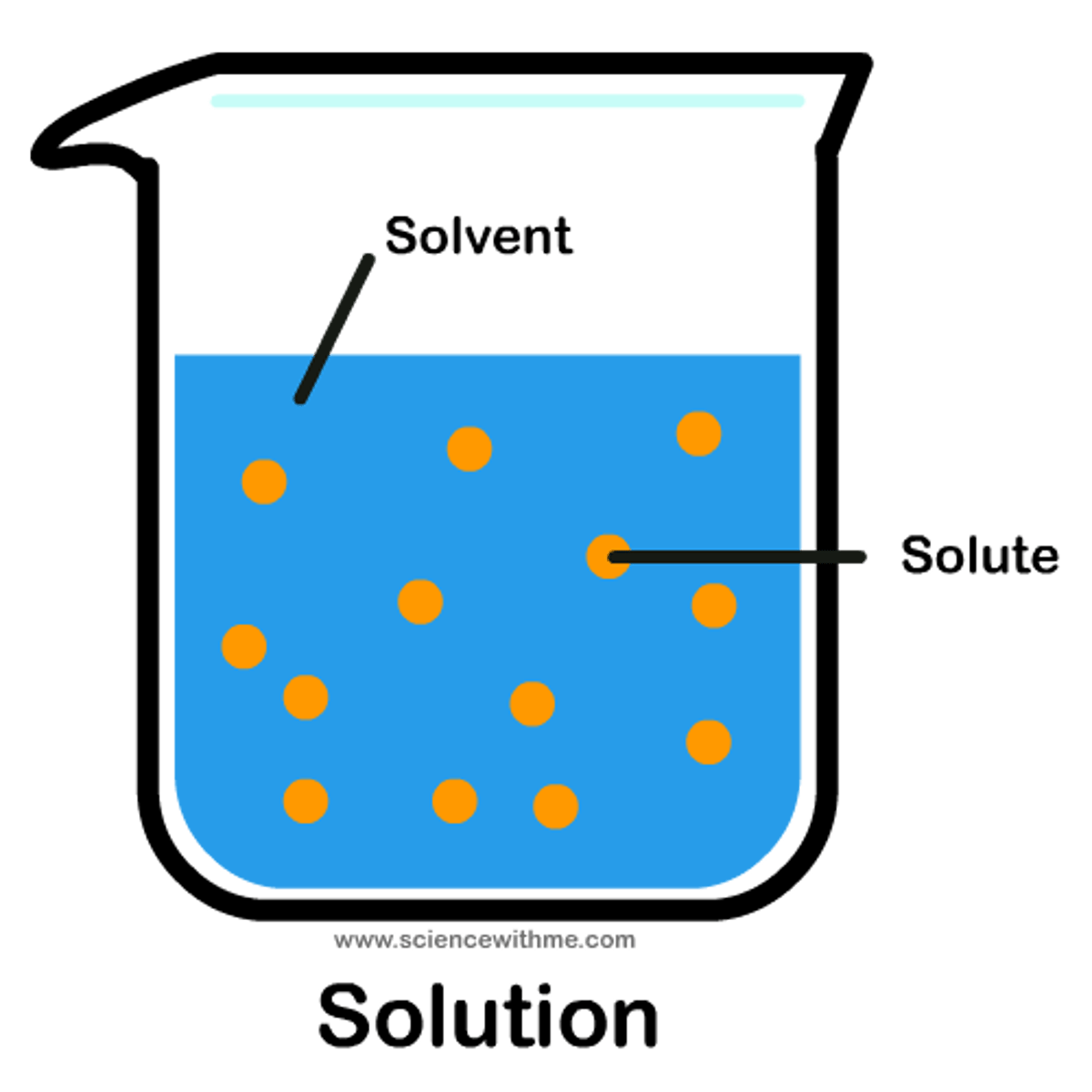
What are solvents and solutes?
Solvent: Substance that dissolves other substances (e.g., water).
Solute: Substance that is dissolved (e.g., salt in water).
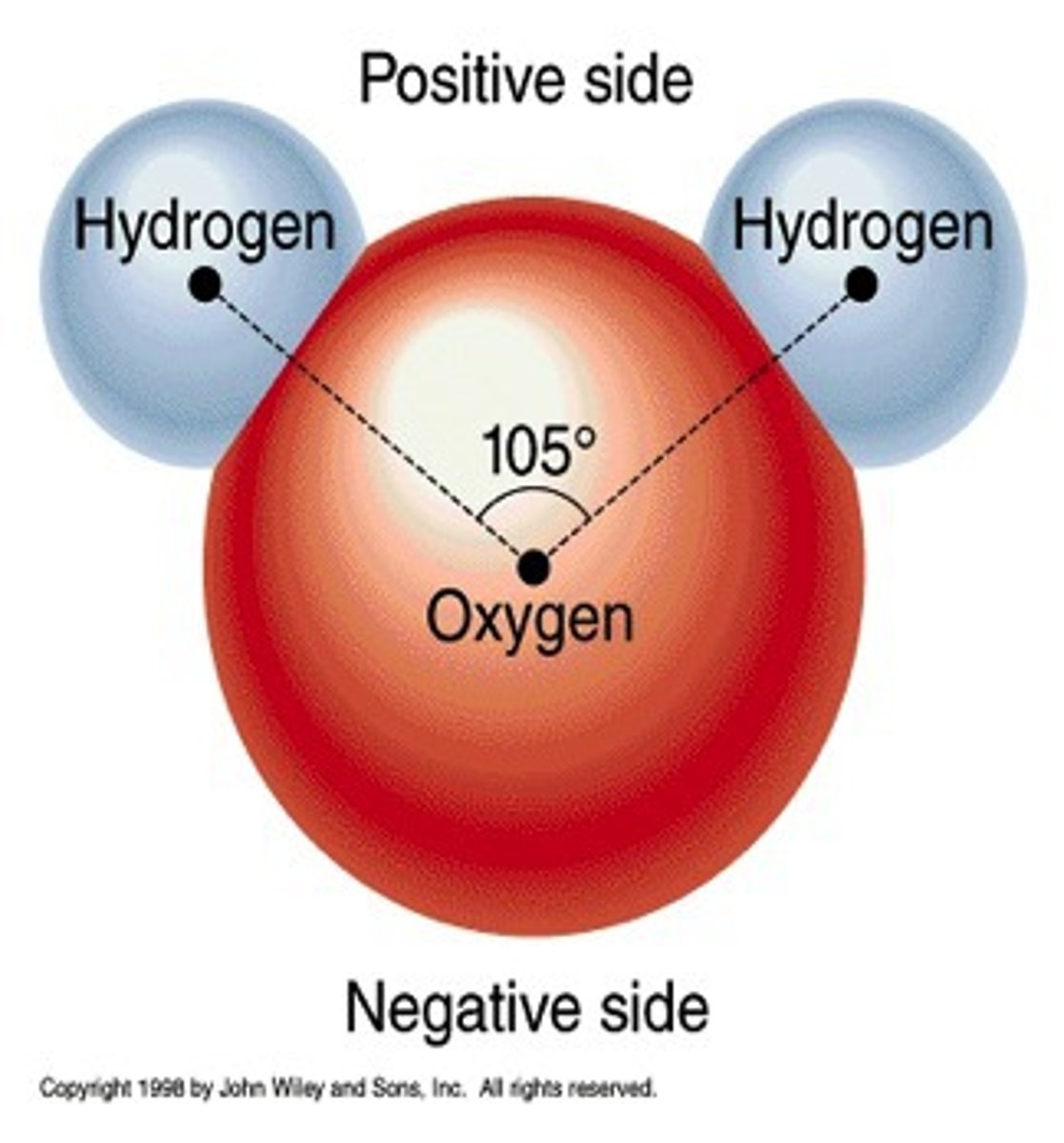
How does the polarity of water influence its ability as a solvent?
Water is a polar molecule with partial positive and negative charges.
"Like dissolves like" – Polar substances dissolve in water.
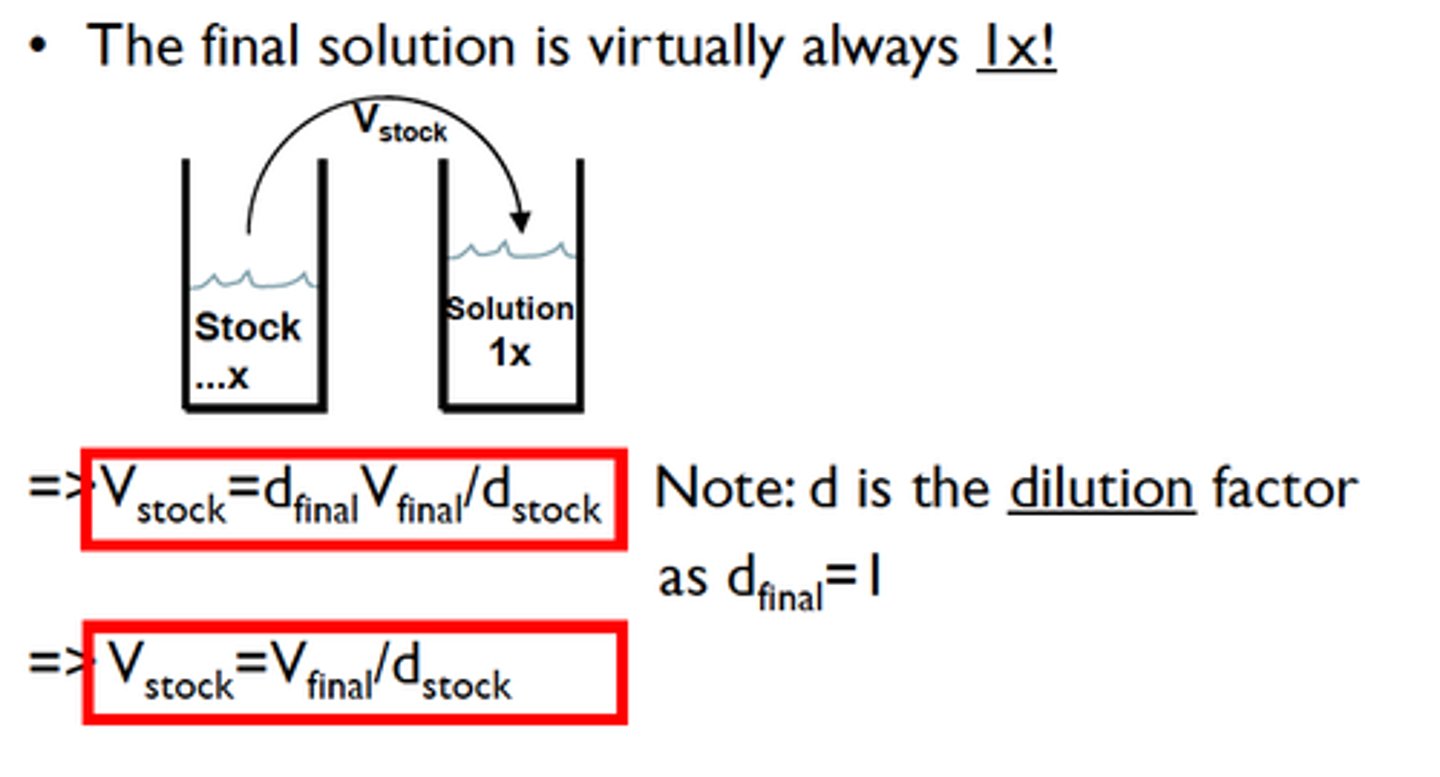
What is the difference between concentration and dilution in solutions?
Concentration: The amount of solute in a solution.
Dilution: The process of adding solvent to reduce the concentration of the solute.
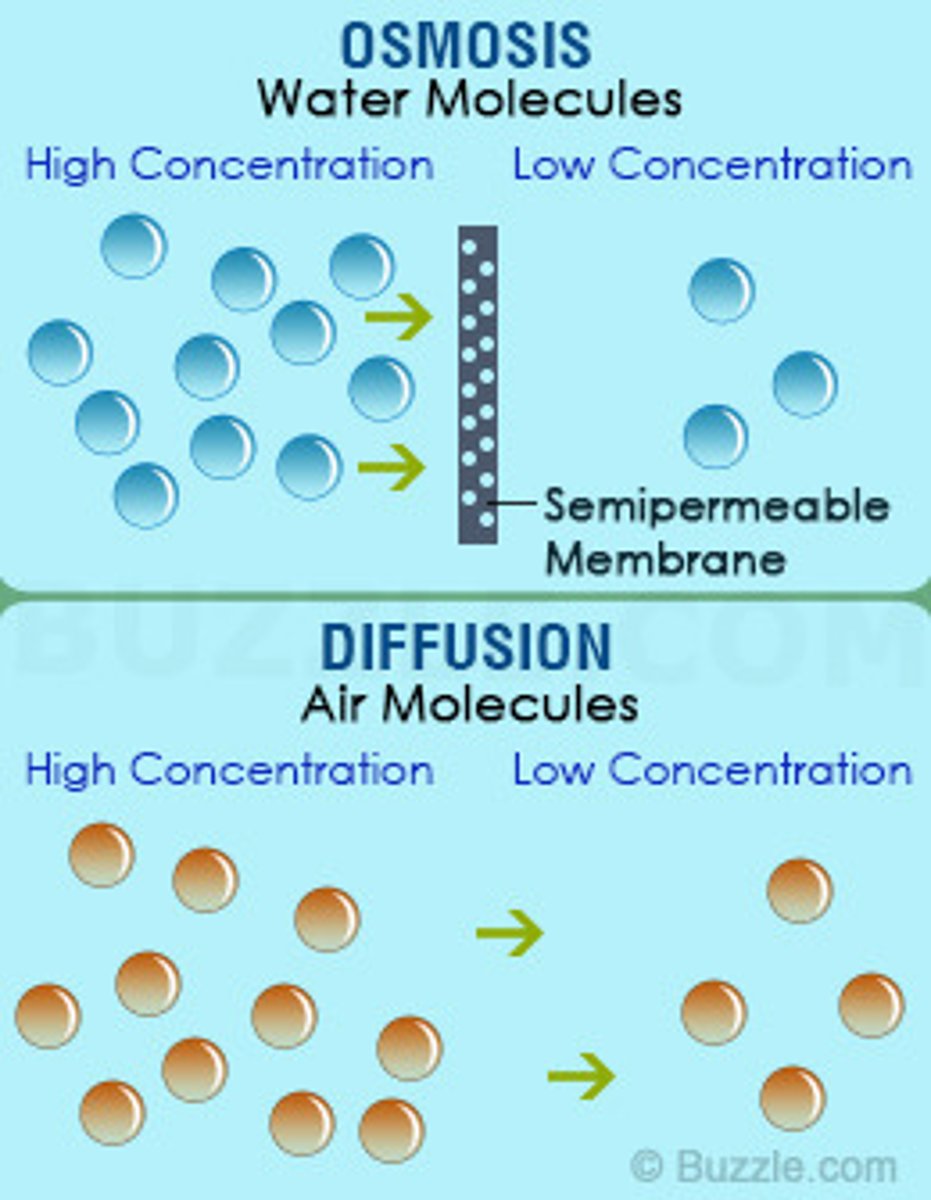
What are diffusion and osmosis?
Diffusion: Movement of particles from high to low concentration.
Osmosis: Movement of water across a semipermeable membrane from high to low concentration.
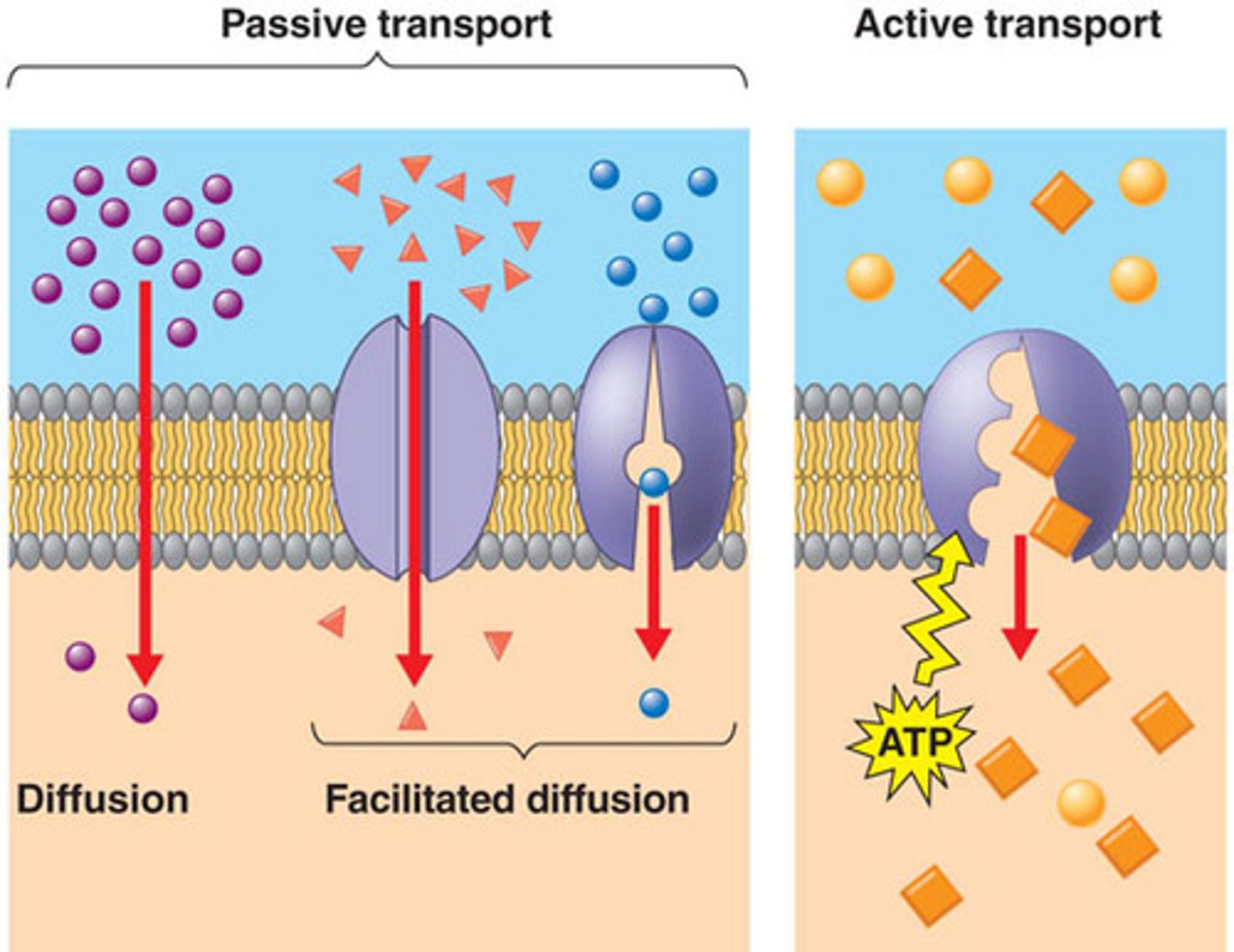
What is the difference between passive and active transport?
Passive Transport: Does not require energy (e.g., diffusion, osmosis).
Active Transport: Requires energy to move substances against the concentration gradient (e.g., pumps, endocytosis).
Define acids, bases, and the pH scale.
Acid: Releases H⁺ ions, pH < 7.
Base: Releases OH⁻ ions, pH > 7.
Neutral pH: 7 (pure water).

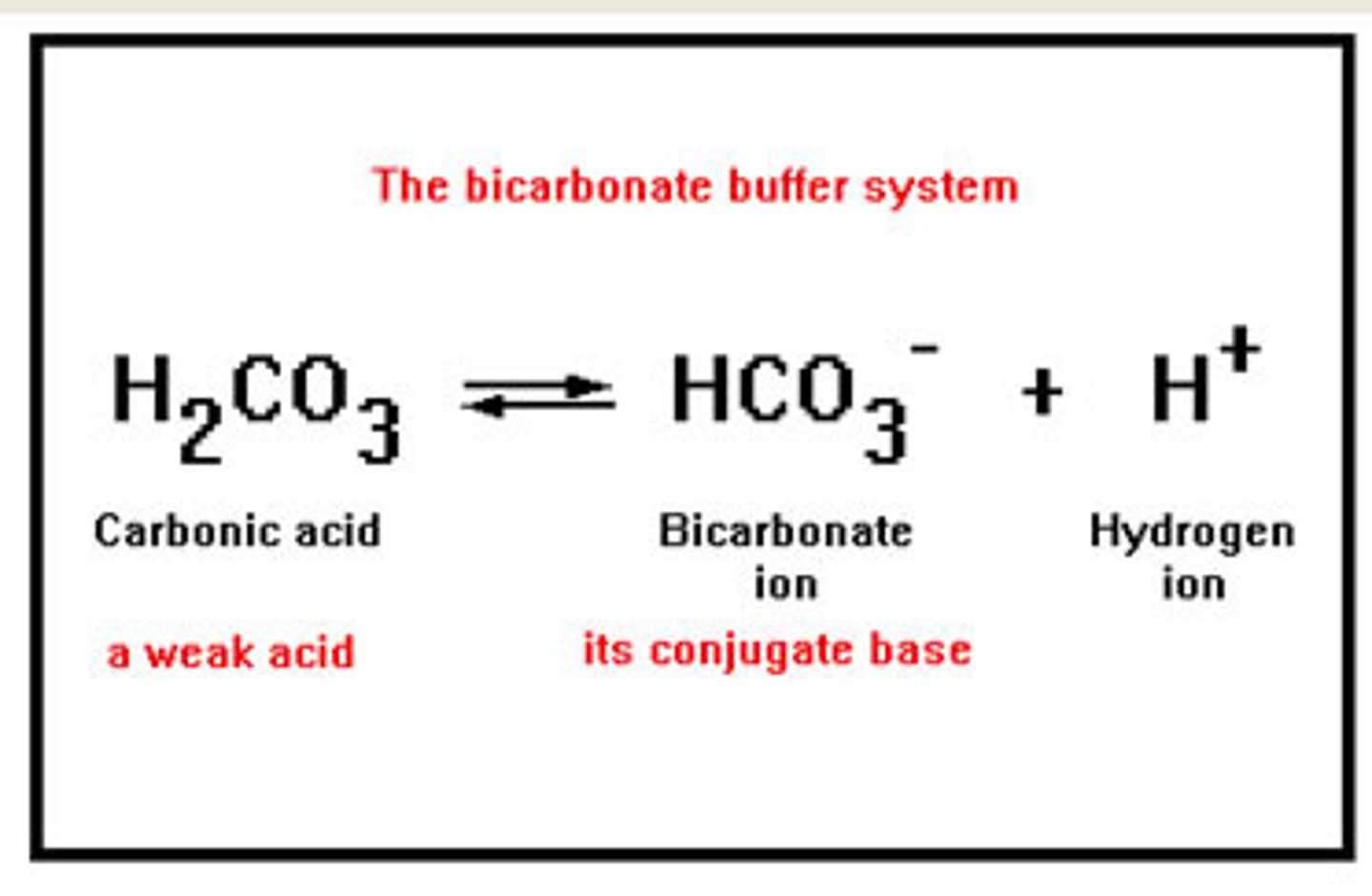
What role do biological buffers play in maintaining pH?
Biological buffers maintain a stable pH in the body by neutralizing excess acids or bases (e.g., bicarbonate in blood).
What happens in a neutralization reaction?
An acid reacts with a base to form salt and water.
Example: HCl + NaOH → NaCl + H₂O
